-
抗体試薬
- フローサイトメトリー用試薬
-
ウェスタンブロッティング抗体試薬
- イムノアッセイ試薬
-
シングルセル試薬
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD® OMICS-One Protein Panels
-
細胞機能評価のための試薬
-
顕微鏡・イメージング用試薬
-
細胞調製・分離試薬
-
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD® OMICS-One Protein Panels
- Japan (Japanese)
-
Change country/language
Old Browser
Looks like you're visiting us from United States.
Would you like to stay on the current country site or be switched to your country?
BD Transduction Laboratories™ Purified Mouse Anti-IKKγ
クローン 54/IKKγ/NEMO (RUO)
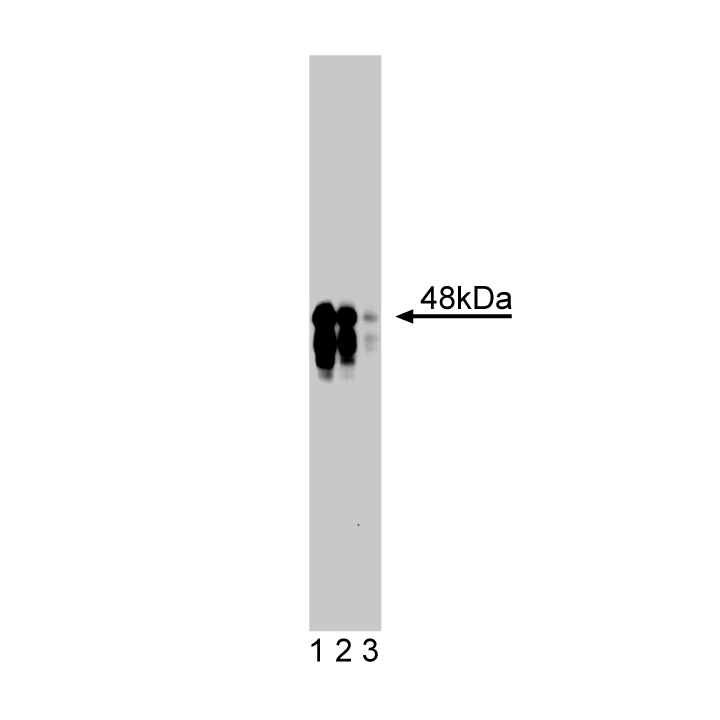


Western blot analysis of IKKγ on rat kidney lysate. Lane 1: 1:250, lane 2: 1:500, lane 3: 1:1000 dilution of anti-IKKγ antibody.

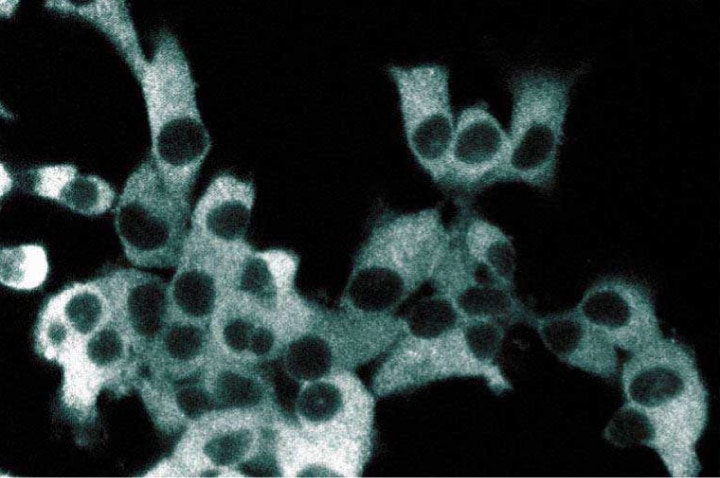
Immunofluorescent staining of ES2 cells with anti-IKKγ antibody.


BD Transduction Laboratories™ Purified Mouse Anti-IKKγ

BD Transduction Laboratories™ Purified Mouse Anti-IKKγ

Regulatory Statusの凡例
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation and Storage
推奨アッセイ手順
Western blot: Please refer to http://www.bdbiosciences.com/pharmingen/protocols/Western_Blotting.shtml .
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
In most cells, NF-κB is sequestered in an inactive cytoplasmic form via interactions with the inhibitory proteins IκBα, IκBβ, and IκBε. Cell stimulation induces the release, activation, and nuclear translocation of NF-κB. Release of NF-κB results from the phosphorylation and subsequent proteolytic degradation of the IκB proteins. Two cytokine-inducible IκB kinases (IKKα and IKKβ) phosphorylate and target the IκB proteins for degradation by the ubiquitin pathway. These kinases are components of a 700-900 kDa multisubunit complex that also contains NF-κB/RelA, IκBα, MEKK1, NIK, IKAP, and IKKγ/NEMO (NF-κB essential modulator). IKKγ contains two coiled-coil domains and a leucine zipper which allow it to form dimers and trimers that interact directly with IKKβ. IKKγ, essential for IKKα/IKKβ activation of NF-κB, is located functionally and physically upstream of these subunits. In addition, IKKγ enables the HTLV-1 Tax oncoprotein to interact with IKKβ, resulting in constitutive activation of NF-κB and maintenance of cellular transformation. Thus, IKKγ is an essential element of the IκB complex and an adaptor that mediates stable formation of Tax-IKK complexes.
Development References (5)
-
Chu ZL, Shin YA, Yang JM, DiDonato JA, Ballard DW. IKKgamma mediates the interaction of cellular IkappaB kinases with the tax transforming protein of human T cell leukemia virus type 1. J Biol Chem. 1999; 274(22):15297-15300. (Biology). View Reference
-
Jin DY, Giordano V, Kibler KV, Nakano H, Jeang KT. Role of adapter function in oncoprotein-mediated activation of NF-kappaB. Human T-cell leukemia virus type I Tax interacts directly with IkappaB kinase gamma. J Biol Chem. 1999; 274(25):17402-17405. (Biology). View Reference
-
Rothwarf DM, Zandi E, Natoli G, Karin M. IKK-gamma is an essential regulatory subunit of the IkappaB kinase complex. Nature. 1998; 395(6699):297-300. (Biology). View Reference
-
Weil R, Schwamborn K, Alcover A, Bessia C, Di Bartolo V, Israël A. Induction of the NF-kappaB cascade by recruitment of the scaffold molecule NEMO to the T cell receptor. Immunity. 2003; 18(1):13-26. (Clone-specific: Immunofluorescence, Immunoprecipitation, Western blot). View Reference
-
Yamaoka S, Courtois G, Bessia C. Complementation cloning of NEMO, a component of the IkappaB kinase complex essential for NF-kappaB activation. Cell. 1998; 93(7):1231-1240. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.