-
抗体試薬
- フローサイトメトリー用試薬
-
ウェスタンブロッティング抗体試薬
- イムノアッセイ試薬
-
シングルセル試薬
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD® OMICS-One Protein Panels
-
細胞機能評価のための試薬
-
顕微鏡・イメージング用試薬
-
細胞調製・分離試薬
-
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD® OMICS-One Protein Panels
- Japan (Japanese)
-
Change country/language
Old Browser
Looks like you're visiting us from United States.
Would you like to stay on the current country site or be switched to your country?
BD Pharmingen™ Alexa Fluor® 488 Mouse anti-E-Cadherin
クローン 36/E-Cadherin (RUO)

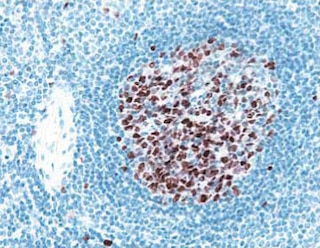
Immunofluorescent staining of mouse small intestine (left panel) and human breast adenocarcinoma (middle panel). Mouse small intestine and MCF-7 cells (ATCC HTB-22) were processed and stained according to the Recommended Assay Procedures for tissue sections and cultured cells, respectively. Both specimens were stained with Alexa Fluor® 488 Mouse anti-E-Cadherin monoclonal antibody (pseudo-colored green) and counter-stained with Hoechst 33342 (pseudo-colored blue). The images were captured on a BD Pathway™ 435 Bioimager System with a 20x objective and merged using BD Attovision™ software.
Right Panel: Western blot analysis of E-Cadherin using Purified Mouse Anti-E-Cadherin (Cat. No. 610181/610182). E-Cadherin is observable at 120kDa. Left Panel: A431 lysate (ATCC CRL-1555; human epithelial carcinoma) was blotted at 1:10000 & 1:20000 (Lanes 1 & 2 respectively; 30 second exposure). Middle Left Panel: 293F control lysate was blotted at 1:250 & 1:500 (Lanes 1 & 2 respectively; 30 second exposure). Middle Right Panel: 293F cells transfected with human E-Cadherin (CDH1) was blotted at 1:2500 & 1: 5000 (Lanes 1 & 2 respectively; 5 second exposure). Right Panel: 293 cells transfected with human P-Cadherin (CDH3) was blotted using Purified Mouse Anti-E-Cadherin (Cat. No. 610181/610182) at 1:2500 & 1: 5000 (Lanes 1 & 2 respectively; 5 second exposure).

.png)
Immunofluorescent staining of mouse small intestine (left panel) and human breast adenocarcinoma (middle panel). Mouse small intestine and MCF-7 cells (ATCC HTB-22) were processed and stained according to the Recommended Assay Procedures for tissue sections and cultured cells, respectively. Both specimens were stained with Alexa Fluor® 488 Mouse anti-E-Cadherin monoclonal antibody (pseudo-colored green) and counter-stained with Hoechst 33342 (pseudo-colored blue). The images were captured on a BD Pathway™ 435 Bioimager System with a 20x objective and merged using BD Attovision™ software.
Right Panel: Western blot analysis of E-Cadherin using Purified Mouse Anti-E-Cadherin (Cat. No. 610181/610182). E-Cadherin is observable at 120kDa. Left Panel: A431 lysate (ATCC CRL-1555; human epithelial carcinoma) was blotted at 1:10000 & 1:20000 (Lanes 1 & 2 respectively; 30 second exposure). Middle Left Panel: 293F control lysate was blotted at 1:250 & 1:500 (Lanes 1 & 2 respectively; 30 second exposure). Middle Right Panel: 293F cells transfected with human E-Cadherin (CDH1) was blotted at 1:2500 & 1: 5000 (Lanes 1 & 2 respectively; 5 second exposure). Right Panel: 293 cells transfected with human P-Cadherin (CDH3) was blotted using Purified Mouse Anti-E-Cadherin (Cat. No. 610181/610182) at 1:2500 & 1: 5000 (Lanes 1 & 2 respectively; 5 second exposure).

Immunofluorescent staining of mouse small intestine (left panel) and human breast adenocarcinoma (middle panel). Mouse small intestine and MCF-7 cells (ATCC HTB-22) were processed and stained according to the Recommended Assay Procedures for tissue sections and cultured cells, respectively. Both specimens were stained with Alexa Fluor® 488 Mouse anti-E-Cadherin monoclonal antibody (pseudo-colored green) and counter-stained with Hoechst 33342 (pseudo-colored blue). The images were captured on a BD Pathway™ 435 Bioimager System with a 20x objective and merged using BD Attovision™ software.
Right Panel: Western blot analysis of E-Cadherin using Purified Mouse Anti-E-Cadherin (Cat. No. 610181/610182). E-Cadherin is observable at 120kDa. Left Panel: A431 lysate (ATCC CRL-1555; human epithelial carcinoma) was blotted at 1:10000 & 1:20000 (Lanes 1 & 2 respectively; 30 second exposure). Middle Left Panel: 293F control lysate was blotted at 1:250 & 1:500 (Lanes 1 & 2 respectively; 30 second exposure). Middle Right Panel: 293F cells transfected with human E-Cadherin (CDH1) was blotted at 1:2500 & 1: 5000 (Lanes 1 & 2 respectively; 5 second exposure). Right Panel: 293 cells transfected with human P-Cadherin (CDH3) was blotted using Purified Mouse Anti-E-Cadherin (Cat. No. 610181/610182) at 1:2500 & 1: 5000 (Lanes 1 & 2 respectively; 5 second exposure).
.png)

BD Pharmingen™ Alexa Fluor® 488 Mouse anti-E-Cadherin
.png)
Regulatory Statusの凡例
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation and Storage
推奨アッセイ手順
For tissue sections
1. Harvest target organs or tissues and flush with PBS.
2. Place the tissues in cassettes and fix with 10% neutral buffered formalin (Fisher SF100-4) for 16 hrs.
3. Remove the fixative and proceed with processing and embedding in paraffin using standard protocols.*
4. Cut paraffin embedded tissue sections (5 µm) using a microtome, adhere sections onto colorfrost slides (Fisher 12-550-17), and allow them to air dry.*
5. Deparaffinize and re-hydrate the sections.*
6. Treat with BD Retrievagen A solution (Cat. No.550524) by heating the slides in a pressure cooker at 121°C for 15 minutes at 17 psi.*
7. Allow the slides to cool to room temperature in the Retrievagen A, rinse the slides with tap water, and store in PBS prior to antibody staining.
8. Sections can be simultaneously stained with a cocktail of multiple antibodies at pre-optimized concentration, for 2 hours at room temperature.
9. Wash the sections in 1× PBS, label cellular DNA with 2 µg/ml Hoechst 33342 (Cat. No. 561908) for 30 minutes, and wash with 1× PBS.
10. Place cover slips on the sections using Aqua-Mount (Lerner Labs 13800).
11. View and analyze the samples on an appropriate imaging instrument, such as the BD Pathway™ 435 high-content bioimager system.
* for more details please see http://www.bdbiosciences.com/research/cellularimaging/resources/index.jsp
For cultured cells
1. Seed the cells in appropriate culture medium at an appropriate cell density in a 96-well Imaging Plate and culture overnight to 48 hours.
2. Remove the culture medium from the wells, and wash (one to two times) with 100 μl of 1× PBS.
3. Fix the cells by adding 100 µl of fresh 3.7% Formaldehyde in PBS or BD Cytofix™ fixation buffer (Cat. No. 554655) to each well and incubating for 10 minutes at room temperature (RT).
4. Remove the fixative from the wells, and wash the wells (one to two times) with 100 μl of 1× PBS.
5. Permeabilize the cells using either cold methanol (a), Triton™ X-100 (b), or Saponin (c):
a. Add 100 µl of -20°C 90% methanol or -20°C BD Phosflow™ Perm Buffer III (Cat. No. 558050) to each well and incubate for 5 minutes at RT.
b. Add 100 µl of 0.1% Triton™ X-100 to each well and incubate for 5 minutes at RT.
c. Add 100 µl of 1× Perm/Wash buffer (Cat. No. 554723) to each well and incubate for 15 to 30 minutes at RT. Continue to use 1× Perm/Wash buffer for all subsequent wash and dilutions steps.
6. Remove the permeabilization buffer from the wells, and wash one to two times with 100 μl of appropriate buffer (either 1× PBS or 1× Perm/Wash buffer, see step 5.c.).
7. Optional blocking step: Remove the wash buffers, and block the cells by adding 100 µl of blocking buffer BD Pharmingen™ Stain Buffer (FBS) (Cat. No. 554656) or 3% FBS in appropriate dilution buffer to each well and incubating for 15 to 30 minutes at RT.
8. Dilute the antibody to its optimal working concentration in appropriate dilution buffer. Titrate purified (unconjugated) antibodies and second-step reagents to determine the optimal concentration. If using a Bioimaging Certified antibody conjugate, dilute it 1:10.
9. Add 50 µl of diluted antibody per well and incubate for 60 minutes at RT. Incubate in the dark if using fluorescently labeled antibodies.
10. Remove the antibody, and wash the wells three times with 100 μl of wash buffer. An optional detergent wash (100 μl of 0.05% Tween in 1× PBS) can be included prior to the regular wash steps.
11. If the antibody being used is fluorescently labeled, then move to step 12. Otherwise, if using a purified unlabeled antibody, repeat steps 8 to 10 with a fluorescently labeled second-step reagent to detect the purified antibody.
12. After the final wash, counter-stain the nuclei by adding 100 μl of a 2 μg/ml solution of Hoechst 33342 (Cat. No. 561908) in 1× PBS to each well at least 15 minutes before imaging.
13. View and analyze the cells on an appropriate imaging instrument. Recommended filters for the BD Pathway™ instruments are:
Instrument Excitation Emission Dichroic
BD Pathway 855 488/10 515 LP Fura/FITC
BD Pathway 435 482/35 536/40 FF506
Product Notices
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- This reagent has been pre-diluted for use at the recommended Volume per Test when following the Recommended Assay Procedure. A Test is typically ~10,000 cells cultured in a well of a 96-well imaging plate.
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Alexa Fluor® 488 fluorochrome emission is collected at the same instrument settings as for fluorescein isothiocyanate (FITC).
- Alexa Fluor® is a registered trademark of Molecular Probes, Inc., Eugene, OR.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Triton is a trademark of the Dow Chemical Company.
- Species cross-reactivity detected in product development may not have been confirmed on every format and/or application.
関連製品


E-Cadherin is a 120-kDa transmembrane glycoprotein that is localized in the adherens junctions of epithelial cells. There it interacts with the cytoskeleton through the associated cytoplasmic catenin proteins. In addition to being a calcium-dependent adhesion molecule, E-Cadherin is also a critical regulator of epithelial junction formation. Its association with catenins is necessary for cell-cell adhesion. These E-cadherin/catenin complexes associate with cortical actin bundles at both the zonula adherens and the lateral adhesion plaques. Tyrosine phosphorylation can disrupt these complexes, leading to changes in cell adhesion properties. E-Cadherin expression is often down-regulated in highly invasive, poorly differentiated carcinomas. Increased expression of E-Cadherin in these cells reduces invasiveness. Thus, loss of expression or function of E-Cadherin appears to be an important step in tumorigenic progression. The 36/E-Cadherin monoclonal antibody recognizes the cytoplasmic domain of E-Cadherin, regardless of phosphorylation status. The peptide immunogen was generated from human E-Cadherin aa. 735-883.
Note: Investigators are advised that this antibody has some degree of cross-reactivity to P-Cadherin.
Development References (5)
-
Jaksits S, Kriehuber E, Charbonnier AS, Rappersberger K, Stingl G, Maurer D. CD34+ cell-derived CD14+ precursor cells develop into Langerhans cells in a TGF-beta 1-dependent manner. J Immunol. 1999; 163(9):4869-4877. (Clone-specific: Flow cytometry). View Reference
-
Miyoshi K, Shillingford JM, Smith GH, et al. Signal transducer and activator of transcription (Stat) 5 controls the proliferation and differentiation of mammary alveolar epithelium. J Cell Biol. 2001; 155(4):531-542. (Clone-specific: Immunohistochemistry). View Reference
-
Sheibani N, Sorenson CM, Frazier WA. Differential modulation of cadherin-mediated cell-cell adhesion by platelet endothelial cell adhesion molecule-1 isoforms through activation of extracellular regulated kinases. Mol Biol Cell. 2000; 11(8):2793-2802. (Clone-specific: Immunofluorescence, Western blot). View Reference
-
Takeichi M. The cadherins: cell-cell adhesion molecules controlling animal morphogenesis.. Development. 1988; 102(4):639-55. (Biology). View Reference
-
Weng Z, Xin M, Pablo L, et al. Protection against anoikis and down-regulation of cadherin expression by a regulatable beta-catenin protein. J Biol Chem. 2002; 277(21):18677-18686. (Clone-specific: Immunofluorescence, Immunoprecipitation, Western blot). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.