-
抗体試薬
- フローサイトメトリー用試薬
-
ウェスタンブロッティング抗体試薬
- イムノアッセイ試薬
-
シングルセル試薬
- BD® AbSeq Assay | シングルセル試薬
- BD Rhapsody™ Accessory Kits | シングルセル試薬
- BD® Single-Cell Multiplexing Kit | シングルセル試薬
- BD Rhapsody™ Targeted mRNA Kits | シングルセル試薬
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit | シングルセル試薬
- BD Rhapsody™ TCR/BCR Profiling Assays (VDJ Assays) | シングルセル試薬
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
-
細胞機能評価のための試薬
-
顕微鏡・イメージング用試薬
-
細胞調製・分離試薬
-
- BD® AbSeq Assay | シングルセル試薬
- BD Rhapsody™ Accessory Kits | シングルセル試薬
- BD® Single-Cell Multiplexing Kit | シングルセル試薬
- BD Rhapsody™ Targeted mRNA Kits | シングルセル試薬
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit | シングルセル試薬
- BD Rhapsody™ TCR/BCR Profiling Assays (VDJ Assays) | シングルセル試薬
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- Japan (Japanese)
-
Change country/language
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

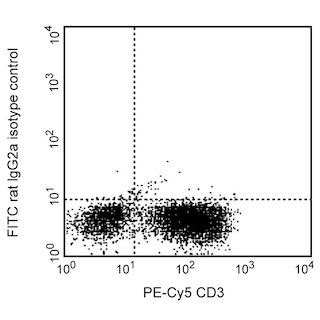
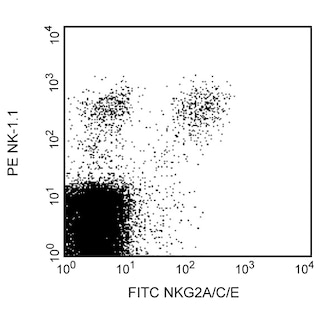
Expression of NKG2A/C/E on mouse splenic NK cells. C57BL/6 splenocytes were simultaneously stained with PE Mouse Anti-Mouse NK-1.1 (Cat. no. 557391) and FITC Rat Anti-Mouse NKG2A/C/E mAb clone 20d5 (Cat. no. 562063) (figure right panel) or a FITC isotype control clone R35-95 (Cat. no. 553929) (figure left panel). Flow cytometry was performed on a BD FACSCalibur™ flow cytometry system.
.png)

BD Pharmingen™ FITC Rat Anti-Mouse NKG2A/C/E
.png)
Regulatory Statusの凡例
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation and Storage
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
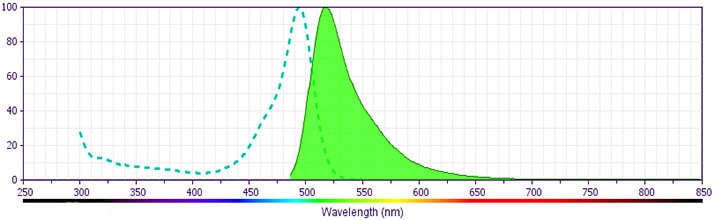
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
関連製品

The 20d5 monoclonal antibody specifically recognizes NKG2A, NKG2C, and NKG2E (also known as CD159a, CD159c, and CD159e which are encoded by Klrc1, Klrc2, and Klrc3, respectively) on a subset of NK and NK-T cells in most strains tested (eg, AKR/J, BALB/c, C3H/He, C57BL/6, CBA/J, DBA/1, FVB/N, 129/Sv, NOD, SWR, and most DBA/2 substrains, but not DBA/2J). The NKG2 molecules are a family of lectin-like receptors that form heterodimers with CD94 on the surface of NK cells. DBA/2J mice do not express CD94, and the lack of CD94 is responsible for the absence of NKG2 expression in this substrain. NKG2 receptors are also expressed on CD8+ T lymphocytes activated in vivo and in vitro. The heterodimers of CD94 with NKG2A, C, or E recognize Qa-1, a nonclassical MHC class I antigen, presenting the Qdm peptide. Studies of CD94/NKG2 heterodimers on human NK cells have demonstrated that the NKG2 components mediate signal transduction for the receptor, with NKG2A being inhibitory and NKG2C being stimulatory. The CD94/NKG2E heterodimer is also thought to be stimulatory. The mouse NKG2A molecule contains two intracytoplasmic sequences that resemble the ITIM (Immunoreceptor Tyrosine- based Inhibitory Motif) consensus sequence. NKG2A transcripts have been shown to be up to 20-fold more abundant than NKG2C and NKG2E mRNA in NK cells of adult mice. The CD94/NKG2 receptors show increased expression on neonatal NK cells compared to the Ly-49 MHC class I receptors, suggesting that CD94/NKG2 receptors and their ligand, Qa-1, may play a role in maintenance of self-tolerance in developing NK cells. The 20d5 antibody is useful for identification of NK cells expressing functional CD94/NKG2 receptors, in contrast to the non-functional CD94 expressed alone, and it blocks the binding of Qdm-complexed Qa-1b tetramers to CD94/NKG2-transfected CHO cells.
Development References (7)
-
Kubota A, Kubota S, Lohwasser S, Mager DL, Takei F. Diversity of NK cell receptor repertoire in adult and neonatal mice. Eur J Immunol. 1999; 163(1):212-216. (Biology). View Reference
-
Lohwasser S, Hande P, Mager DL, Takei F. Cloning of murine NKG2A, B and C: second family of C-type lectin receptors on murine NK cells. Eur J Immunol. 1999; 29(3):755-761. (Biology). View Reference
-
McMahon CW, Zajac AJ, Jamieson AM. Viral and bacterial infections induce expression of multiple NK cell receptors in responding CD8(+) T cells. J Immunol. 2002; 169(3):1444-1452. (Biology). View Reference
-
Vance RE, Jamieson AM, Cado D, Raulet DH. Implications of CD94 deficiency and monoallelic NKG2A expression for natural killer cell development and repertoire formation. Proc Natl Acad Sci U S A. 2002; 99(2):868-873. (Biology). View Reference
-
Vance RE, Jamieson AM, Raulet DH. Recognition of the class Ib molecule Qa-1(b) by putative activating receptors CD94/NKG2C and CD94/NKG2E on mouse natural killer cells. J Exp Med. 1999; 190(12):1801-1812. (Immunogen). View Reference
-
Vance RE, Kraft JR, Altman JD, Jensen PE, Raulet DH. Mouse CD94/NKG2A is a natural killer cell receptor for the nonclassical major histocompatibility complex (MHC) class I molecule Qa-1(b). J Exp Med. 1998; 188(10):1841-1848. (Biology). View Reference
-
Yokoyama WM. Natural killer cell receptors. Curr Opin Immunol. 1998; 10(3):298-305. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.