-
抗体試薬
- フローサイトメトリー用試薬
-
ウェスタンブロッティング抗体試薬
- イムノアッセイ試薬
-
シングルセル試薬
- BD® AbSeq Assay | シングルセル試薬
- BD Rhapsody™ Accessory Kits | シングルセル試薬
- BD® Single-Cell Multiplexing Kit | シングルセル試薬
- BD Rhapsody™ Targeted mRNA Kits | シングルセル試薬
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit | シングルセル試薬
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
-
細胞機能評価のための試薬
-
顕微鏡・イメージング用試薬
-
細胞調製・分離試薬
-
- BD® AbSeq Assay | シングルセル試薬
- BD Rhapsody™ Accessory Kits | シングルセル試薬
- BD® Single-Cell Multiplexing Kit | シングルセル試薬
- BD Rhapsody™ Targeted mRNA Kits | シングルセル試薬
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit | シングルセル試薬
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- Japan (Japanese)
-
Change country/language
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


APC Mouse Anti-Human Terminal-Deoxynucleotidyl Transferase (TdT)
Regulatory Statusの凡例
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation and Storage
The FITC conjugate is supplied as 12.5 µg in 1.0 mL (12.5 µg/mL) of PBS. The PE conjugate is supplied as 6.0 µg in 1.0 mL (6.0 µg/mL) of PBS. The APC conjugate is supplied as 12.5 µg in 0.5 mL (25 µg/mL) of PBS. PBS contains gelatin and 0.1% sodium azide. Vials should be stored at 2° to 8°C. Conjugated forms should not be frozen and should be protected from prolonged exposure to light. Each reagent is stable for the period shown on the bottle label when stored as directed.
Anti-TdT, clone E17-1519, is generated from the fusion of FO myeloma cells with spleen cells from BALB/c mice immunized with purified TdT enzyme. Anti–terminal-deoxynucleotidyl transferase (TdT) recognizes a 60-kilodalton (kd) polymerase, a nuclear enzyme that catalyzes the template-independent addition of nucleotides to single-stranded DNA primers. It has been reported that TdT is involved in the regulation or translocation or both of DNA and gene rearrangement during normal T- and B-cell development.
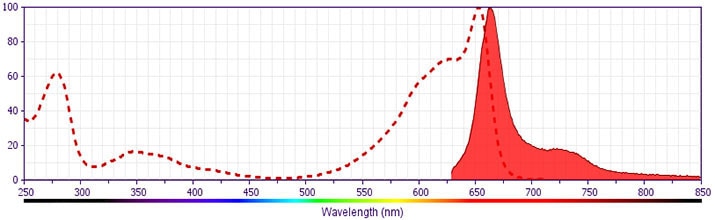
Development References (7)
-
Fuller SA, Phillips A, Coleman MS. Affinity purification and refined structural characterization of terminal deoxyribonucleotidyl transferase. Biochem J. 1985; 231:105-113. (Biology).
-
Gore SD, Kastan MB, Civin CI. Normal human bone marrow precursors that express terminal deoxynucleotidyl transferase include T-cell precursors and possible lymphoid stem cells.. Blood. 1991; 77(8):1681-90. (Biology). View Reference
-
Horvatinovich JM, Sparks SD, Borowitz MJ. Detection of terminal deoxynucleotidyl transferase by flow cytometry: a three color method. Cytometry. 1994; 18:228-230. (Biology).
-
Komori T, Okada A, Stewart V, Alt FW. Lack of N regions in antigen receptor variable regions of TdTdeficient lymphocytes. Science. 1993; 261:1171-1175. (Biology).
-
Landau NR, Schatz DG, Rosa M, Baltimore D. Increased frequency of N-region insertion in a murine pre–B-cell line infected with a terminal deoxynucleotidyl transferase retroviral expression vector. Mol Cell Biol. 1987; 7:3237-3243. (Biology).
-
Paietta E, Meenan B, Heavey C, Thomas D. Detection of terminal transferase in acute myeloid leukemia by flow cytometry. Cytometry. 1994; 16:256-261. (Biology).
-
Roma AO, Kutok JL, Shaheen G, Dorfman DM. A novel, rapid, multiparametric approach for flow cytometric analysis of intranuclear terminal deoxynucleotidyl transferase. Am J Clin Pathol. 1999; 112:343-348. (Biology).
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Although not required, these products are manufactured in accordance with Good Manufacturing Practices.