-
Reagents
- Flow Cytometry Reagents
-
蛋白质印迹试剂
- 免疫分析 试剂
-
Single-Cell Multiomics Reagents
- BD® AbSeq Assay
- BD Rhapsody™ 附件试剂盒
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
-
Functional Assays
-
显微成像试剂
-
Cell Preparation and Separation Reagents
-
- BD® AbSeq Assay
- BD Rhapsody™ 附件试剂盒
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- China (Chinese)
-
更改国家/语言
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


监管状态图例
未经BD明确书面授权,严禁使用未经许可的任何商品。
准备和存储
商品通知
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
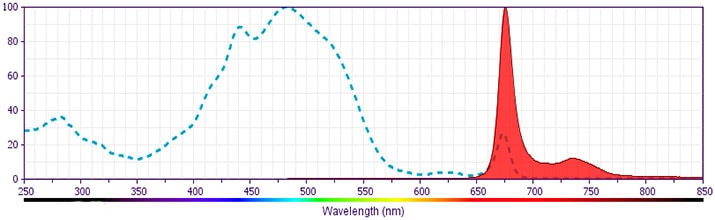
- PerCP is a photosynthetic accessory pigment from Glenodinium species of dinoflagellates, which is excited by the 488-nm light of an Argon ion laser and fluoresces at 675 nm. Therefore, PerCP-labelled antibodies can be used with FITC- and R-PE–labelled reagents in most single-laser flow cytometers with no significant spectral overlap of PerCP fluorescence with that of FITC or R-PE. PerCP has been reported to undergo significant photobleaching, the magnitude of which increases as laser power is increased or beam focus is narrowed. For third-color flow¬cytometric analysis using ≥25-mW laser power, we recommend PE-Cy5-, PE-Cy7–, or PerCP-Cy5.5-conjugated reagents.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
The RA3-6B2 monoclonal antibody specifically binds to an epitope on the extracellular domain of the transmembrane CD45 glycoprotein which is dependent upon the expression of exon A and specific carbohydrate residues. It is expressed on B lymphocytes at all stages from pro-B through mature and activated B cell, but it is decreased on plasma cells and a subset of memory B cells. The levels of CD45R expression on the B-cell lineage appear to be developmentally regulated. It is also reportedly found on the abnormal T cells involved in the lymphadenopathy of lpr/lpr and gld/gld mutant mice, on lytically active subsets of lymphokine-activated killer cells (NK cells and non-MHC-restricted CTL), on apoptotic T lymphocytes of mice injected with bacterial superantigen, on a population of NK-cell precursors in the bone marrow, and on B-lymphocyte, T-lymphocyte, and macrophage progenitors in fetal liver. The CD45R antigen has been reported not to be on hematopoietic stem cells, naive T lymphocytes, or MHC-restricted CTL. CD45 is a member of the Protein Tyrosine Phosphatase (PTP) family: Its intracellular (COOH-terminal) region contains two PTP catalytic domains, and the extracellular region is highly variable due to alternative splicing of exons 4, 5, and 6 (designated A, B, and C, respectively), plus differing levels of glycosylation. The CD45 isoforms detected in the mouse are cell type-, maturation, and activation state-specific. The CD45 isoforms play complex roles in T-cell and B-cell antigen receptor signal transduction. CD45R is commonly used as a pan B-cell marker; however, CD19 expression, detectable by the rat anti-mouse CD19 antibody (clone 1D3), is reported to be more restricted to the B-cell lineage. The rat anti-mouse CD45R antibody (clone RA3-6B2) has been reported to enhance isotype switching during in vitro B-cell responses and to inhibit in vivo B-cell responses. Cross-reaction of the RA3-6B2 clone with activated human T lymphocytes has also been reportedly observed.
研发参考 (22)
-
Afar B, Merrill J, Clark EA. Detection of lymphocyte subsets using three-color/single-laser flow cytometry and the fluorescent dye peridinin chlorophyll-alpha protein. J Clin Immunol. 1991; 11(5):254-261. (Biology). 查看参考
-
Allman DM, Ferguson SE, Cancro MP. Peripheral B cell maturation. I. Immature peripheral B cells in adults are heat-stable antigenhi and exhibit unique signaling characteristics. J Immunol. 1992; 149(8):2533-2540. (Biology). 查看参考
-
Asensi V, Kimeno K, Kawamura I, Sakumoto M, Nomoto K. Treatment of autoimmune MRL/lpr mice with anti-B220 monoclonal antibody reduces the level of anti-DNA antibodies and lymphadenopathies. Immunology. 1989; 68(2):204-208. (Clone-specific). 查看参考
-
Ballas ZK, Rasmussen W. Lymphokine-activated killer cells. VII. IL-4 induces an NK1.1+CD8 alpha+beta- TCR-alpha beta B220+ lymphokine-activated killer subset. J Immunol. 1993; 150(1):17-30. (Biology). 查看参考
-
Bleesing JJ, Morrow MR, Uzel G, Fleisher TA. Human T cell activation induces the expression of a novel CD45 isoform that is analogous to murine B220 and is associated with altered O-glycan synthesis and onset of apoptosis. Cell Immunol. 2001; 213(1):72-81. (Clone-specific). 查看参考
-
Coffman RL. Surface antigen expression and immunoglobulin gene rearrangement during mouse pre-B cell development. Immunol Rev. 1982; 69:5-23. (Biology). 查看参考
-
Domiati-Saad R, Ogle EW, Justement LB. Administration of anti-CD45 mAb specific for a B cell-restricted epitope abrogates the B cell response to a T-dependent antigen in vivo. J Immunol. 1993; 151(11):5936-5947. (Clone-specific). 查看参考
-
Driver DJ, McHeyzer-Williams LJ, Cool M, Stetson DB, McHeyzer-Williams MG. Development and maintenance of a B220- memory B cell compartment. J Immunol. 2001; 167(3):1393-1405. (Biology). 查看参考
-
George A, Rath S, Shroff KE, Wang M, Durdik JM. Ligation of CD45 on B cells can facilitate production of secondary Ig isotypes. J Immunol. 1994; 152(3):1014-1021. (Clone-specific). 查看参考
-
Greimers R, Trebak M, Moutschen M, Jacobs N, Boniver J. Improved four-color flow cytometry method using fluo-3 and triple immunofluorescence for analysis of intracellular calcium ion ([Ca2+]i) fluxes among mouse lymph node B- and T-lymphocyte subsets. Cytometry. 1996; 23(3):205-217. (Biology). 查看参考
-
Hardy RR, Carmack CE, Shinton SA, Kemp JD, Hayakawa K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J Exp Med. 1991; 173(5):1213-1225. (Biology). 查看参考
-
Hathcock KS, Hirano H, Murakami S, Hodes RJ. CD45 expression by B cells. Expression of different CD45 isoforms by subpopulations of activated B cells. J Immunol. 1992; 149(7):2286-2294. (Biology). 查看参考
-
Johnson P, Maiti A, Ng DHW. CD45: A family of leukocyte-specific cell surface glycoproteins. In: Herzenberg LA, Weir DM, Herzenberg LA, Blackwell C , ed. Weir's Handbook of Experimental Immunology, Vol 2. Cambridge: Blackwell Science; 1997:62.1-62.16.
-
Kobata T, Takasaki K, Asahara H, et al. Apoptosis with FasL+ cell infiltration in the periphery and thymus of corrected autoimmune mice. Immunology. 1997; 92(2):206-213. (Biology). 查看参考
-
Krop I, de Fougerolles AR, Hardy RR, Allison M, Schlissel MS, Fearon DT. Self-renewal of B-1 lymphocytes is dependent on CD19. Eur J Immunol. 1996; 26(1):238-242. (Biology). 查看参考
-
Laouar Y, Ezine S. In vivo CD4+ lymph node T cells from lpr mice generate CD4-CD8-B220+TCR-beta low cells. J Immunol. 1994; 153(9):3948-3955. (Biology). 查看参考
-
Puzanov IJ, Bennett M, Kumar V. IL-15 can substitute for the marrow microenvironment in the differentiation of natural killer cells. J Immunol. 1996; 157(10):4282-4285. (Biology). 查看参考
-
Renno T, Hahne M, Tschopp J, MacDonald HR. Peripheral T cells undergoing superantigen-induced apoptosis in vivo express B220 and upregulate Fas and Fas ligand. J Exp Med. 1996; 183(2):431-437. (Biology). 查看参考
-
Rolink A, ten Boekel E, Melchers F, Fearon DT, Krop I, Andersson J. A subpopulation of B220+ cells in murine bone marrow does not express CD19 and contains natural killer cell progenitors. J Exp Med. 1996; 183(1):187-194. (Biology). 查看参考
-
Sagara S, Sugaya K, Tokoro Y, et al. B220 expression by T lymphoid progenitor cells in mouse fetal liver. J Immunol. 1997; 158(2):666-676. (Biology). 查看参考
-
Shapiro HM. Practical Flow Cytometry, 3rd Edition. New York: Wiley-Liss, Inc; 1995:280-281.
-
Waggoner AS, Ernst LA, Chen CH, Rechtenwald DJ. PE-CY5. A new fluorescent antibody label for three-color flow cytometry with a single laser. Ann N Y Acad Sci. 1993; 677:185-193. (Biology). 查看参考
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.
