-
Reagents
- Flow Cytometry Reagents
-
蛋白质印迹试剂
- 免疫分析 试剂
-
Single-Cell Multiomics Reagents
- BD® AbSeq Assay
- BD Rhapsody™ 附件试剂盒
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
-
Functional Assays
-
显微成像试剂
-
Cell Preparation and Separation Reagents
-
- BD® AbSeq Assay
- BD Rhapsody™ 附件试剂盒
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- China (Chinese)
-
更改国家/语言
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




Two-color flow cytometric analysis of IL-10 expression in stimulated mouse T cells. Splenic CD4+ T cells were cultured (2 days) with plate-bound Purified NA/LE Hamster Anti-Mouse CD3e (Cat. No. 553057; 25 μg/ml for coating) and soluble Purified NA/LE Hamster Anti-Mouse CD28 (Cat. No. 553294; 2 μg/ml) antibodies and with Recombinant IL-2 (Cat. No. 550069; 10 ng/ml) and IL-4 (Cat. No. 550067; 25 ng/ml). The stimulated cells were subsequently cultured (3 days) with Recombinant IL-2 and IL-4. The cells were then stimulated with PMA (Sigma Cat. No. P-8139) and Ionomycin (Sigma Cat. No. I-0634) in the presence of BD GolgiStop™ Protein Transport Inhibitor (containing Monensin) (Cat. No. 554724). After 6 hours, the cells were harvested, washed, fixed and permeabilized with BD Cytofix/Cytoperm™ Fixation and Permeabilization Solution (Cat. No. 554722). The cells were then washed and stained in BD Perm/Wash™ Buffer (Cat. No. 554723) with BD Horizon™ BV605 Rat IgG2b, κ Isotype Control (Cat. No. 563145; Left Panel) or BD Horizon BV605 Rat Anti-Mouse IL-10 antibody (Cat. No. 564082; Right Panel). Flow cytometric contour plots showing correlated expression of IL-10 (or Ig Isotype control staining) versus Autofluorescence were derived from gated events with the forward and side light-scatter characteristics of intact stimulated lymphocytes. Flow cytometric analysis was performed using a BD LSRFortessa™ Cell Analyzer System.


BD Horizon™ BV605 Rat Anti-Mouse IL-10

监管状态图例
未经BD明确书面授权,严禁使用未经许可的任何商品。
准备和存储
商品通知
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Please observe the following precautions: Absorption of visible light can significantly alter the energy transfer occurring in any tandem fluorochrome conjugate; therefore, we recommend that special precautions be taken (such as wrapping vials, tubes, or racks in aluminum foil) to prevent exposure of conjugated reagents, including cells stained with those reagents, to room illumination.
- Although every effort is made to minimize the lot-to-lot variation in the efficiency of the fluorochrome energy transfer, differences in the residual emission from BD Horizon™ BV421 may be observed. Therefore, we recommend that individual compensation controls be performed for every BD Horizon™ BV605 conjugate.
- CF™ is a trademark of Biotium, Inc.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
配套商品





The JES5-16E3 monoclonal antibody specifically binds to the mouse cytokine, Interleukin-10 (IL-10). IL-10 is also known as Cytokine Synthesis Inhibitory Factor (CSIF). It is produced by various activated cell types including CD4+ T cells, CD8+ T cells, T regulatory cells, NK T cells, B1 B cells, NK cells, macrophages, dendritic cells, mast cells, granulocytes and keratinocytes. IL-10 plays a pivotal role in regulating immune responses and protecting the host from damage caused by inflammatory and autoimmune responses. IL-10 has numerous biological activities including the inhibition of cytokine synthesis by activated T cells, NK cells, monocytes, and macrophages. In the presence of accessory cells, IL-10 inhibits mitogen- or anti-CD3 induced proliferation of T lymphocytes. IL-10 has also been shown to costimulate the development of thymocytes, B cell differentiation and the generation of cytotoxic T cells. The immunogen used to generate the JES5-16E3 hybridoma was recombinant mouse IL-10. JES5-16E3 is a neutralizing antibody.
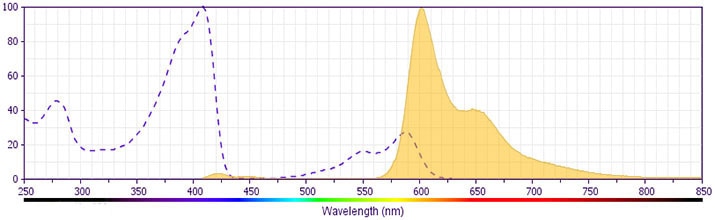
This antibody is conjugated to BD Horizon BV605 which is part of the BD Horizon Brilliant™ Violet family of dyes. With an Ex Max of 407-nm and Em Max of 602-nm, BD Horizon BV605 can be excited by a violet laser and detected with a standard 610/20-nm filter set. BD Horizon BV605 is a tandem fluorochrome of BD Horizon BV421 and an acceptor dye with an Em max at 605-nm. Due to the excitation of the acceptor dye by the green (532 nm) and yellow-green (561 nm) lasers, there will be significant spillover into the PE and BD Horizon PE-CF594 detectors off the green or yellow-green lasers. BD Horizon BV605 conjugates are very bright, often exhibiting brightness equivalent to PE conjugates and can be used as a third color off of the violet laser.
For optimal and reproducible results, BD Horizon Brilliant Stain Buffer should be used anytime two or more BD Horizon Brilliant dyes are used in the same experiment. Fluorescent dye interactions may cause staining artifacts which may affect data interpretation. The BD Horizon Brilliant Stain Buffer was designed to minimize these interactions. More information can be found in the Technical Data Sheet of the BD Horizon Brilliant Stain Buffer (Cat. No. 563794).
研发参考 (4)
-
Andersson U, Andersson J. Immunolabeling of cytokine-producing cells in tissues and in suspension. In: Fradelizie D, Emelie D, ed. Cytokine Producing Cells. Paris: Inserm; 1994:32-49.
-
Litton MJ, Sander B, Murphy E, O'Garra A, Abrams JS. Early expression of cytokines in lymph nodes after treatment in vivo with Staphylococcus enterotoxin B. J Immunol Methods. 1994; 175(1):47-58. (Clone-specific: Neutralization). 查看参考
-
Prussin C, Metcalfe DD. Detection of intracytoplasmic cytokine using flow cytometry and directly conjugated anti-cytokine antibodies. J Immunol Methods. 1995; 188(1):117-128. (Methodology: Flow cytometry). 查看参考
-
Sander B, Hoiden I, Andersson U, Moller E, Abrams JS. Similar frequencies and kinetics of cytokine producing cells in murine peripheral blood and spleen. Cytokine detection by immunoassay and intracellular immunostaining. J Immunol Methods. 1993; 166(2):201-214. (Clone-specific: Immunocytochemistry (cytospins), Neutralization). 查看参考
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.