Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
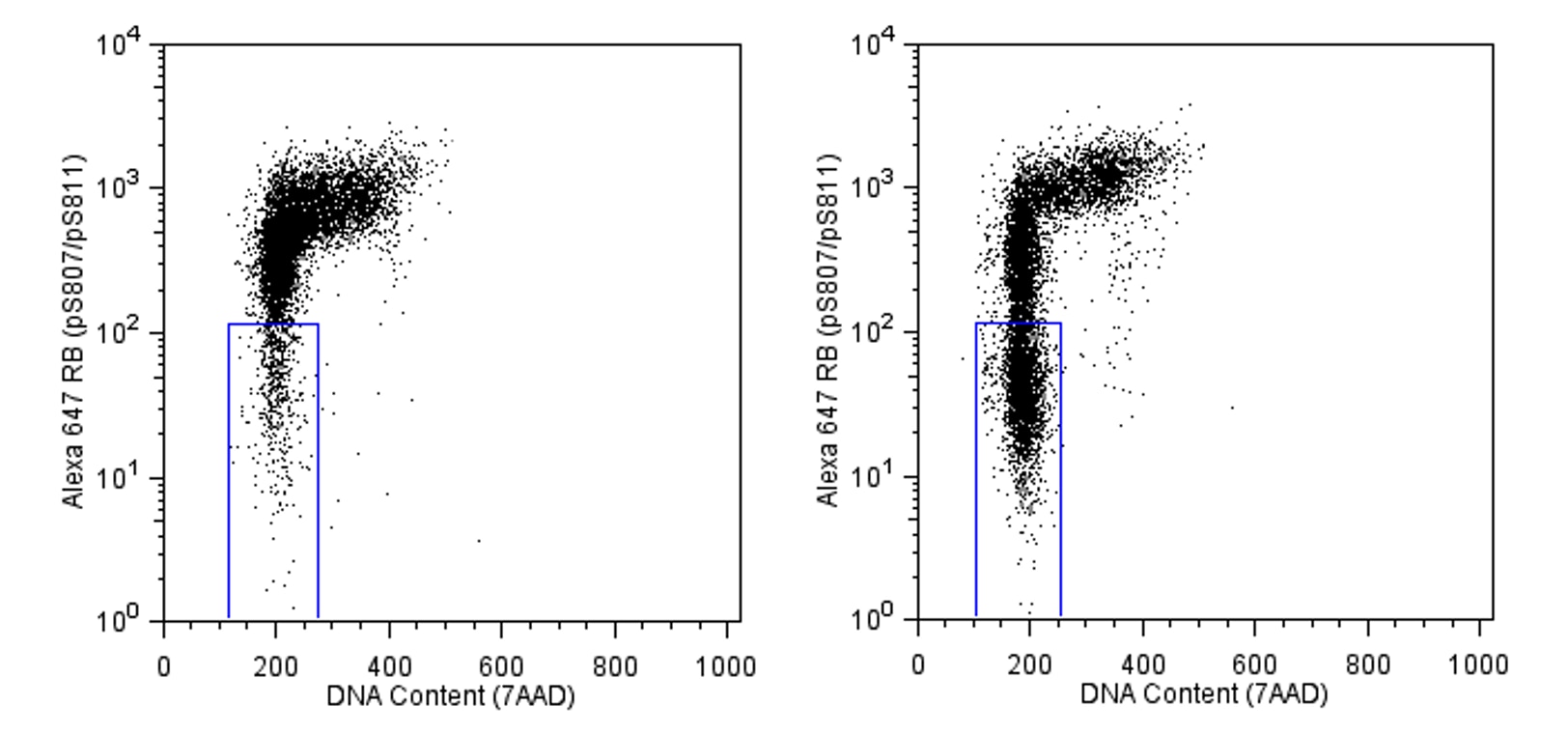



Analysis of Rb (pS807/pS811) in activated human acute lymphoblastic leukemia. MOLT-4 cells (without serum starvation) were either stimulated with 200 ng/ml PMA (Sigma, Cat. No. P8139) for 24 hours at 37°C (right panel) or unstimulated (left panel). The cells were fixed (BD Phosflow™ Fix Buffer I, Cat. No. 557870) for 10 minutes at 37°C, then permeabilized (BD Phosflow™ Perm Buffer III, Cat. No. 558050) on ice for at least 30 minutes, and then stained with Alexa Fluor® 647 Mouse anti-Rb (pS807/pS811). 7AAD staining solution (Cat. No. 559925) was used to monitor the cell cycle of the stimulated and unstimulated cells. Flow cytometry was performed on a BD FACSCalibur™ flow cytometry system. The boxed region in the data demonstrates that PMA treatment causes cell cycle arrest at the G1 phase, which is associated with dephosphorylation of Rb.


BD™ Phosflow Alexa Fluor® 647 Mouse anti-Rb (pS807/pS811)

监管状态图例
未经BD明确书面授权,严禁使用未经许可的任何商品。
准备和存储
商品通知
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Alexa Fluor® 647 fluorochrome emission is collected at the same instrument settings as for allophycocyanin (APC).
- Alexa Fluor® is a registered trademark of Molecular Probes, Inc., Eugene, OR.
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
The retinoblastoma gene product (Rb) is well known as a tumor suppressor and is either absent or mutated in many human tumors. Retrovirus-mediated gene transfer of the wild-type Rb gene into several Rb mutant neoplastic cell lines suppresses their tumorgenicity. Rb is a 110-kDa nuclear phosphoprotein that undergoes differential phosphorylation during the cell cycle. During G1 phase, Rb is predominantly in a hypophosphorylated state. It becomes increasingly phosphorylated throughout the cell cycle until late mitosis, when substantial dephosphorylation occurs. Hypophosphorylated Rb interacts with a number of cellular proteins including the E2F transcription factor, several cyclins, RBP-1, RBP-2, c-Abl, c-myc, N-myc, and p46. Phosphorylation of Rb at various sites, by Cyclin-dependent protein kinases, inhibits the binding of Rb to these proteins. Rb is thought to mediate its effects, in part, via the repression of genes required for proliferation. For example, Rb is specifically recruited to promoters containing E2F sites and actively represses E2F mediated transcription. Rb also stimulates the activity of other transcription factors, although the mechanisms are less clearly defined. Thus, Rb appears to regulate transcription in its aim to control cell growth.
The J112-906 monoclonal antibody recognizes Rb phosphorylated at serines 807 and 811 (pS807/pS811), which regulate c-Abl binding and cell cycle progression. The orthologous phosphorylation sites in mouse and rat Rb are pS800/pS804 and pS778/pS782, respectively.
研发参考 (3)
-
Cobrinik D. Pocket proteins and cell cycle control. Oncogene. 2005; 24:2796-2809. (Biology).
-
Knudsen ES, Wang JY. Differential regulation of retinoblastoma protein function by specific Cdk phosphorylation sites. J Biol Chem. 1996; 271(14):8313-8320. (Biology).
-
Ren S, Rollins BJ. Cyclin C/Cdk3 promotes Rb-dependent G0 exit. Cell. 2004; 117:239-251. (Biology).
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.