Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


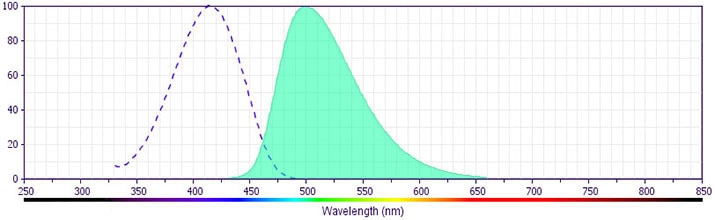
CD8 V500-C
监管状态图例
未经BD明确书面授权,严禁使用未经许可的任何商品。
The CD8 antibody, clone SK1, is derived from hybridization of mouse NS-1 myeloma cells with spleen cells from BALB/c mice immunized with human peripheral blood T lymphocytes.
The CD8 antibody recognizes the 32-kilodalton (kDa) α-subunit of a disulfide-linked bimolecular complex. The majority of peripheral blood CD8+ T lymphocytes expresses an α/β heterodimer (Mr 32, 30 kDa), while CD8+CD16+ natural killer (NK) lymphocytes and CD8+ T-cell receptor (TCR)-γ/δ+ T lymphocytes express an α/α homodimer (Mr 30 kDa). CD8+TCR-α/β+ T lymphocytes can express either an α/α homodimer or α/β heterodimer. The CD8 antigenic determinant binds to class I major histocompatibility (MHC) molecules, resulting in increased adhesion between the CD8+ T lymphocytes and target cells. Binding of the CD8 antigen to class I MHC molecules enhances the activation of resting T lymphocytes. The CD8 antigen is coupled to a protein tyrosine kinase, p56lck. The CD8:p56lck complex can play a role in T-lymphocyte activation through mediation of the interactions between the CD8 antigen and the CD3/TCR complex.
研发参考 (19)
-
Anderson P, Blue ML, Morimoto C, Schlossman SF. Cross-linking of T3 (CD3) with T4 (CD4) enhances the proliferation of resting T lymphocytes. J Immunol. 1987; 139:678-682. (Biology).
-
Bernard A, Boumsell L, Hill C. Joint report of the first international workshop on human leucocyte differentiation antigens by the investigators of the participating laboratories. In: Bernard A, Boumsell L, Dausset J, Milstein C, Schlossman SF, ed. Leucocyte Typing. New York, NY: Springer-Verlag; 1984:9-108.
-
Centers for Disease Control. Update: universal precautions for prevention of transmission of human immunodeficiency virus, hepatitis B virus, and other bloodborne pathogens in healthcare settings. MMWR. 1988; 37:377-388. (Biology).
-
Clinical and Laboratory Standards Institute. 2005. (Biology).
-
Cosimi AB. Anti-T-cell monoclonal antibodies in transplantation therapy. Trans Proc. 1983; XV:1889-1892. (Biology).
-
Eichmann K, Johnson J, Falk I, Emmrich F. Effective activation of resting mouse T lymphocytes by cross-linking submitogenic concentrations of the T-cell antigen receptor with either Lyt-2 or L3T4. Eur J Immunol. 1987; 17:643-650. (Biology).
-
Engleman EG, Benike CJ, Evans RL. Circulating antigen-specific suppressor T cells in a healthy woman: mechanism of action and isolation with a monoclonal antibody. Clin Res. 1981; 29:365A. (Biology).
-
Engleman EG, Benike CJ, Glickman E, Evans RL. Antibodies to membrane structures that distinguish suppressor/cytotoxic and helper T lymphocyte subpopulations block the mixed leukocyte reaction in man. J Exp Med. 1981; 154(1):193-198. (Biology). 查看参考
-
Evans RL, Wall DW, Platsoucas CD, et al. Thymus-dependent membrane antigens in man: inhibition of cell-mediated lympholysis by monoclonal antibodies to TH2 antigen. Proc Natl Acad Sci U S A. 1981; 78(1):544-548. (Biology). 查看参考
-
Gallagher PF, Fazekas de St. Groth B, Miller JFAP. CD4 and CD8 molecules can physically associate with the same T-cell receptor. Proc Natl Acad Sci USA. 1989; 86:10044-10048. (Biology).
-
Gatenby PA, Kansas GS, Xian CY, Evans RL, Engleman EG. Dissection of immunoregulatory subpopulations of T lymphocytes within the helper and suppressor sublineages in man. J Immunol. 1982; 129(5):1997-2000. (Biology). 查看参考
-
Kotzin BL, Benike CJ, Engleman EG. Induction of immunoglobulin-secreting cells in the allogeneic mixed leukocyte reaction: regulation by helper and suppressor lymphocyte subsets in man. J Immunol. 1981; 127(9):931-935. (Biology). 查看参考
-
Lanier LL, Le AM, Phillips JH, Warner NL, Babcock GF. Subpopulations of human natural killer cells defined by expression of the Leu-7 (HNK-1) and Leu-11 (NK-15) antigens. J Immunol. 1983; 131(4):1789-1796. (Biology). 查看参考
-
Ledbetter JA, Evans RL, Lipinski M, Cunningham-Rundles C, Good RA, Herzenberg LA. Evolutionary conservation of surface molecules that distinguish T lymphocyte helper/inducer and cytotoxic/suppressor subpopulations in mouse and man. J Exp Med. 1981; 153(2):310-323. (Biology). 查看参考
-
Ledbetter JA, Frankel AE, Herzenberg. Human Leu T-cell differentiation antigens: quantitative expression on normal lymphoid cells and cell lines. In: Hammerling G, Hammerling U, Kearney J, ed. Monoclonal Antibodies and T Cell Hybridomas: Perspectives and Technical News. New York: Elsevier/North Holland Biomedical Press; 1981:16-22.
-
Moebius U. Knapp W, Dörken B, Gilks W, et al, ed. Leucocyte Typing IV. White Cell Differentiation Antigens. New York: Oxford University Press; 1989:342-343.
-
Reichert T, DeBruyere M, Deneys V, et al. Lymphocyte subset reference ranges in adult Caucasians. Clin Immunol Immunopathol. 1991; 60(2):190-208. (Biology). 查看参考
-
Rudd CE, Burgess KE, Barber EK, Schlossman SF. Knapp W, Dörken B, Gilks WR, et al, ed. Leucocyte Typing IV: White Cell Differentiation Antigens. New York, NY: Oxford University Press; 1989:326-327.
-
Terry LA, DiSanto JP, Small TN, Flomenberg N. Knapp W, Dörken B, Gilks WR, et al, ed. Leucocyte Typing IV: White Cell Differentiation Antigens. New York, NY: Oxford University Press; 1989:345-346.
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.