-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ CRYO Preservation Buffer
- BD OMICS-Guard™ Sample Preservation Buffer
- BD OMICS-One™ Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD OMICS-One™ WTA Next Assay
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- Advanced Training
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Cell Therapy
Tremendous advances have been made in cell therapy in the past decade. Building off experience in hematopoietic stem cell transplant and exploiting advances in genetic engineering, genome editing, immunology, immunotherapy, stem cell biology, understanding disease mechanisms and cell therapy manufacturing, considerable progress has been made with hundreds of clinical trials underway. Engineered chimeric antigen receptor (CAR) T cell therapies (CAR-T) are now commercially available to treat certain leukemias and lymphomas. Investigational cell therapies are addressing a wide range of hematological malignancies. Solid tumors, autoimmune diseases, graft versus host disease (GvHD), genetic diseases and neurodegenerative diseases are investigated using many types of autologous and allogeneic cells with rapidly evolving cell engineering and editing approaches.
A common theme in cell therapy is the need to characterize and understand cell phenotypes and heterogeneity at every step of the process. This includes:
- Characterization of patient disease and development of predictive biomarkers
- Characterization of cells isolated from patients as input to cell therapy manufacturing
- Study of candidate cell therapies in vitro and in preclinical models
- Development and optimization of the cell therapy manufacturing process
- Cell product release testing
- Development of monitoring tools of cell therapies and patient response post-infusion
BD Biosciences supports each stage of this journey, with research tools for single-cell multiomics, high-parameter analysis and informatics to deeply characterize cell samples in discovery.
Utilizing T Cells and NK Cells for Cell Therapy Research
T Cell Therapies
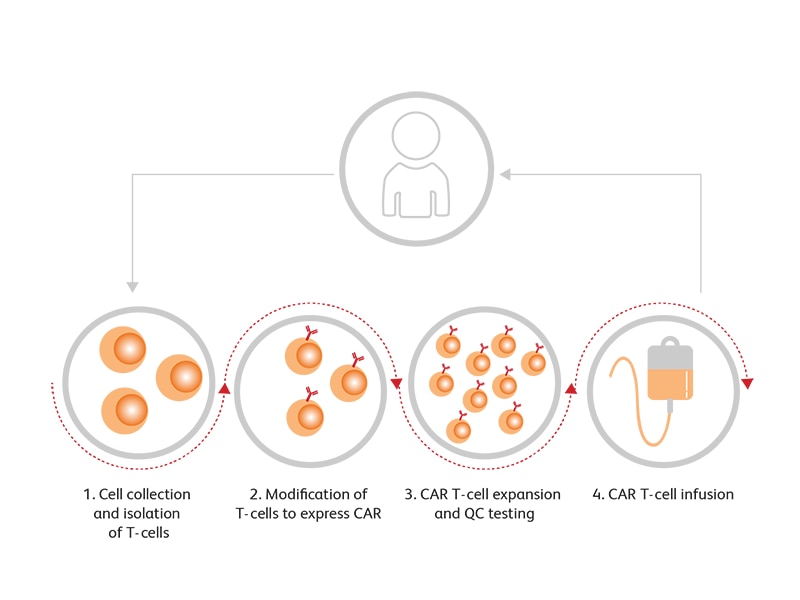
CAR-T cells, which incorporate an antibody-derived extracellular receptor and T cell derived intracellular signaling domains, have shown convincing outcomes in certain types of leukemia and lymphoma, including commercial licensure of CD19 CAR-Ts for the treatment of relapsed/ refractory large B cell lymphoma and acute lymphoblastic leukemia. The technique has also shown preliminarily promising outcomes in B cell acute lymphoblastic leukemia (B-ALL), B cell non-Hodgkin’s lymphoma (B-NHL), chronic lymphocytic leukemia (CLL), Hodgkin’s lymphoma and multiple myeloma in clinical trials targeting CD19, CD20, CD22, CD30 and B-cell maturation antigen (BCMA), with many more under investigation.1 T cells are engineered to express CARs through viral vectors, enabling them to recognize specific target antigens. The resultant activation of T cells is then exploited for tumor elimination. T cell–targeted immunomodulators such as monoclonal antibodies against PD-1 or CTLA4 may be used in combination with CAR-T cells in clinical trials.2 Generic steps in (CAR) T cell therapy are shown below:
Step 1: T Cell Collection
Patient PBMCs are collected by leukapheresis followed by enrichment approaches to isolate T cells while removing other cells such as B cells, monocytes, neutrophils, dendritic cells and contaminating tumor cells.
Step 2: (CAR) T Cell Generation
T cells are modified ex vivo by transduction with viral vectors containing CAR or other modifications of interest.
Step 3: (CAR) T Cell Expansion and QC Testing
(CAR) T cells are then grown and expanded in defined cell culture conditions and harvested. Quality control testing is essential to ensure the CAR-T cell product meets established specifications for manufacturing release and patient infusion.
Step 4: (CAR) T Cell Infusion
Harvested cells are then administered to the patient.
Augmenting the Effectiveness of the (CAR) T Cell Therapy Approach
(CAR) design has undergone several levels of enhancements, each step adding to the efficacy and safety of the outcome. The fourth generation of CAR-T design attempts to deliver cytokines to modulate the tumor microenvironment either by activating host effector T cells or hampering host suppressors and reinforcing memory T cells. These experimental cytokine-producing CAR-Ts, called T cells redirected for universal cytokine killing (TRUCKs), can deliver a variety of cytokines, such as IL-12, IL-15, IL-18 or IL-21.3
Related T cell approaches such as engineered T cell receptors also show promise. Current efforts use sophisticated cell engineering to direct novel T cell therapies to hematological and solid tumors while avoiding toxicity (particularly cytokine release syndrome and neurotoxicity), counteracting tumor escape mechanisms with dual CARs or combination therapies, enhancing tumor cell killing with cytokine-secreting CAR-T cells to improve immune response in the tumor microenvironment, adding modified checkpoint receptors to prevent blockade, reducing cell exhaustion while maintaining CAR-T cell proliferation and persistence, and removing immunogenic components to enable allogeneic therapies.
Utilizing NK cells for Cell Therapy Research
Besides T cells, NK Cells are also being explored as potential candidates for use in cell therapy based on several lines of evidence. NK cells are part of the innate immune system and programmed to protect normal autologous cells with a balance between appropriate levels of self–HLA alleles and non-HLA-specific activating receptors. Tumor cells manipulate this balance of HLA-I levels and block the cytotoxic activity of NK cells. Downregulation of HLA-I levels can induce NK cell–mediated killing through a “missing-self” recognition mechanism.4 The inhibitory mechanism includes killer immunoglobulin-like receptors (KIRs) and CD94/NKG2A, which can recognize major histocompatibility complex (MHC) class I molecules. This property enables manipulations that can create mismatch between donor and recipient KIRs and their ligands, which can be effective in overcoming alloreactivity after transplantation. This strategy is employed in allogenic cell therapy using NK cells.5 Engineered NK cells expressing CAR (CAR-NK cells) have been evaluated in clinical trials for several types of cancers.6 In addition, the activated NK cell line NK-92 is also being investigated for cell therapy in clinical trials as it can be expanded easily in culture, effectively transfected, and can overcome some of the limitations of NK cells.7
Hematopoietic Stem Cell Transplantation (HSCT)*
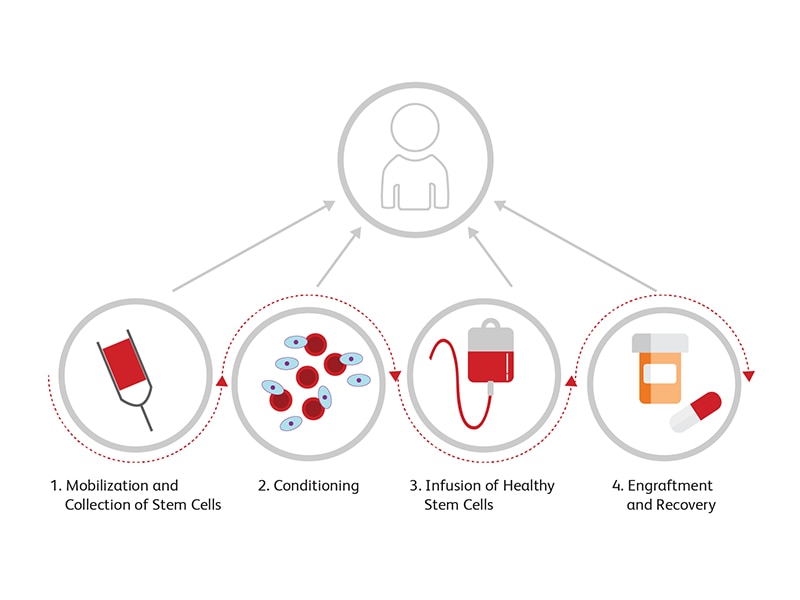
HSCT is well-established both as a rescue therapy after chemotherapy and radiotherapy paradigms in cancer patients and as an adoptive immune therapy for patients with autoimmune disorders. In allogenic HCST, the entire immune system is replaced by the donor cells by matching human histocompatibility locus (HLA) alleles. Current innovations are focused on adjuvant therapies (including Treg cell therapies) to reduce the risk of graft versus host disease, on improving outcomes for use of HLA mismatched transplants, and the use of normal donor HSCT to rescue hematologic genetic diseases. HSCT involves the following steps:
Step 1: Mobilization and Collection of Stem Cells
Stem cells can be mobilized from their niches or in the peripheral blood using factors such as G-CSF or filgrastim. These factors are used to boost the counts of stem cells for transplantation.8 Stem cells are then collected and stored.
Step 2: Conditioning
Conditioning therapy is used to prepare the patient for the transplant. A combination of therapies including chemotherapy and radiation are used to clear the bone marrow of the patient of rapidly dividing cells, including malignant cells, and to create space for donor cells during engraftment. Combining immunotherapy to this strategy helps in preventing immunosuppression induced by cancer cells and helps mitigate the risk of rejection of the donor cells.9
Step 3: Infusion of Healthy Stem Cells
After depleting the immune cells, healthy cells are infused into the patient’s body.
Step 4: Engraftment and Recovery
After transplantation, the patient is monitored to verify the engraftment status and to ensure that there are no signs of rejection.
*BD Biosciences clinical flow cytometry solutions, including instrumentation, software, and reagents, offer the building blocks for laboratory-developed tests used in the identification of markers associated with several immune-related diseases. These solutions are not FDA cleared or approved for the diagnosis of these diseases. Analyte Specific Reagent. Analytical and performance characteristics are not established.
References
- Wang Z, Wu Z, Liu Y, Han W. New development in CAR-T cell therapy. J Hematol Oncol 2017;10(1):53. doi: 10.1186/s13045-017-0423-1
- Fins S, Kong W, Williams EF, Milone MC, Fraietta JA. An introduction to chimeric antigen receptor (CAR) T-cell immunotherapy for human cancer. Am J Hematol. 2019;94(S1):S3-S9. doi: 10.1002/ajh.25418<
- Knochelmann HM, Smith AS, Dwyer CJ, Wyatt MM, Mehrotra S, Paulos CM. CAR T cells in solid tumors: Blueprints for building effective therapies. Front Immunol. 2018;9:1740. doi: 10.3389/fimmu.2018.01740
- Minetto P, Guolo F, Pesce S, et al. Harnessing NK cells for cancer treatment. Front Immunol. 2019;10:2836. doi: 10.3389/fimmu.2019.02836
- Karantalis V, Schulman IH, Balkan W, Hare JM. Allogenic cell therapy: a new paradigm in therapeutics. Circ Res. 2015;116(1):12-15. doi: 10.1161/CIRCRESAHA.114.305495
- Rezvani K. Adoptive cell therapy using engineered natural killer cells. Bone Marrow Transplant. 2019;54(Suppl):785-788. doi:10.1038/s41409-019-0601-6
- Romanski A, Uherek C, Bug G, et al. CD-19-CAR engineered NK-92 cells are sufficient to overcome NK cell resistance in B-cell malignancies. J Cell Mol Med.2016;20(7):1287-1294. doi: 10.1111/jcmm.12810
- Karpova D, Rettig MP, DiPersio JF. Mobilized peripheral blood: an updated perspective. F1000Res. 2019;8:F1000 Faculty Rev-2125. doi: 10.12688/f1000research.21129.1
- Zulu S, Kenyon M. Principles of conditioning therapy and cell infusion. In Kenyon M, Babic A, eds. The European Blood and Marrow Transplantation Textbook for Nurses: Under the Auspices of EBMT [Internet]. Cham (CH): Springer; 2018.
BD Biosciences Offers Tools for Cell Analysis
From sample preparation and cell sorting to flow cytometry and downstream sequencing and informatics analysis, BD Biosciences offers a variety of instruments, reagents and custom offerings that can be used in cell therapy research.
Our research cell analyzers, such as the BD FACSymphony™ Flow Cytometers, can be used for the simultaneous measurement of up to 50 parameters. The BD LSRFortessa™ System can analyze up to 20 parameters. The BD Rhapsody™ Single-Cell Analysis System enables high-throughput capture and analysis of hundreds of single cells. Our single-cell multiomics reagents portfolio enables the analysis of the entire transcriptome or of targeted genes of interest, while targeted panels like the BD Rhapsody™ Immune Response Panel allow profiling of specific immune cell types. The same cells can be interrogated with over 100 antibodies using our BD® AbSeq Technology and clonal diversity can be tracked with our BD® TCR/BCR Analysis Assay for true single-cell multiomics.
Our flagship clinical cell analyzer, the BD FACSLyric™ Flow Cytometer, is also available in an RUO mode offering highly standardized and consistent performance, excellent sensitivity across 12 colors, and audit trail and e-signature to support 21 CFR part 11 compliance, for your cell therapy product release testing and emerging clinical applications.
We also provide several sample preparation tools that can be used in cell therapy research. The BD Vacutainer® family of products can be used for blood cell and biomarker preservation.
The BD Horizon™ Dri Tumor and Tissue Dissociation Reagent (TTDR) offers gentle and effective dissociation with superior epitope preservation. TTDR maximizes cell yields while minimizing cell death, which allows effective dissociation of a variety of tumor types to enable single-cell studies to support the development of cell therapies for solid tumors. The dried reagent cocktails of BD Horizon™ Dri Panels are predesigned, ready to use multicolor panels optimized and tested for memory T cell, monocyte subset and T, B, and NK cell characterization.
In addition, our flow cytometry panel design solutions and a comprehensive portfolio of over 9,000 immunology research reagents can support cell therapy research at every step, from testing and discovery phases to uncovering the identity, purity, potency and safety of cell therapy product attributes. Custom panels, including dried cocktails for consistent performance over time and locations, can also be manufactured based on customer specifications. Finally, special order cell processing reagents are available to meet the GMP manufacturing needs for ancillary materials in cell therapy product manufacturing.
All of this is supported by leading informatics capabilities with FlowJo™ v10 Software, providing the latest analytical tools for flow cytometry and single-cell multiomics analysis.
Custom cGMP reagents for cell isolation
| Specificity | Clone | Format |
|---|---|---|
CD4 |
(L200) |
PerCP |
CD25 |
(2A3) |
APC |
CD127 |
(40131) |
PE |
Cell characterization antibodies for cell therapy development
| Regulatory Status | Format | Product Highlights | |
|---|---|---|---|
Single Vials |
RUO or RUO GMP |
Liquid |
>6,000 directly conjugated RUO antibodies (ISO certified)
>300 directly conjugated RUO GMP antibodies
Innovative dye technology developed by Sirigen provides brighter dye options for better cell separation and easier multicolor analysis |
Custom Antipbody Cocktails |
RUO |
Liquid or lyophilized
In vials or 96-well plates |
BD Lyoplate™ Plate format flexibility permits user-defined experiment layouts, including the exact content of each well within the plate |
Custom Antibody Conjugates |
RUO/GMP |
Liquid |
Conjugation of any fluorescent dye to any antibody within the BD portfolio or to customer-provided antibodies. A broad portfolio of fluorochromes featuring the BD Horizon Brilliant™ Dyes offers flexibility for experimental design. |
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
BD flow cytometers are Class 1 Laser Products.