-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ CRYO Preservation Buffer
- BD OMICS-Guard™ Sample Preservation Buffer
- BD OMICS-One™ Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD OMICS-One™ WTA Next Assay
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- Advanced Training
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

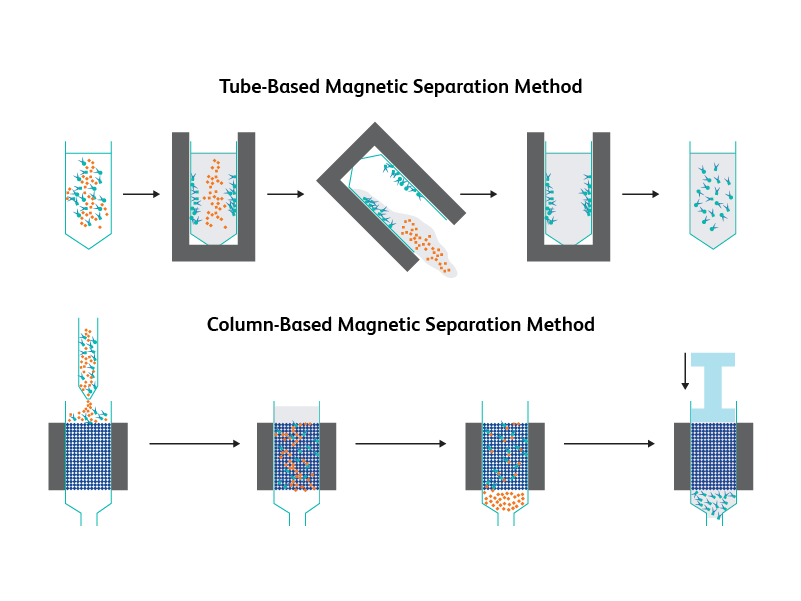
Magnetic Separation
Cell separation is a powerful technique and an indispensable tool for basic and clinical research applications. The heterogeneity of biological cell populations often necessitates separation of individual cell types for deeper investigation. Traditionally, cell separation is carried out based on the physical properties of cells, such as adherence, size, density or affinity to electrostatic or magnetic forces. Biochemical characteristics, such as expression of surface antigens, are also used for cell separation.
What is magnetic cell separation and how is it achieved?
This cell separation technique utilizes the potential to label cell surface markers with magnetic bead–tagged antibodies and the ability of a magnetic field to migrate the labeled particles from a distance.1 This controlled migration by a magnetic force (magnetophoresis) is invaluable in separating heterogeneous cell populations and is the basis for magnetic-activated cell sorting (MACS). Cells can be separated by tube-based or column-based methods.2
In a tube-based magnetic-separation method, a suspension of magnetically labeled cells are placed in a magnet, allowing movement of labeled cells, which can then be separated.
In a column-based magnetic-separation method, a suspension of magnetically labeled cells are separated through a column within a magnet, capturing labeled cells, which can then be separated.
Positive selection versus negative selection of cells
Based on what is being retained or isolated, two types of selections are possible:
- Positive selection of cells
- Negative selection of cells
What is positive selection of cells and why is it important?
Positive selection selects the cells that need to be collected as the target population. The method uses magnetic particles with antibodies targeting a subpopulation of interest covalently bound to their surface. Once placed within the magnet, targeted cells migrate toward the magnet and are retained within the magnetic field while the unlabeled cells are drawn off and discarded. The targeted cells can then be collected and used in the desired application after removal from the magnetic field.
Positive cell selections yield excellent results with respect to purity, recovery, and viability of selected cells. However, depending on the cell type being selected and the surface antigen being targeted by the particle, positive selections can result in cells becoming activated or otherwise functionally altered. Even though the probability of activation is low, this magnetic particle-induced activation may be an issue if you specifically require purified yet unstimulated cells. In that case, you should consider negative selection for your cell separations.
What is negative selection of cells and why is it important?
Negative selection magnetically isolates cells that are not needed, while the target population of cells can be aspirated and collected prior to downstream application, such as cell sorting.
Enrichment by depletion or negative selection is used for research applications that require a cell population with high levels of purity and no antibody or particles bound to their surface.
In this procedure, all unwanted cells are first labeled with a cocktail containing monoclonal antibodies against antigens expressed by them. After washing away unbound antibody, a second-step reagent is used to magnetically label these cells. The labeled cells migrate to the magnet leaving in suspension a pure and untouched subpopulation of cells to be collected. A large percentage (>95%) of unwanted cell populations can be removed through negative selection.1
Enrichment using negative selection is recommended for downstream single-cell multiomic analysis. Such pre-enrichment helps in minimizing manipulation of cells.
Pre-enrichment prior to sorting
Enrichment of cells before sorting is very beneficial for obtaining faster and better sorting results, especially for very rare cell populations. In this procedure, the cells of interest are first enriched through negative selection. The process can remove 20–80% of unwanted cells, thus enriching the untouched cell population of interest and enabling faster and more efficient cell sorting.
What are the main considerations for utilizing magnetic cell separation?
Three factors determine the outcome of cell separation:
- Target cell purity
- Target cell recovery
- Cell viability
Target cell purity
The percentage of target cells collected compared to nontarget cells in the heterogeneous population determines the purity of the collected target cell fraction.
Target cell recovery
The percentage of target cells obtained after sorting compared to the total number of target cells in the original cell suspension determines recovery.
Cell viability
Viability refers to the percentage of cells that are alive and could indicate cell quality after separation.
References
- Plouffe BD, Murthy SK, Lewis LH. Fundamentals and application of magnetic particles in cell isolation and enrichment: a review. Rep Prog Phys. 2015;78(1):016601. doi: 10.1088/0034-4885/78/1/016601
- Tomlinson MJ, Tomlinson S, Yang XB, et al. Cell separation: Terminology and practical considerations. J of Tissue Eng. 2013;4:2041731412472690. doi: 10.1177/2041731412472690
BD Biosciences magnetic separation product portfolio
BD Biosciences offers a variety of reagents and powerful magnets for magnetic cell separation.
Our portfolio includes a selection of magnetic separation reagents for positive and negative selection of cells. Reagents to enrich B lymphocytes, CD4 and CD8 T lymphocytes, NK cells and certain types of murine dendritic cells are available.
BD Biosciences magnetic separation workflow protocol
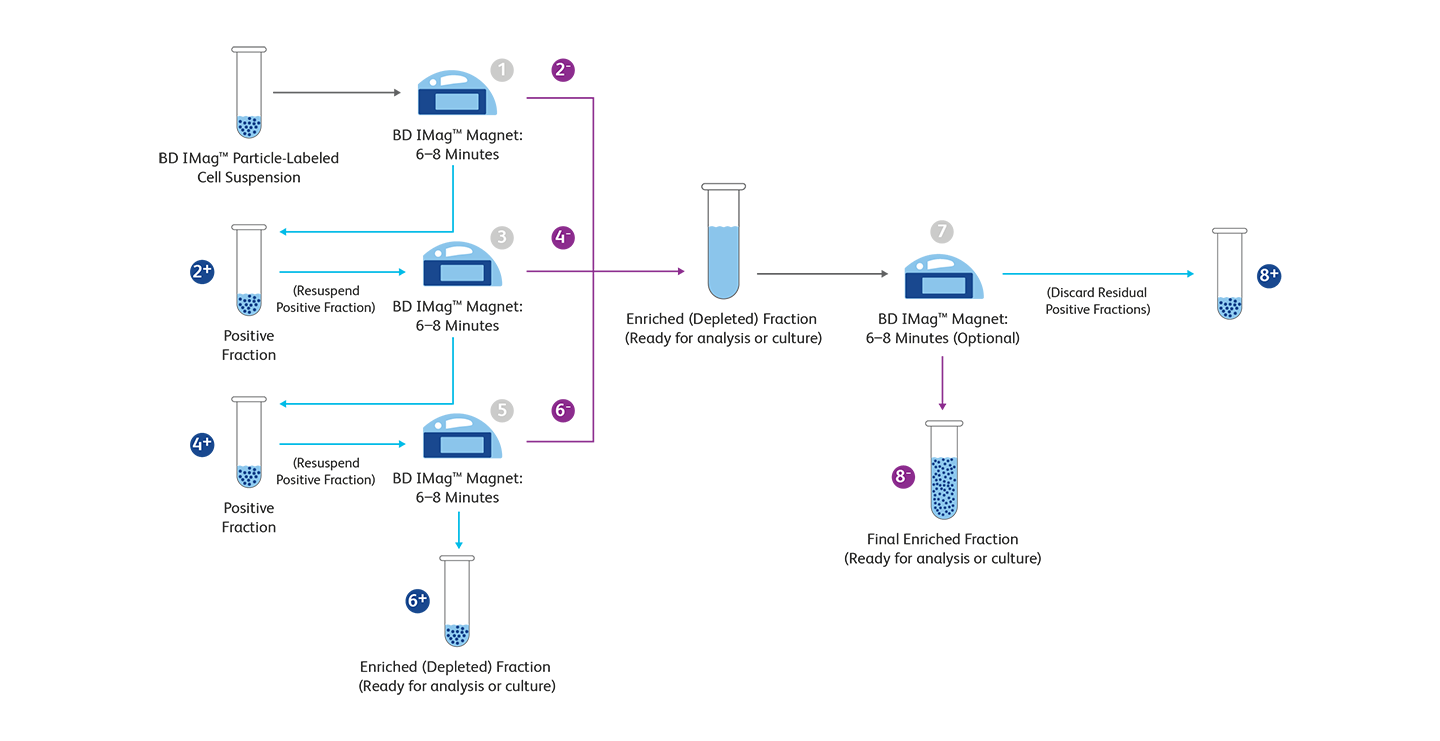
| Step | Positive Selection (+) | Negative Selection (–) |
|---|---|---|
| 1 | Place the BD IMag™ Particle-labeled cell suspension onto the BD IMag™ Magnet. | Place the BD IMag™ Particle-labeled cell suspension onto the BD IMag™ Magnet. |
| 2 | Remove the tube containing the positive fraction. | Remove the supernatant containing the negative fraction and place in an appropriately labeled tube. |
| 3 | Resuspend the positive fraction and place it back onto the BD IMag™ Magnet. | Resuspend the positive fraction and place it back onto the BD IMag™ Magnet. |
| 4 | Remove the tube containing the positive fraction. | Remove the negative fraction and pool with the negative fraction from step 2. |
| 5 | Resuspend the positive fraction and place it back onto the BD IMag™ Magnet. | Resuspend the positive fraction and place it back onto the BD IMag™ Magnet. |
| 6 | Remove the positive fraction and resuspend the cells for use. | Remove the negative fraction and pool with the negative fraction from steps 2 and 4. |
| 7 | Place the combined negative fractions on the BD IMag™ Magnet. | Place the combined negative fractions on the BD IMag™ Magnet. |
| 8 | Remove and discard residual positive fraction. | Remove remaining twice-enriched fraction and place in an appropriately labeled tube for use. |
+ and – refer to the steps in the positive and negative selection process, respectively.
Sample data generated using the BD Biosciences magnetic separation reagents
Expression of activation markers CD25 and CD69 after positive or negative selections
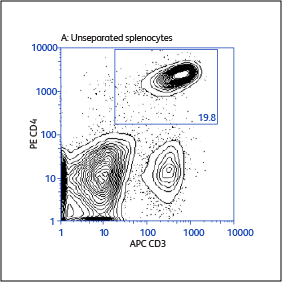
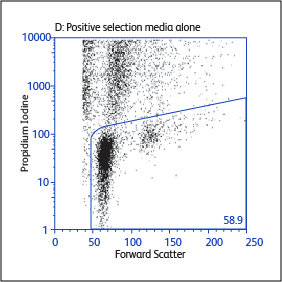
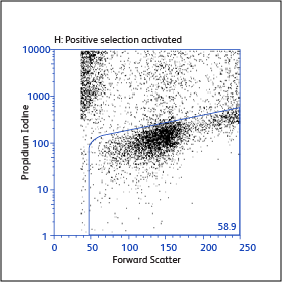
Expression of activation markers CD25 and CD69 after either positive or negative selection (enrichment) of CD4 T cells using BD IMag™ Mouse CD4 Particles–DM and BD IMag™ Mouse CD4 T Lymphocyte Enrichment Set–DM, respectively.
Data showing flexibility of the enrichment protocol
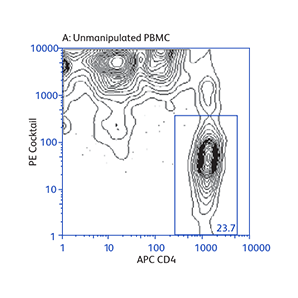
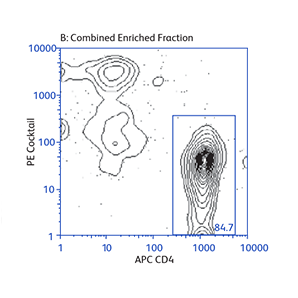
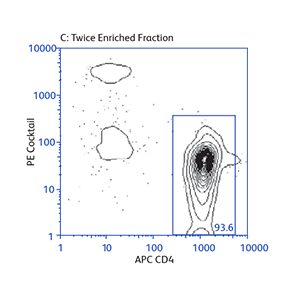
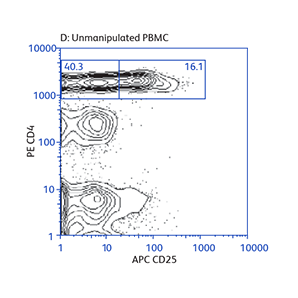

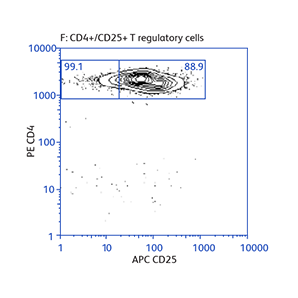
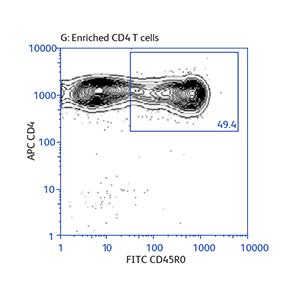
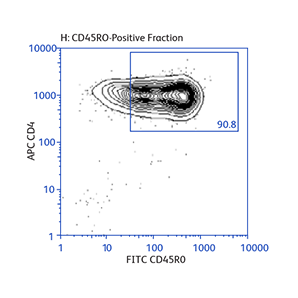
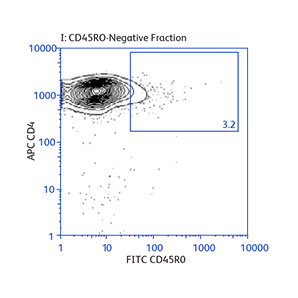
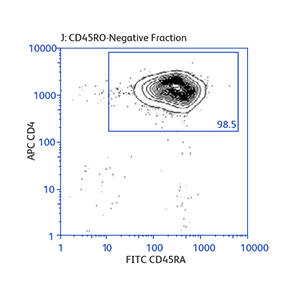
Demonstration of how the basic enrichment protocol can be manipulated for different experimental needs and how positive selections can be coupled with enrichments to isolate uncommon cell subpopulations.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
23-23108-00