-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ CRYO Preservation Buffer
- BD OMICS-Guard™ Sample Preservation Buffer
- BD OMICS-One™ Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD OMICS-One™ WTA Next Assay
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- Advanced Training
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Multiple Sclerosis
Multiple sclerosis (MS) is an inflammatory autoimmune disease of the central nervous system (CNS) affecting both the brain and the spinal cord. It is characterized by demyelination and axonal loss, leading to poor communication between the CNS and the rest of the body. The etiology of MS is unknown; however, genetic and environmental factors seem to play a role in causing immune dysregulation. Major histocompatibility complex (MHC) HLA DR15 and DQ alleles are implicated in MS. Epstein-Barr virus (EBV) infection and vitamin D deficiency are some of the environmental risk factors strongly associated with MS.1
Immunology of MS
Environmental factors, such as EBV infection and genetic susceptibility, could induce, in some individuals, a tendency to destroy the myelin sheet around the nerves, resulting in autoactivation of T cells and release of autoantigens (a process called breach of immune tolerance). A process of molecular mimicry, wherein cross reactivity between endogenous proteins like myelin and exogenous viral proteins ensues, results in further activation of T cells. The activated peripheral T cells then enter the blood brain barrier and get reactivated when they encounter major histocompatibility complex (MHC) class II molecules expressed by dendritic cells, macrophages and B cells, triggering a release of inflammatory cytokines such as interferons and tumor necrosis factor (TNF). This results in additional activation of other immune cells, leading to a persistent activated state of these cells, leading to myelin damage.1
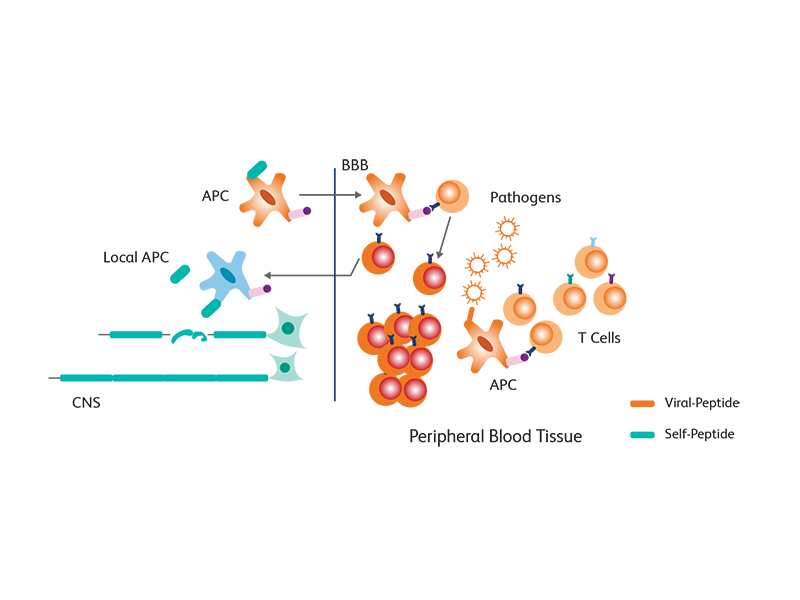
Immune cell subsets associated with pathogenesis in MS
Flow cytometry is an invaluable tool for obtaining insights into the immunological disturbances occurring in MS. Peripheral blood provides a key resource for investigating the type and extent of immunopathogenesis.2 Several immune subsets, such as T and B lymphocyte subsets, have been associated with MS:3
CD4+ T helper (Th) cells
Th17 cells are involved in CNS demyelination by activating inflammatory immune cells on brain epithelial cells. Th cells can be identified using cell surface markers.
CD4+ regulatory (Treg) cells
Treg cells are responsible for regulating the number and function of autoreactive T cells. This balance, which provides immunological tolerance, is compromised in MS. Evaluation of the FoxP3 transcription factor can provide an ability to phenotype Treg cells.
CD4+ follicular T helper (Tfh) and regulatory (Tfr) cells
These cells are involved in the formation and regulation of germinal centers and the differentiation of B cells and elimination of autoreactive B cells.
CD8+ cytotoxic and Treg cells
CD8+ cytotoxic T cells are implicated in the defense against EBV and also in autoreactivity and immune activation associated with MS.
B cells
These cells produce proinflammatory cytokines and autoantibodies to myelin proteins and are also implicated in the activation of autoreactive T cells. CD19 and CD20 counts are also used as markers for evaluating treatment efficacy monoclonal antibodies directed at CD20+ B cells.4
Cytokines
Inflammatory cytokines such as TNF-α, interferon-gamma and interleukin-2 (IL-2) play a key role in mediating inflammation in MS. TNF-α can cross the blood brain barrier and its levels are elevated in the CNS of autoimmune patients. Anti TNF-α blockers are used for CNS demyelination.5
BD Biosciences tools for MS research
BD Biosciences provides an expansive selection of research cell analyzers that can be used on a routine basis in a wide range of applications; and highly advanced, high-parameter research cell analyzers to resolve and analyze rare cell populations and distinctive phenotypes in a heterogeneous cell population.
The dried, unit-sized, preformulated and optimized BD Horizon™ Dri Treg Panel contains markers used for the characterization of FoxP3+ naïve, translational and effector Treg subsets, directly relevant for autoimmune research.
The BD Horizon™ Dri TBNK + CD20 Panel enables efficient characterization of T, B and NK cells.
BD Horizon™ Dyes and Antibodies are ideal for characterizing immune cells surface receptors of varied densities and their brightness makes it easy to distinguish these dim cells from others in a sample.
References
- Sospedra M, Martin R. Immunology of multiple sclerosis. Annu Rev Immunol. 2005;23:683-747. doi: 10.1146/annurev.immunol.23.021704.115707
- Christensen JR, Bornsen L, Ratzer R, et al. Systemic inflammation in progressive multiple sclerosis involves follicular T-helper, Th17 and activated B cells and correlates with progression. PLOS One. 2013;8:e57820. doi: 10.1371/journal.pone.0057820
- Jones AP, Kermode AG, Lucas RM, Carroll WM, Nolan D, Hart PH. Circulating immune cells in multiple sclerosis. Clin Exp Immunol. 2017;187(2):193-203. doi:10.1111/cei.12878
- Barra ME. Experience with long-term rituximab use in a multiple sclerosis clinic. Mult Scler J Exp Transl Clin.
2016;2:2055217316672100. doi: 10.1177/2055217316672100
- Kemanetzoglour E, Andreadou E. CNS demyelination with TNF-α blockers. Curr Neurol Neurosci. 2017;17(4):36. doi: 10.1007/s11910-017-0742-1
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
23-23031-00