-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® OMICS-One Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Promotions
-
Spectral Sorter Promotion
-
BD Primer Program
-
New Lab Promotion
-
BD FACSLyric™ Flow Cytometers 50th Anniversary Promo
-
BD FACSAria™ Customer Loyalty Promotion
-
FlowJo™ Software Promotion
-
BD® Research Cloud Promotion
-
30% off + Free Shipping on BD Horizon Brilliant™ Violet and Ultraviolet Reagents!
-
30% off BD Horizon RealYellow ™ & BD Horizon RealBlue™ Reagents!
-
Spectral Sorter Promotion
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- Advanced Training
-
- BD® OMICS-Guard Sample Preservation Buffer
- BD® OMICS-One Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- United States (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
Applications and Solutions
A comprehensive suite of trusted products, integrated solutions, useful tools and a wealth of information to advance your flow cytometry applications.
Promotions
Innovative solutions to power your diagnostics and research
Backed by cutting-edge technology and more than 50 years of flow cytometry expertise
Utilize our featured resources with invaluable information to advance your science.
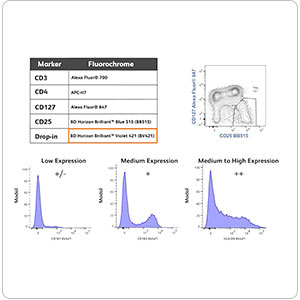
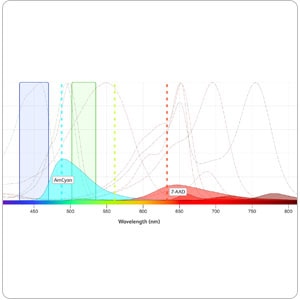
Explore a variety of tools for your multicolor flow cytometry, from panel design to selection tools—an array of aids to support your research.
An interactive tool containing key cell information to help plan your flow cytometry experiments.
A unique ecosystem to collaborate on flow cytometry panel design, improve cytometer efficiency and manage your laboratory.
Houses many of our pre-designed multicolor flow cytometry panels. Search or filter to find and learn more about the panels.
Use our excellent product and technical support with our >50 years of flow cytometry expertise and support resources.
Find a seamless customer experience and an easy way to create and manage accounts.
Explore a variety of ways that work with your purchasing process to place and track orders.
Explore our webinar series spanning a broad range of topics and in-depth reviews by senior scientists, professionals and clinicians.
Use a variety of training courses for instruments and applications to take full advantage of the capabilities of BD products.