-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® OMICS-One Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- Advanced Training
-
- BD® OMICS-Guard Sample Preservation Buffer
- BD® OMICS-One Protein Panels
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
-
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- United States (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?

Instruments
Backed by cutting-edge technology and more than 50 years of expertise, BD Biosciences’ large and extensive portfolio of flow cytometry instruments are designed to empower you with precise tools that can transform your diagnostic, clinical and research applications.
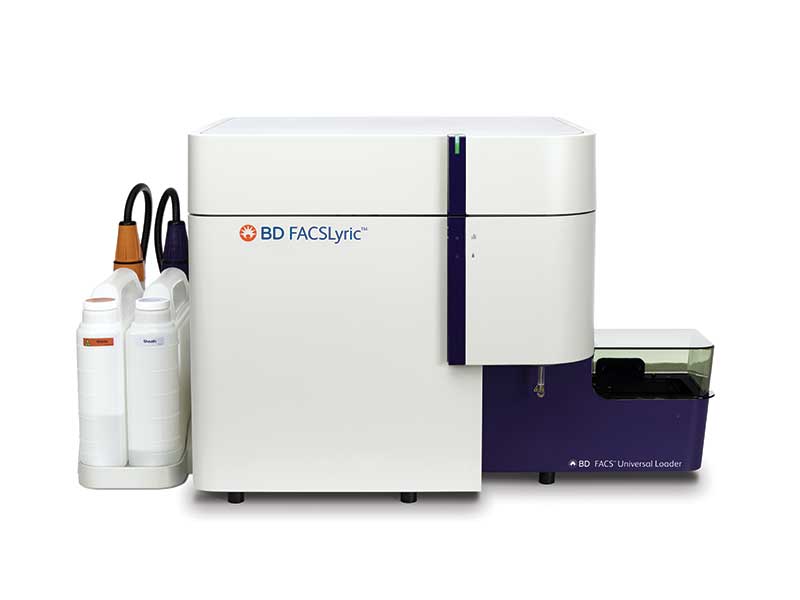
Flow Cytometers
As pioneers in flow cytometry, we at BD Biosciences take pride in the high performance, reliability and reproducibility of our instruments. Transform your clinical lab by enabling easy-to-use inter- and intra-lab standardization with our accurate and flexible flow cytometry systems and intuitive data analysis tools. Further your research needs with our cell analyzers and sorters—and their exceptional multicolor flow cytometry performance.

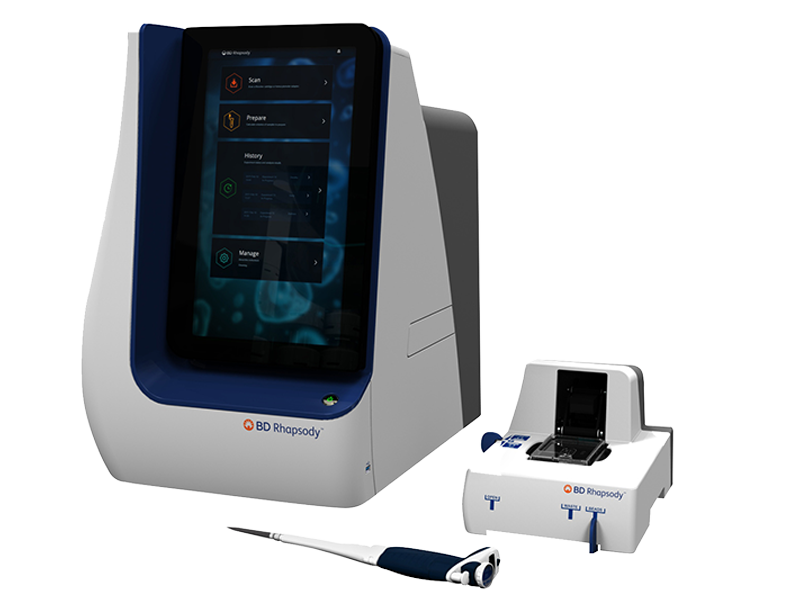
Single-Cell Multiomics Systems
With our single-cell multiomics instruments, you can simultaneously analyze hundreds to thousands of genes, transcripts, clonotypes and proteins at the single-cell level. Dramatically increase your proteomics capabilities by combining single-cell analysis methods with next-generation sequencing using antibodies linked to oligonucleotide barcodes. Find the right tools you need to further your single-cell multiomics research from BD Biosciences, your trusted partner for high-parameter cell sorting and analysis.

Sample Preparation Systems
Automate your sample preparation and minimize errors and variability. As part of our clinical solution, our sample prep systems are designed to work in tandem with our flow cytometry instruments to standardize the processing steps for flow cytometry analysis. Our efficient automation process decreases the number of manual steps necessary to complete your analysis, thereby reducing errors, while maintaining workflow traceability and saving you time.


The BD FACSDuet™ Sample Preparation System is a Class 1 Laser Product.
The BD FACSDuet™ Sample Preparation System is for In Vitro Diagnostic Use.
Sample preparation for user-defined protocols and cocktailing functions are for laboratory use. Not for use in diagnostic or therapeutic procedures. Class 1 Laser Product.
