-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- Advanced Training
-
- BD Accuri™ C6 Plus Cell Analyzer
- BD FACSAria™ Cell Sorter Cell Sorter
- BD FACSCanto™ Cell Analyzer
- BD FACSDiscover™ A8 Cell Analyzer
- BD FACSDiscover™ S8 Cell Sorter
- BD FACSDuet™ Sample Preparation System
- BD FACSLyric™ Cell Analyzer
- BD FACSMelody™ Cell Sorter
- BD FACSymphony™ Cell Analyzer
- BD LSRFortessa™ Cell Analyzer
- United States (English)
-
Change location/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?
BD Transduction Laboratories™ Purified Mouse Anti-Hip1R
Clone 44/Hip1R (RUO)
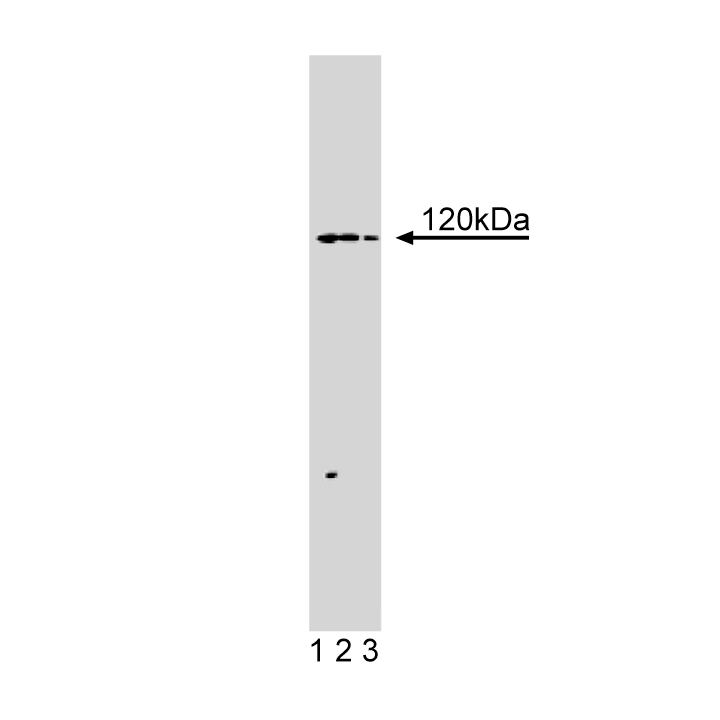



Western blot analysis of Hip1R on BC3H1 lysate. Lane 1: 1:250, lane 2: 1:500, lane 3: 1:1000 dilution of anti-Hip1R.

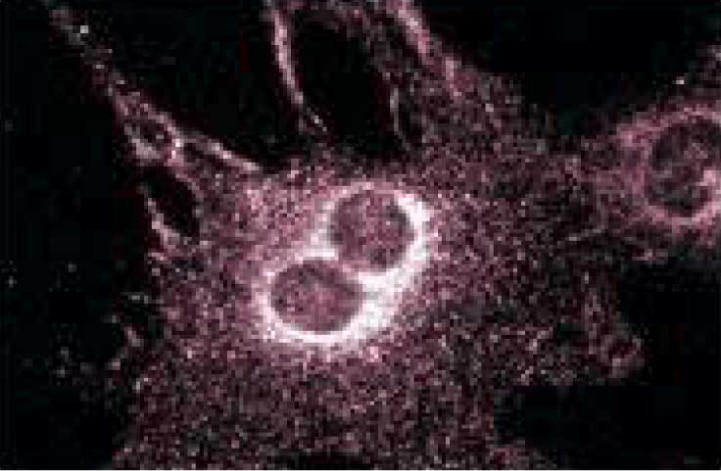
Immunofluorescent staining of BC3H1 cells.




Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Western blot: Please refer to http://www.bdbiosciences.com/pharmingen/protocols/Western_Blotting.shtml.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
Clathrin is the major protein component in the coat formed around pits and vesicles involved in receptor-mediated endocytosis. Clathrin forms a non-covalently bound triskelion structure composed of three heavy chains (192kDa each) and three light chains (23-25kDa each). A variety of proteins facilitate receptor-mediated endocytosis through association with clathrin-coated vesicles. Huntingtin interacting protein 1 (Hip1) is an actin-binding protein that interacts with Huntingtin protein, and has been implicated in vesicular transport defects found in Huntingtin's disease. Hip1 related protein (Hip1R) is another actin binding protein that contains an epsin NH2-terminal homology (ENTH)domain, three coiled-coil regions, a leucine zipper, and a talin-like actin binding domain. Hip1R mRNA is widely expressed, and Hip1R protein is enriched in the cell cortex and perinuclear region. The ENTH domain of Hip1R is required for binding to phosphatidylinositol-4,5-bisphosphate, and this complex is essential for clathrin-mediated endocytosis. In addition, Hip1R colocalizes with clathrin, AP-2 and endocytosed transferrin. Thus, Hip1R may facilitate interactions between clathrin-coated pits and actin during endocytosis.
Development References (3)
-
Engqvist-Goldstein AE, Kessels MM, Chopra VS, Hayden MR, Drubin DG. An actin-binding protein of the Sla2/Huntingtin interacting protein 1 family is a novel component of clathrin-coated pits and vesicles. J Cell Biol. 1999; 147(7):1503-1518. (Biology). View Reference
-
Itoh T, Koshiba S, Kigawa T, Kikuchi A, Yokoyama S, Takenawa T. Role of the ENTH domain in phosphatidylinositol-4,5-bisphosphate binding and endocytosis. Science. 2001; 291(5506):1047-1051. (Biology). View Reference
-
Seki N, Muramatsu M, Sugano S. Cloning, expression analysis, and chromosomal localization of HIP1R, an isolog of huntingtin interacting protein (HIP1). J Hum Genet. 1998; 43(4):268-271. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.