-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays for Human and Mouse
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD FACSDiscover™ S8 Cell Sorter Product Training
- Accuri C6 Plus Product-Based Training
- FACSAria Product Based Training
- FACSCanto Product-Based Training
- FACSLyric Product-Based Training
- FACSMelody Product-Based Training
- FACSymphony Product-Based Training
- HTS Product-Based Training
- LSRFortessa Product-Based Training
- Advanced Training
-
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays for Human and Mouse
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
-
- BD FACSDiscover™ S8 Cell Sorter Product Training
- Accuri C6 Plus Product-Based Training
- FACSAria Product Based Training
- FACSCanto Product-Based Training
- FACSLyric Product-Based Training
- FACSMelody Product-Based Training
- FACSymphony Product-Based Training
- HTS Product-Based Training
- LSRFortessa Product-Based Training
- United States (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

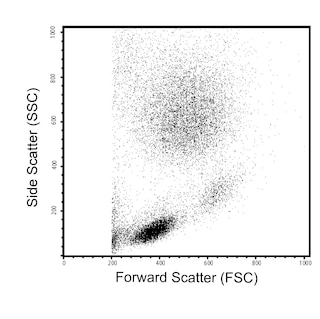
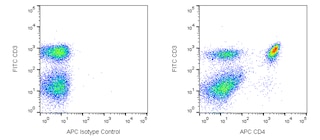
Flow cytometric analysis of PE anti-mouse Foxp3 on mouse splenocytes. Balb/c mouse splenocytes were surface stained with APC Rat Anti-Mouse CD4 (clone RM4-5, Cat. No. 553051), then fixed and permeabilized using working solutions of mouse Foxp3 Buffers (see recommended assay procedure, Cat. No. 560409) followed by intracellular staining with PE Rat anti-Mouse Foxp3 (0.25 µg/test). The dot plots were derived from the gated events based on light scattering characteristics of lymphocytes. Flow cytometry was performed on a BD FACSCalibur™ System.
.png)

BD Pharmingen™ PE Rat anti-Mouse Foxp3
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Cell Preparation and Staining Procedures for Conjugated Anti-Mouse Foxp3 Antibody
1. Prepare working solutions of the BD Pharmingen™ Mouse Foxp3 Buffer Set Cat. No. 560409 (for the buffer preparation, please see TDS
Cat. No. 560409 buffer instructions for details).
2. Pre-warm Permeabilization buffer to 37°C before use. Keep Fixation buffer on ice.
3. Prepare a single-cell suspension from the peripheral lymphoid tissue of interest. Remove clumps of cells and/or debris by passing the
suspension through a 70-µm nylon cell strainer. Use 1 × BD PharmLyse™ Lysing Buffer (Cat. No. 555899) to lyse red blood cells
if necessary. Dilute the cells with BD Pharmingen™ Stain Buffer (FBS, Cat. No. 554656) to ten million cells/ml.
4. Pipette appropriate amount of CD4 or other surface staining reagent(s) to bottom of each 12 x 75 mm tube.
5. Add 100 µl of cells per tube, mix well. Incubate for 20 minutes at RT in the dark.
6. To wash cells, add 2 ml of BD Pharmingen™ Stain Buffer (FBS) to each tube. Centrifuge 250 x g for 10 minutes, and remove buffer.
7. To fix the cells, gently re-suspend pellet in residual volume of staining buffer and then add 2 ml of freshly prepared cold 1 x BD
Pharmingen™ Mouse Foxp3 Fixation Buffer. Mix well. Incubate for 30 minutes at 4°C in the dark.
8. Centrifuge 500 x g for 5 minutes, and remove fixative.
9. To wash cells, re-suspend each pellet in 2 ml of freshly prepared pre-warmed 1 × BD Pharmingen™ Mouse Foxp3 Permeabilization
buffer, and centrifuge 500 x g for 5 minutes. Remove permeabilization buffer.
10. To permeabilize the cells, gently re-suspend pellet in another 2 ml of freshly prepared pre-warmed 1 x BD Pharmingen™ Mouse Foxp3
Permeabilization buffer. Incubate for 30 minutes at 37°C in the dark.
11. Centrifuge 500 x g for 5 minutes, and remove buffer.
12. To wash cells, add 2 ml of BD Pharmingen™ Stain Buffer (FBS) to each tube, centrifuge 500 x g for 5 minutes. Remove buffer.
13. Add 20 µl of conjugated Foxp3 antibody diluted with BD Pharmingen™ Stain Buffer (FBS) at appropriate concentrations (check the
figure legend from each format for the concentration) to re-suspend the pellet. Gently shake or vortex briefly.
14. Incubate for 20 minutes at RT in the dark.
15. Repeat wash step #12 two times.
16. Resuspend the cells in 0.5 ml BD Pharmingen™ Stain Buffer (FBS) and analyze immediately.*
Note: We recommend using the BD Pharmingen™ Stain Buffer for all wash steps and covering tubes during incubation steps with caps or parafilm. We also recommend optimizing forward scatter and side scatter voltages to visualize lymphocytes as separate from debris, red cells, etc. before acquisition.
* Acquire at least 15,000 to 25,000 CD4 positive lymphocytes.
BD® CompBeads can be used as surrogates to assess fluorescence spillover (compensation). When fluorochrome conjugated antibodies are bound to BD® CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cell and BD® CompBeads to ensure that BD® CompBeads are appropriate for your specific cellular application.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
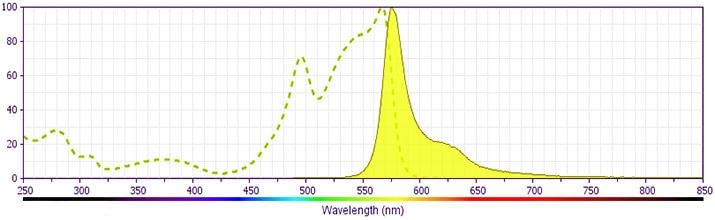
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
Companion Products


Foxp3 is a 50-55 kDa protein also known as Forkhead box P3, JM2, or IPEX. It is a member of the forkhead or winged helix family of
transcription factors and is specifically expressed by T regulatory (Treg) cells. Foxp3 has been reported to be a key regulatory protein for Treg
cell development and function. Ectopic expression of Foxp3 in conventional T cells is sufficient to induce suppressive activity, repress the
production of cytokines such as IL2 and IFN-γ, and upregulate Treg cell-associated molecules such as CD25, CTLA4 and GITR. It has been
found that the mutation of Foxp3 is responsible for "scurfy" mice. When overexpressed, Foxp3 leads to poor T cell proliferation and
activation
Development References (3)
-
Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003; 299(5609):1057-1061. (Biology). View Reference
-
Ono M, Yaguchi H, Ohkura N, et al. Foxp3 controls regulatory T-cell function by interacting with AML1/Runx1. Nature. 2007; 446(7136):685-689. (Biology). View Reference
-
Zheng Y, Rudensky AY. Foxp3 in control of the regulatory T cell lineage. Nat Immunol. 2007; 8:457-462. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.