Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

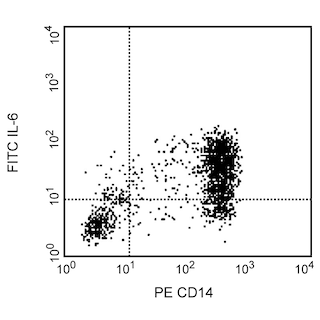
Expression of IFN-γ by stimulated CD8+ and CD8- BALB/c spleen cells. BALB/c spleen cells were cultured for 72h in medium containing Staphylococcus aureus entertoxin B (SEB, 2 µg/ml; Sigma Cat. No. S-4881), recombinant mouse IL-2 (10 U/ml, BD Cat. No. 550069) and recombinant mouse IL-4 (2 ng/ml, BD Cat. No. 550067). The cells were harvested and restimulated for 5 h with immobilized anti-CD3 (145-2C11, BD Cat. No. 553057 at 10 µg/ml) and anti-CD28 (clone 37.51, BD Cat. No. 553294 at 2 µg/ml) antibodies in the presence of BD GolgiStop™ (2 µM final concentration, Cat. No. 554724). The splenocytes were stained with 0.25 µg of PE rat anti-mouse CD8 (PE-53-6.7, Cat. No. 553033), fixed permeabilized, and subseqently stained with 0.1 µg of FITC rat anti-mouse IFN-γ (FITC-XMG1.2, left panel). To demonstrate specificity of staining, the binding by FITC-XMG1.2 was blocked by preincubation of the fixed/permeabilized cells with excess unlabeled XMG1.2 mAb (5 µg; Cat. No. 554409, right panel) prior to staining with the FITC-XMG1.2. The quadrant markers for the bivariate dot plots were set based on the autofluorescence controls and verified using the recombinant cytokine blocking and unlabelled antibody blocking specificity control.
.png)

BD Pharmingen™ FITC Rat Anti-Mouse IFN-γ
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Immunofluorescent Staining and Flow Cytometric Analysis: The FITC-conjugated XMG1.2 antibody can be used for multicolor immunofluorescent staining and flow cytometric analyses to identify and enumerate IFN-γ producing cells within mixed cell populations. For optimal immunofluorescent staining with flow cytometric analysis, this anti-cytokine antibody should be titrated (≤0.5 µg mAb/million cells).
A useful control for deomonstrating specificity of staining is either of the following: (1) pre-block the conjugated XMG1.2 antibody with a molar excess of ligand (e.g., recombinant mouse IFN-γ; Cat. No. 554587) prior to staining, or (2) pre-block the fixed/permeabilized cells with un-conjugated XMG1.2 antibody (Cat. No. 554409) prior to staining. The staining technique and use of blocking controls are described in detail by C. Prussin and D. Metcelfe. A suitable rat IgG1 isotype control for assessing the level of background staining on paraformaldehyde-fixed/saponin-permeabilized mouse cells is FITC-R3-34 (Cat. No. 554684); use at comparable concentrations to antibody of interest.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- An isotype control should be used at the same concentration as the antibody of interest.
Companion Products

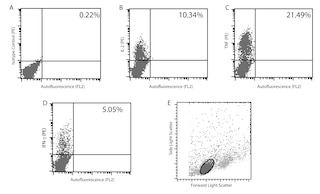

The XMG1.2 monoclonal antibody specifically binds to mouse interferon-γ (IFN-γ) protein. IFN-γ is a pleiotropic cytokine, of approximately 15-17 kDa, involved in the regulation of inflammatory and immune responses. It plays an important role in activation, growth, and differentiation of T and B lymphocytes, macrophages, NK cells and other non-hematopoietic cell types. IFN-γ production is associated with the Th1 cell differentiation. The purified form of this antibody has been reported to be a neutralizing antibody.
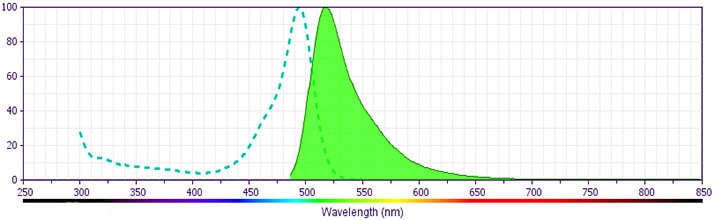
Development References (7)
-
Abrams JS, Roncarolo MG, Yssel H, Andersson U, Gleich GJ, Silver JE. Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples. Immunol Rev. 1992; 127:5-24. (Clone-specific). View Reference
-
Cherwinski HM, Schumacher JH, Brown KD, Mosmann TR. Two types of mouse helper T cell clone. III. Further differences in lymphokine synthesis between Th1 and Th2 clones revealed by RNA hybridization, functionally monospecific bioassays, and monoclonal antibodies. J Exp Med. 1987; 166(5):1229-1244. (Clone-specific). View Reference
-
Ferrick DA, Schrenzel MD, Mulvania T, Hsieh B, Ferlin WG, Lepper H. Differential production of interferon-gamma and interleukin-4 in response to Th1- and Th2-stimulating pathogens by gamma delta T cells in vivo. Nature. 1995; 373(6511):255-257. (Clone-specific: Flow cytometry). View Reference
-
Hsieh B, Schrenzel MD, Mulvania T, Lepper HD, DiMolfetto-Landon L, Ferrick DA. In vivo cytokine production in murine listeriosis. Evidence for immunoregulation by gamma delta+ T cells. J Immunol. 1996; 156(1):232-237. (Clone-specific: Flow cytometry). View Reference
-
Prussin C, Metcalfe DD. Detection of intracytoplasmic cytokine using flow cytometry and directly conjugated anti-cytokine antibodies. J Immunol Methods. 1995; 188(1):117-128. (Methodology: Immunofluorescence). View Reference
-
Sander B, Hoiden I, Andersson U, Moller E, Abrams JS. Similar frequencies and kinetics of cytokine producing cells in murine peripheral blood and spleen. Cytokine detection by immunoassay and intracellular immunostaining. J Immunol Methods. 1993; 166(2):201-214. (Clone-specific). View Reference
-
Vikingsson A, Pederson K, Muller D. Enumeration of IFN-gamma producing lymphocytes by flow cytometry and correlation with quantitative measurement of IFN-gamma. J Immunol Methods. 1994; 173(2):219-228. (Clone-specific: Flow cytometry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.