-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD Rhapsody™ TCR/BCR Profiling Assays for Human and Mouse
- BD® OMICS-One Protein Panels
- BD OMICS-Guard™ CRYO Preservation Buffer
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
Overview
We deliver an integrated portfolio of solutions that helps you accelerate time to insight, streamline scaleup and safeguard manufacturing quality—so your team can focus on what matters mos. From early-stage discovery through commercial manufacturing, BD stands beside you at every stage with the tools, technologies and expertise to help you move forward with clarity, confidence and purpose.
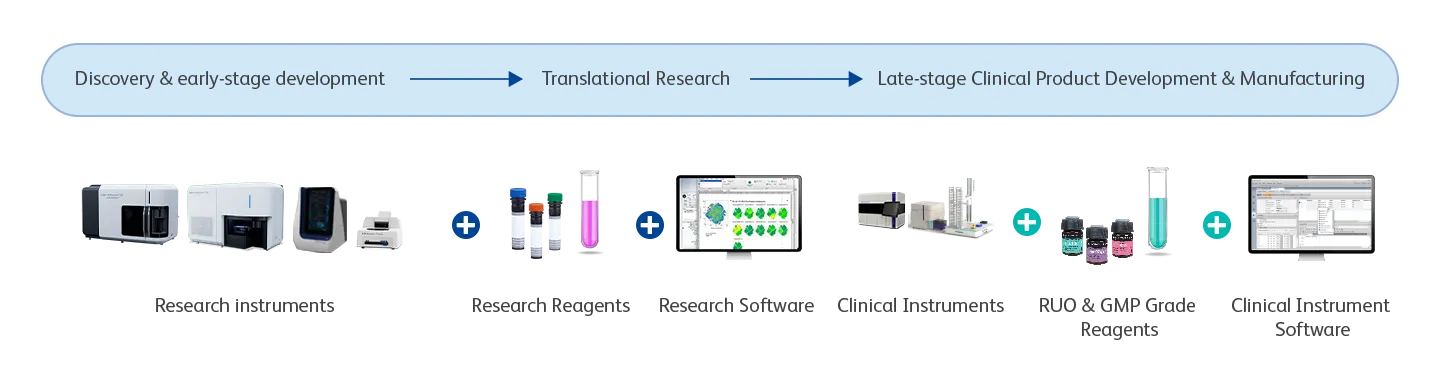
Cell Therapy Solution Portfolio
Backed by industry‑leading technical and scientific experts, BD delivers the precision, scalability and site‑to‑site consistency your cell therapy programs demand. Our comprehensive portfolio of flow cytometry and single‑cell instruments, reagents and software helps you stay competitive —from early discovery through translational development and into full‑scale manufacturing QC.
Accelerate Discovery, Power Cell Therapy Innovation
Cell therapy research is reshaping the future of medicine—and bringing these therapies to market requires unmatched speed, precision and scalability. BD solutions can help you to meet the complex demands your teams face across the entire cell therapy development journey.
With a robust portfolio that includes spectral flow cytometers featuring new-to-world imaging technologies, innovative BD Horizon™ spectral dye fluorochromes, novel BD® CAR Detection Reagents and best-in-class cell analysis software, BD empowers you to confidently advance even the most demanding workflows.
From target identification and validation to process refinement, preclinical studies and GMP process development, BD delivers the tools, technologies and expertise to accelerate your CAR-T and broader cell therapy programs with clarity and consistency.
Late-stage Clinical Product Development and Manufacturing QC Workflow
Optimize Your Manufacturing Processes

BD delivers truly end-to-end commercialization solutions useful for late-stage clinical product development and manufacturing. Our focus is simple: help you drive efficiency, increase productivity and establish repeatable, standardized analytical workflows that scale.
From preanalytical characterization to postanalytical QC, we provide a complete ecosystem—including high‑quality reagents, ready‑to‑use panels, advanced flow cytometry instrumentation and integrated software—to help meet the demands of regulated cell therapy environments.
With fully automated technologies like sample preparation and robotic arm workcell integration, 21 CFR Part 11 features to support customers with their compliance and standardized flow cytometry solutions, we empower your teams to achieve scalable manufacturing QC, site-to-site data consistency and seamless technology transfer from R&D to full commercial production.
Learn more about BD cell therapy solution supporting late-stage manufacturing
Accelerate Discovery, Power Cell Therapy Innovation
Cell therapy research is reshaping the future of medicine—and bringing these therapies to market requires unmatched speed, precision and scalability. BD solutions can help you to meet the complex demands your teams face across the entire cell therapy development journey.
With a robust portfolio that includes spectral flow cytometers featuring new-to-world imaging technologies, innovative BD Horizon™ spectral dye fluorochromes, novel BD® CAR Detection Reagents and best-in-class cell analysis software, BD empowers you to confidently advance even the most demanding workflows.
From target identification and validation to process refinement, preclinical studies and GMP process development, BD delivers the tools, technologies and expertise to accelerate your CAR-T and broader cell therapy programs with clarity and consistency.
Late-stage Clinical Product Development and Manufacturing QC Workflow
Optimize Your Manufacturing Processes

BD delivers truly end-to-end commercialization solutions useful for late-stage clinical product development and manufacturing. Our focus is simple: help you drive efficiency, increase productivity and establish repeatable, standardized analytical workflows that scale.
From preanalytical characterization to postanalytical QC, we provide a complete ecosystem—including high‑quality reagents, ready‑to‑use panels, advanced flow cytometry instrumentation and integrated software—to help meet the demands of regulated cell therapy environments.
With fully automated technologies like sample preparation and robotic arm workcell integration, 21 CFR Part 11 features to support customers with their compliance and standardized flow cytometry solutions, we empower your teams to achieve scalable manufacturing QC, site-to-site data consistency and seamless technology transfer from R&D to full commercial production.
Learn more about BD cell therapy solution supporting late-stage manufacturing
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
BD Flow Cytometers are Class I Laser Products.