-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD Rhapsody™ TCR/BCR Profiling Assays for Human and Mouse
- BD® OMICS-One Protein Panels
- BD OMICS-Guard™ CRYO Preservation Buffer
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?

CD56 (NCAM)
CD56 (NCAM)
CD56 is also known as neural cell adhesion molecule 1 (NCAM-1), neural cell adhesion molecule (NCAM) or embryonic NCAM (E-NCAM).1 It is a member of the immunoglobulin superfamily and expressed on nearly all tissues. Depending on the cell type, three primary isoforms (NCAM-120, NCAM-140 and NCAM-180) are generated by alternative splicing from one single gene NCAM1.2 BD carries several anti-CD56 pan antibody clones for CD56 detection in human, non-human primates and mouse targets.
Function
CD56 is a cell adhesion molecule involved in cell-to-cell and cell-matrix interactions during development and differentiation. CD56 interacts with components of the extracellular matrix such as fibroblast growth factor receptors and N-cadherin. It also triggers signaling cascades involving FYN-focal adhesion kinase (FAK), mitogen-activated protein kinase (MAPK) and phosphatidylinositol 3-kinase (PI3K).
During nervous system development, CD56 regulates neurogenesis, neurite outgrowth, axonal guidance, synapse formation and cell migration. In the hematopoietic system, CD56 is involved in the expansion of T lymphocytes, B lymphocytes and natural killer (NK) cells.3,4 In humans, CD56 is a common phenotypic marker for NK cells.
Structure
The CD56 isoforms are heavily glycosylated proteins named by their molecular weights.5 The larger isoforms are transmembrane proteins while NCAM-120 has no intracellular residues.
- NCAM-180 has a large cytoplasmic domain.
- NCAM-140 has a short cytoplasmic tail.
- NCAM-120 is glycophosphatidylinositol (GPI)-linked to the cell membrane.
NCAM-120 is the predominant isoform in immune cells.6
CD56 in Diseases
CD56 can be used as a biomarker to detect malignant tumors in the nervous system (e.g., medulloblastoma and astrocytoma), malignant NK/T-cell lymphomas and neuroendocrine carcinoma.7 Due to its overexpression in several cancers, anti-CD56 antibody-drug conjugates, such as lorvotuzumab mertansine7 and promiximab-duocarmycin, have been developed.8 CD56 can also be a surrogate marker for other diseases such as COVID-19 and dementia associated with Alzheimer’s disease.9
Antigen Distribution
CD56 is expressed in brain, on CD16+ natural killer (NK) cells, T cell subsets and dendritic cells but not on myeloid cells, erythrocytes or B cells. CD56 expression on NK lymphocytes increases upon activation.7 CD56 expression on non-human primate lymphocytes is similar to that observed on human peripheral blood lymphocytes, with a subset of CD16+ cells co-expressing CD56.10 Clone MY31 is appropriate for detection of CD56 in non-human primates.
CD56 and CD16 expression can be used to characterize NK cell populations. CD16+CD56dim NK cells make up 90% of peripheral blood NK cell populations that can migrate to sites of inflammation. They are characterized by their cytolytic activity, expressing high levels of granzymes, perforin and cytolytic granules but low levels of cytokines. Conversely, CD16-CD56bright NK cells produce cytokines such as IFN-γ, TNF-α, GM-CSF, IL-10 and IL-13. They migrate to secondary lymphoid organs and have low cytotoxic properties.10
Demonstrated Applications of CD56 Detection
Our in-house clone R19-760 was developed to be compatible with select fixation and permeabilization buffers in flow cytometry applications. BD scientists incorporated R19-760 into a 16-color antibody panel to characterize subsets of cytotoxic T cells and NK cells. The cytolytic potential of these cell populations were analyzed through the detection of granzyme K, granzyme B and perforin.11
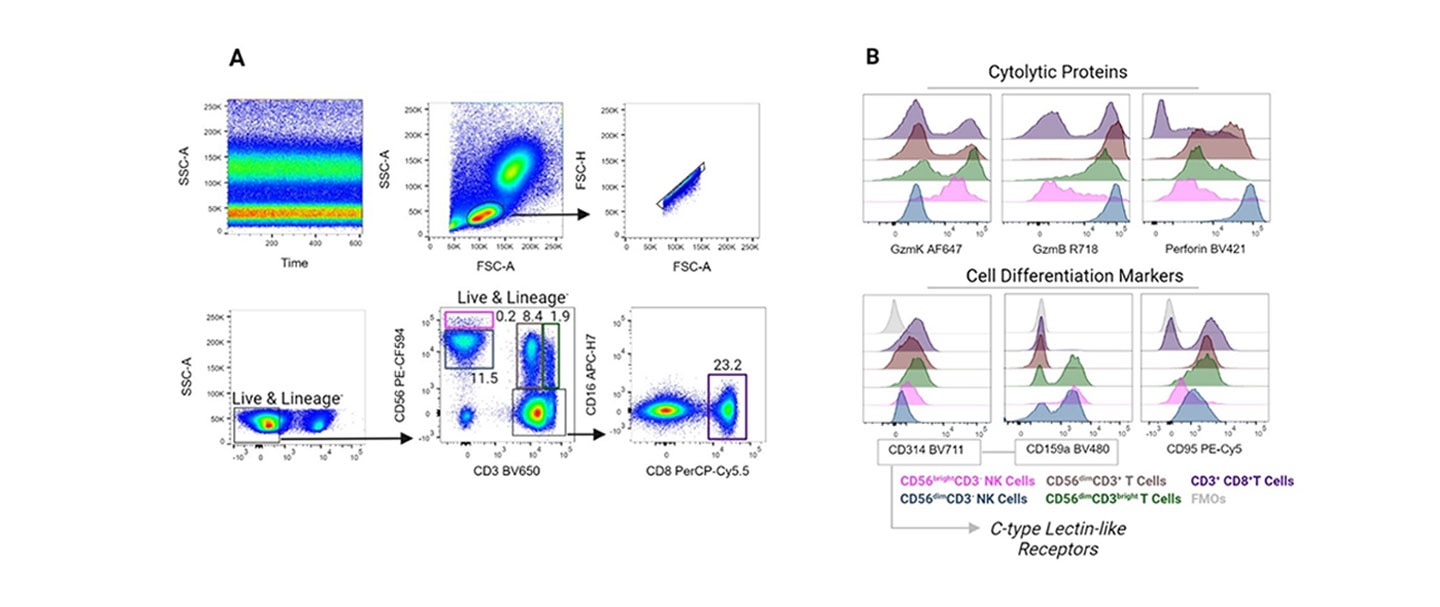
References
- Schlossman SF. Leucocyte Typing V: White Cell Differentiation Antigens: Proceedings of the Fifth International Workshop and Conference, Held in Boston, USA, 3-7 November, 1993. Oxford: Oxford University Press; 1995.
- Bennett IM, Zatsepina O, Zamai L, Azzoni L, Mikheeva T, Perussia B. Definition of a natural killer NKR-P1A+/CD56-/CD16- functionally immature human NK cell subset that differentiates in vitro in the presence of interleukin 12. J Exp Med. 1996;184(5):1845-1856. doi:10.1084/jem.184.5.1845
- Crotta S, Stilla A, Wack A, et al. Inhibition of natural killer cells through engagement of CD81 by the major hepatitis C virus envelope protein. J Exp Med. 2001;195(1):35-42. doi:10.1084/jem.20011124
- Umezawa Y, Kuge S, Kikyo N, et al. Identity of brain-associated small cell lung cancer antigen and the CD56 (NKH-1/Leu-19) leukocyte differentiation antigen and the neural cell adhesion molecule. Jpn J Clin Oncol. 1991. doi:10.1093/oxfordjournals.jjco.a039467
- Van Acker HH, Van Acker ZP, Versteven M, et al. CD56 homodimerization and participation in anti-tumor immune effector cell functioning: A role for interleukin-15. Cancers. 2019;11(7):1029. doi:10.3390/cancers11071029
- Yu L, Lu Y, Yao Y, et al. Promiximab-duocarmycin, a new CD56 antibody-drug conjugates, is highly efficacious in small cell lung cancer xenograft models. Oncotarget. 2017;9(4):5197-5207. doi:10.18632/oncotarget.23708
- Solana C, Tarazona R, Solana R. Immunosenescence of natural killer cells, inflammation, and Alzheimer’s disease. Int J Alzheimer’s Dis. 2018;2018:1-9. doi:10.1155/2018/3128758
- Hong HS, Rajakumar PA, Billingsley JM, Reeves RK, Johnson RP. No monkey business: Why studying NK cells in non-human primates pays off. Front Immunol. 2013;4. doi:10.3389/fimmu.2013.00032
- Baracho GV, Kara N, Rigaud S, Lo E, Widmann SJ, Tyznik AJ. Functional phenotyping of circulating human cytotoxic T cells and NK cells using a 16-color flow cytometry panel. STAR Protocols. 2022;3(1):101069. doi:10.1016/j.xpro.2021.101069
Alexa Fluor™ is a trademark of Life Technologies Corporation.
CD56 Clones and Applications
CD56-specific antibody-binding epitopes can be disrupted when exposed to fixatives, limiting the application of some antibody reagents in intracellular staining workflows. BD offers three clones (MY31, B159 and NCAM16.2) that optimally detect CD56 in unfixed cells. BD has developed clone R19-760 for CD56 detection in unfixed and fixed cells to aid in intracellular staining. Furthermore, clone R19-760 competes with MY31 and NCAM16.2 for CD56 binding, and partially competes with B159, suggesting these clones are possible candidates for receptor occupancy assays.
Clone comparison summary of anti-human CD56 antibodies in the BD catalog.
| Anti-Human CD56 Clones | ||||
|---|---|---|---|---|
| MY31 | B159 | NCAM16.2 | R19-760 | |
| Epitope/immunogen | 220/135 kDa heavily glycosylated antigen from KG1α cell line | Human NK cells | Extracellular immunoglobulin-like domain of human brain | Extracellular domain of recombinant CD56 |
| Cross-reactivity | Baboon, Rhesus, Cynomolgus | None | None | None |
| Isotype | Mouse IgG1, κ | Mouse IgG1, κ | Mouse IgG1, κ | Mouse IgG1, κ |
| Workshop | V NK19 | V NK79 | V NK60 | NA |
| Application | Flow cytometry | Flow cytometry | Flow cytometry | Flow cytometry (intracellular) |
| Staining after cell fixation | No | No | No | Yes |
| Non-competing clones | NCAM16.2, R19-760 | NCAM16.2 | MY31, B159, R19-760 | NCAM16.2, MY31 |
*Compatible with BD Cytofix™ Fixation Buffer, BD Phosflow™ Lyse/Fix Buffer, BD Phosflow™ Perm Buffer I and BD Phosflow™ Perm Buffer III.
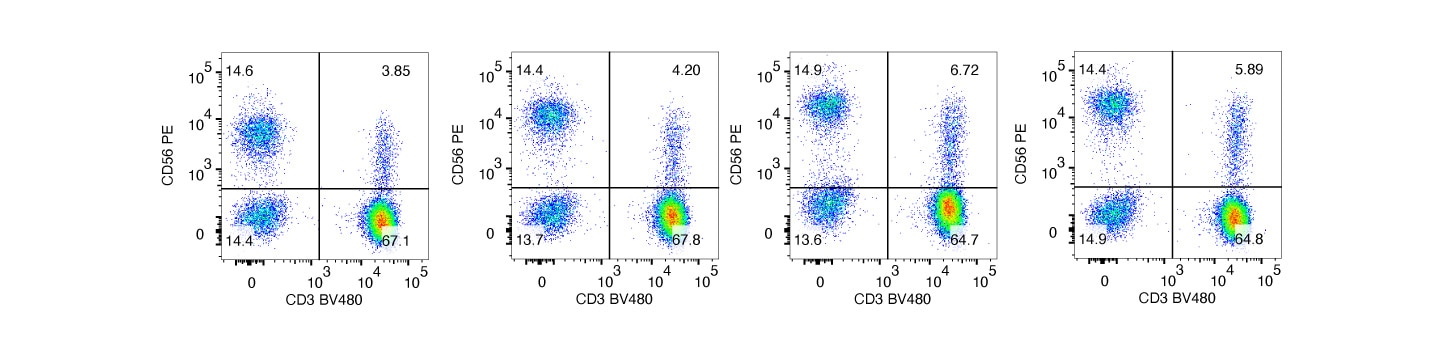
All four clones similarly detected surface CD56 on human peripheral blood.
Analysis of CD56 expression using different anti-CD56 clones
Human whole blood was stained with BD Horizon™ Mouse Anti-Human CD3 and optimal concentrations of PE mouse anti-human CD56 clones: MY31, B159, NCAM16.2 or R19-760 (left to right figures, respectively). Bivariate pseudocolor density plots showing CD3 vs CD56 expression were derived from gated events with the side and forward-light scattering characteristics of intact leucocyte populations. Samples were acquired on a BD FACSymphony™ A5 SE Flow Cytometer and data were analyzed via traditional compensation.
Compatibility with Fixation and Permeabilization Buffers
CD56 Staining in Intracellular Workflows
Intracellular flow cytometry requires cells to be treated with fixation and permeabilization buffers, which may affect antibody binding. Clone R19-760 was developed, and is routinely tested, for CD56 detection in intracellular workflows. The following figures show that R19-760 is compatible with BD Cytofix™ Fixation Buffer, BD Phosflow™ Lyse/Fix Buffer and BD Phosflow™ Perm Buffer III.
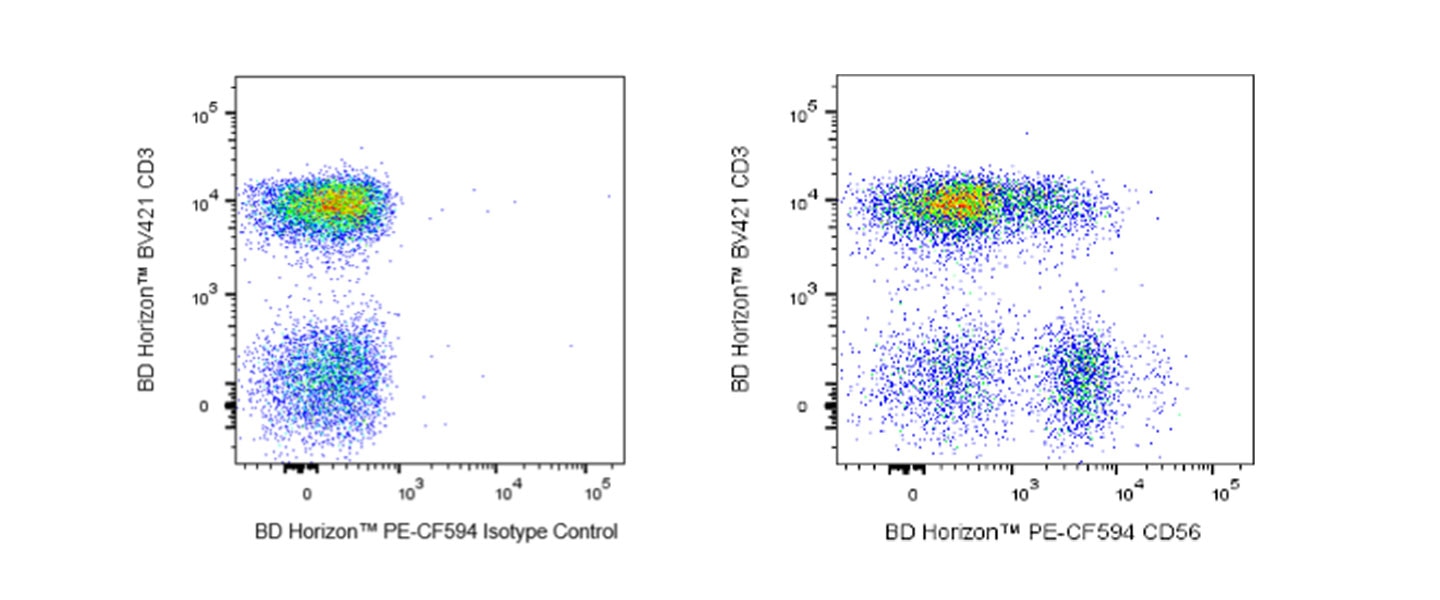
Staining of human peripheral blood lymphocytes with anti-CD56, clone R19-760 after cell fixation and permeabilization.
Whole blood was treated with BD Phosflow™ Lyse/Fix Buffer (10 min, 37 °C) to lyse erythrocytes and fix leucocytes, followed by BD Phosflow™ Perm Buffer III (30 min, on ice) to permeabilize leucocytes. The cells were then stained with BD Horizon™ PE-CF594 Mouse Anti-Human NCAM-1 (CD56) clone R19-760 (Cat. No. 564963) and BD Horizon™ BV421 Mouse Anti-Human CD3 (Cat. No. 562426/562427) antibodies. The bivariate pseudocolor density plot showing correlated CD56 versus CD3 expression was derived from gated events with the forward and side light-scatter characteristics of intact peripheral blood lymphocytes. Flow cytometry and data analysis were performed using a BD LSRFortessa™ Cell Analyzer System and FlowJo™ Software.
Surface Staining of Fixed Cells
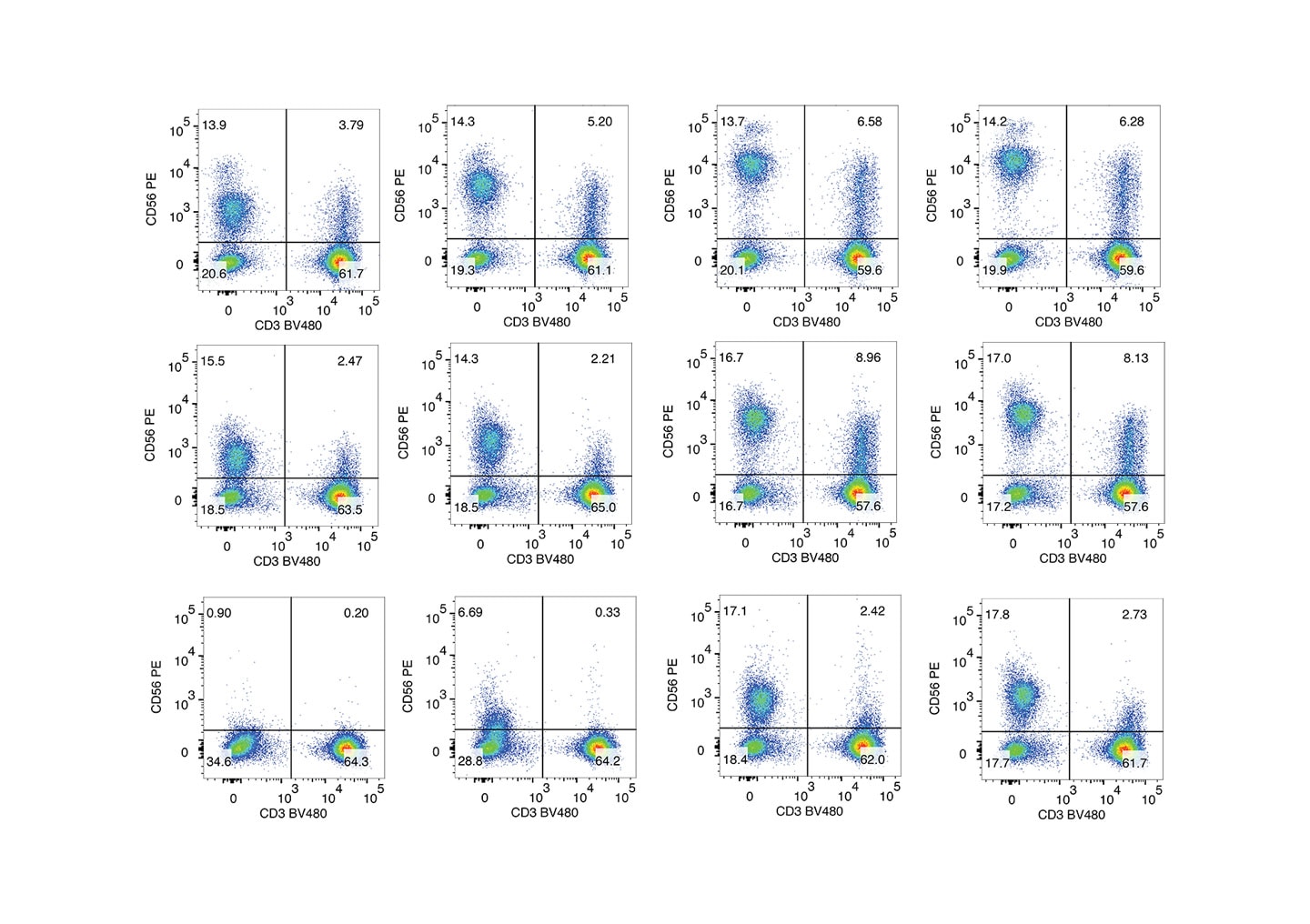
Clone R19-760 was developed to increase the flexibility of intracellular workflow design by staining similarly on unfixed and fixed cells. The following figures represent two possible workflows: CD56 staining on unfixed cells, CD56 staining on fixed cells. In contrast to clones MY31, B159, and NCAM16.2, R19-760 staining is less affected by cell fixation.
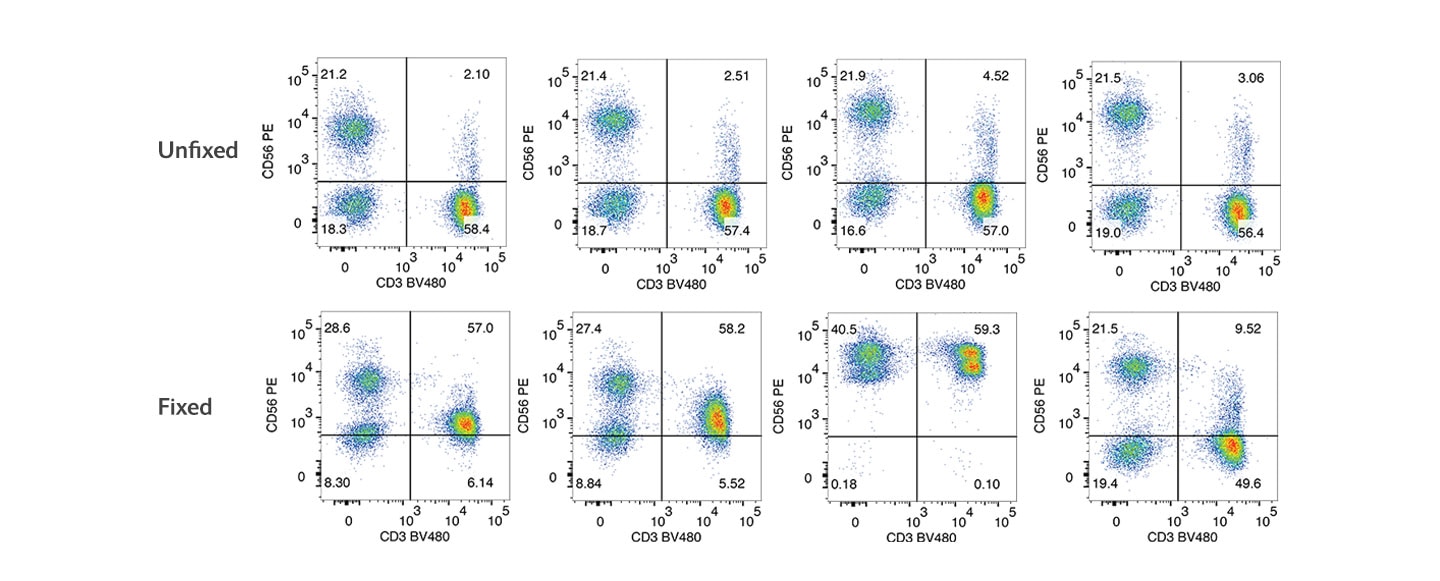
Staining of human PBMCs with different anti-CD56 antibodies.
PBMCs were stained after isolation (unfixed) or fixed with 1X BD Cytofix™ Fixation Buffer for 30 minutes at 4 °C. Both unfixed and fixed cells were stained with BD Horizon™ BV480 Mouse Anti-Human CD3 and one of the PE mouse anti-human CD56 clones: MY31, B159, NCAM16.2 or R19-760. Bivariate pseudocolor density plots were derived from gated events with the side and forwards light-scattering characteristics of intact cells. Samples were acquired on a BD FACSymphony™ A5 SE Flow Cytometer and data were analyzed via traditional compensation.
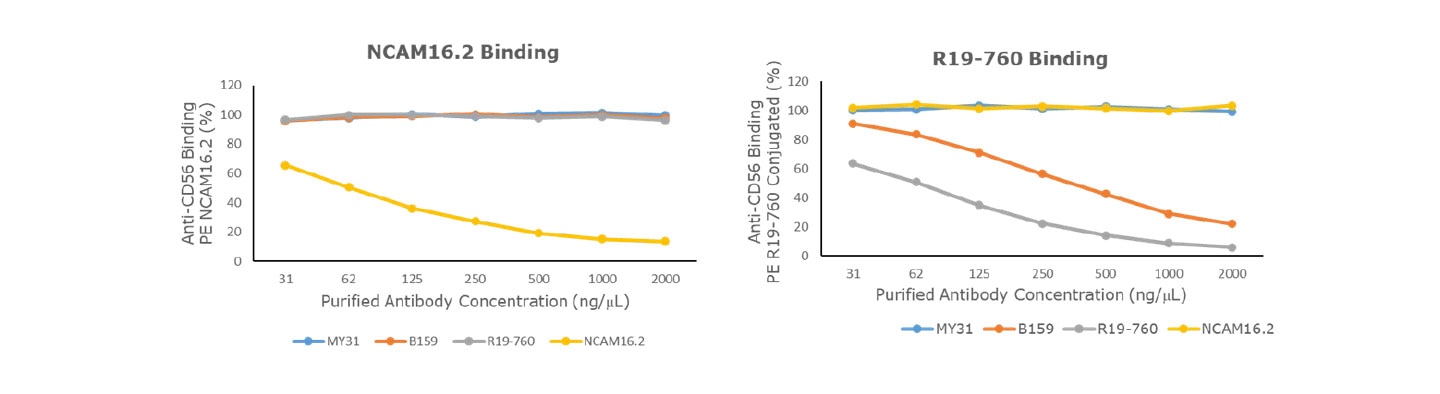
Competitive Binding Assays
Non-competing antibodies against the same antigen can be used in competitive binding assays to measure receptor occupancy. Clone NCAM16.2 does not compete with R19-760, B159 and MY31 for CD56 binding. R19-760 does not compete with MY31 or NCAM16.2 but partially competes with B159.
| MY31 | B159 | NCAM16.2 | R19-760 | |
|---|---|---|---|---|
MY31 | ND | _ | _ | |
B159 | ND |
| _ | + |
NCAM16.2 | _ | _ | _ | |
R19-760 | _ | + | _ |
Epitope recognition overlap of anti-human CD56 clones for use in competitive binding assays.
- = no competition, + = partial competition, ND = no data
Competitive binding assay of anti-human CD56 antibodies
Compatibility with Collagenase Treatment
Collagenase treatment is routinely used to produce single-cell suspensions from tissues. The following figure shows the effect of collagenase on the detection of CD56 on human PBMCs.1 After 60 min of collagenase treatment, none of the clones detected CD56 on CD3+ cells. However, clones R19-760 and NCAM-16.2 enabled CD56 resolution on CD3- cells. At 30 min collagenase treatment, R19-760 and NCAM-16.2 provided greater separation of both CD3+CD56+ and CD3-CD56+ cells compared to the other clones.
References
- Frutoso M, Mair F, Prlic M. OMIP-070: NKp46-Based 27-Color Phenotyping to Define Natural Killer Cells Isolated From Human Tumor Tissues. Cytometry A. 2020;97(10):1052-1056. doi:10.1002/cyto.a.24230
CF™ is a trademark of Biotium, Inc.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.