-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?
Request a Quote BD LSRFortessa™ Cell Analyzer
Please fill in the following information and we will get in touch with you regarding your query.
For Professionals in Research
The BD LSRFortessa™ Cell Analyzer offers the ultimate in choice for flow cytometry, providing power, performance and consistency.
The system is an innovative and proven platform for multicolor analysis with its powerful fluidics systems and novel optics design, all of which work to maximize signal and enable minimum cross talk.
BD FACSDiva™ Software controls the efficient setup, acquisition and analysis of flow cytometry data from the BD LSRFortessa™ Workstation. The software is common across BD FACS™ instrument families, including the BD FACSCanto™ Cell Analyzer and BD FACSAria™ Cell Sorter systems. This affords greater application flexibility allowing you to easily move assays from one platform to another.
If you own our legacy BD® LSR II Flow Cytometer, you can take advantage of our exclusive special offers for trading in your BD® LSR II System. Contact our Technical and Applications Support personnel for maintaining optimal instrument performance and with any other instrument-related support.
View the BD LSRFortessa™ System brochure.

Features
The BD LSRFortessa™ System’s innovative excitation and collection optics design reduces excitation losses and optimizes collection efficiency for increased sensitivity and resolution

- The octagon- and trigon-shaped optical pathways of collection optics maximize signal detection and increase sensitivity and resolution allowing you to identify dim and rare cell populations
- Can be configured with up to 5 lasers—blue, red, violet, UV and yellow-green. This flexibility in laser wavelengths allows you to optimize assay design using the latest fluorescent dyes and substrates
- Can accommodate the detection of up to 18 colors simultaneously with a defined set of optical filters that meet or exceed the majority of today’s assay requirements
Get more information on the excitation and collection optics of the BD LSRFortessa™ System from the system brochure.
The BD LSRFortessa™ System’s fluidics system features a true fixed alignment flow cell that is gel-coupled to the collection optics to maximize detector signal

- The fluidics design ensures that the laser is precisely focused on the sample stream and maximum amount of emitted light can be collected for added sensitivity in multicolor applications
- Fixed alignment also minimizes startup time, improves experiment-to-experiment reproducibility and enables automated daily quality control
- The optional BD FACSFlow™ Supply System Fluidics Cart increases capacity and ease of use while maintaining a stable fluidics pressure. It includes an automated sheath and waste fluid control system that reduces daily maintenance by incorporating two 20-L containers (Cubitainers™)
- Fluidic sensors maintain constant pressure, while a fluidics monitoring system warns when sheath fluid is low or empty or when the waste container is full
Get more information on the fluidics system of the BD LSRFortessa™ System from the system brochure.
The BD® High Throughput Sampler (HTS) option improves experimental workflow and the BD Special Order Research Product (SORP) program provides flexibility

The HTS Option
HTS provides rapid, fully automated sample acquisition from 96- and 384-well microtiter plates. In high-throughput mode, the HTS option can speed through a 96-well plate in less than 15 minutes with less than 0.5% sample carryover from one well to the next. Low carryover is essential in research applications to ensure sample purity and data integrity.
Fast acquisition speed is achieved by synchronizing two high-precision pumps for sample mixing, sample injection and probe washing. Standard throughput mode can be selected for acquisition of larger sample volumes.
Special Order Research Product (SORP) Program for Custom Solutions
The BD Special Order Research Product program allows customers to configure BD flow cytometers and cell sorters to fit precise research and assay needs.
Tailored to the specific needs of research at the leading edge of biomedical discovery, the program offers a wide range of choices to help researchers create the ultimate customized instrument for their requirements.
Learn more about BD custom solutions for flow cytometry.
APPLICATIONS
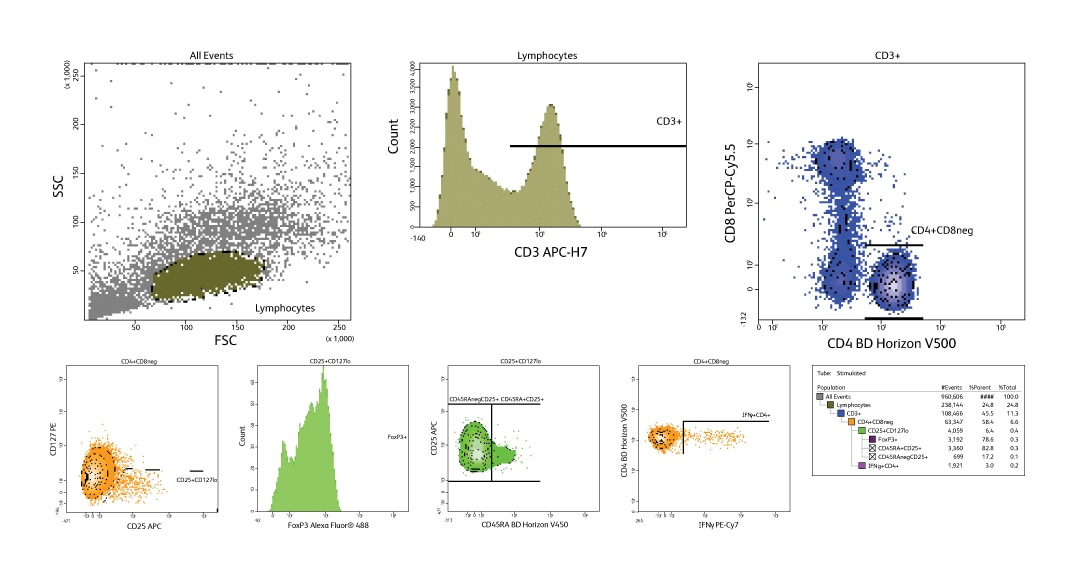
Regulatory T cells (Tregs) play a critical role in maintaining immune modulation and are present in normal peripheral blood in low numbers (5 to 10% of CD4+ T cells or 1 to 2% of total lymphocytes). Forkhead box P3 (FoxP3) is considered to be the definitive marker for this rare cell population. Tregs are known to express the highest levels of CD25. The CD4+CD25+(high) gating strategy shown here was used to identify Treg populations. These plots represent data taken from a BD LSRFortessa™ System using an 8-color panel that applies this gating strategy to identify FoxP3-positive cells.
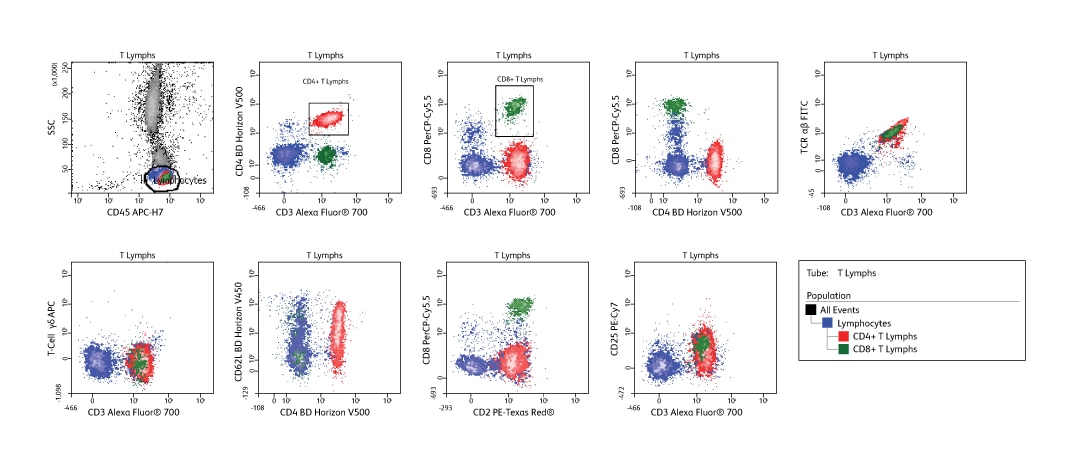
Data show a 10-color panel run on a 3-laser BD LSRFortessa™ System with a blue, red, violet configuration. The panel was used for defining T cell subsets using lysed whole blood (BD Pharm Lyse™ Lysing Buffer) from a normal donor. Use of the BD Horizon™ V450 and BD Horizon™ V500 Dyes allows brighter fluorochromes to be used for more dimly expressed antigens, while the use of 10 colors expands the amount of information gained from a single tube.
-
Brochures
-
Filter Guides
-
Technical Specifications
-
Product Information Sheets
-
Quick Reference Guides
-
BD FACSDiva™ Software v8.0 Administrative Tasks
-
BD FACSDiva™ Software v8.0 for BD LSRFortessa™ Flow Cytometers
-
BD FACSDiva™ Software v8.0 for BD LSRFortessa™ Flow Cytometers (with HTS Option)
-
BD FACSDiva™ Software v6.0: Administrative Tasks
-
BD FACSDiva™ Software v6 (with SPA II and LWA Sample Preppers)
-
BD FACSDiva™ Software v6 (with SPA III and LWA Sample Preppers)
-
BD FACSDiva™ Software v6.0 for BD™ LSR II
-
BD FACSDiva™ Software v6.0 for BD™ LSR (with HTS Option)
-
BD FACSDiva™ Software v6.0 for BD FACSAria™
-
BD FACSDiva™ Software v6.0 for BD FACSCanto™
-
BD FACSDiva™ Software v6.0 for BD FACSCanto™ Loader Option
-
BD FACSDiva™ Software v6.0 for BD FACSCanto™ (with HTS Option)
-
BD FACSDiva™ Software 6.1 for BD FACSAria™
-
BD FACSDiva™ Software v6.1 for BD FACSAria™: Features
-
User's Guides and Manuals
-
Application Notes
BD flow cytometers are Class I (1) laser products.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Purchase does not include or carry any right to resell or transfer this product either as a stand-alone product or as a component of another product. Any use of this product other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited. Product availability and prices are subject to change without notice.
Alexa Fluor®, Texas Red™ and Pacific Blue™ are trademarks of Thermo Fisher Scientific.
CF is a trademark of Biotium, Inc. Cy™ is a trademark of GE Healthcare.
Cy™ dyes are subject to proprietary rights of GE Healthcare and Carnegie Mellon University and are made and sold under license from GE Healthcare only for research and in vitro diagnostic use.
Any other use requires a commercial sublicense from GE Healthcare, 800 Centennial Avenue, Piscataway, NJ 08855-1327, USA.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
