-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Leukemia
For Professionals in Clinical Diagnostics
In leukaemia, immature or abnormal cells undergo uncontrolled expansion, resulting in an overproduction of these white blood cells and frequently the inability to produce enough red blood cells and platelets. In the case of acute leukaemias, the normal differentiation pathway is blocked at a stage where the cells are still proliferating but do not move into terminal differentiation. According to the 2018 GLOBOCAN survey, cases of leukaemia are on the rise worldwide: 437,033 new cases of leukaemia were registered with a global incidence rate of 5.2 and a mortality rate of 3.5 (ASR per 100,000).1 Based on the progenitor cell in the bone marrow, leukaemia can be lymphocytic or myelocytic. Lymphocytic (lymphoblastic) leukaemia arises from T or B lymphocytic progenitors whereas non-lymphocytic or myeloid leukaemia arises from progenitors of erythrocytes, granulocytes, monocytes or platelets.1
What causes leukemia?
Leukemia is caused by genetic and metabolic alterations in lymphocyte or myeloid progenitors in the bone marrow. Environmental and lifestyle risks factors of leukemia include smoking, indoor and outdoor air pollution, exposure to radiation and certain chemicals, radiation therapy, chemotherapy and some genetic disorders such as Down Syndrome.2
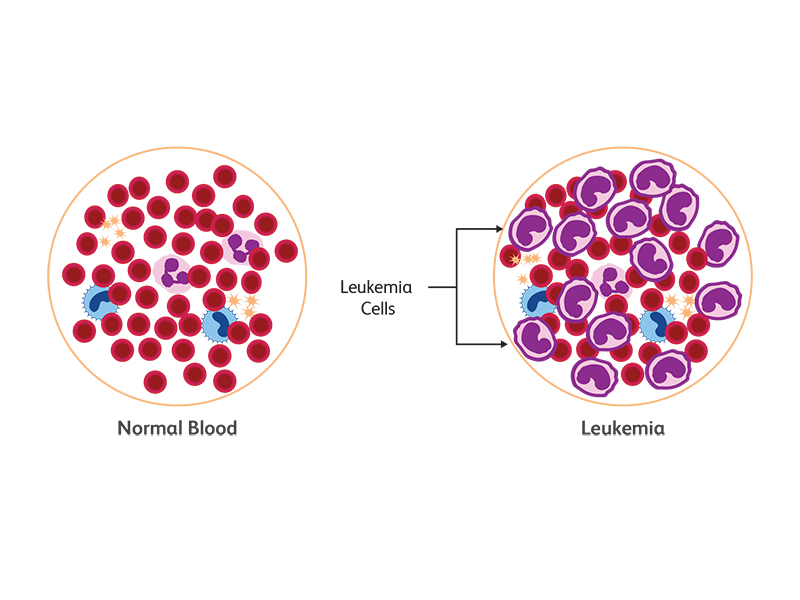
Types of leukemia
Leukemia is categorized as acute or chronic based on the rate of proliferation of the leukemic cells. There are four major types of leukemia, two each for the predominant cell lineages—chronic/acute myeloid leukemia (CML and AML) and chronic/acute lymphocytic leukemia (CLL, ALL).3 Cases of cancer and leukemia are on the rise in the U.S. In 20 years (1998 to 2018) cases of all types of leukemia significantly increased to 187% for CLL, 108% for AML, 96% for CML and 92% for ALL.1,4
How is leukemia diagnosed?
Leukemia is a complex multifactorial disease and leukemia diagnosis requires an interdisciplinary approach, including review of symptoms and physical examination, blood tests, bone marrow biopsy, cerebrospinal fluid (CSF) evaluation, radiology, and genetic testing.
Flow cytometry–based assays for leukemia
Flow cytometry has been reported to be an invaluable tool to aid in both in diagnosis as well as monitoring of the disease.5
Finding the blast population
Flow cytometry offers a commonly used method to discriminate between myeloid and lymphoid blasts.5
- Identifying blasts or immature cells: Immature cells or blasts are typically identified with positive expression of CD34 and low intensity levels of CD45 (CD45low) on an SSC dot plot.
- Aberrant blasts: The expression of abnormal antigens in immature cells is an indication for further investigation for probable leukaemia. These anomalies include arrested maturation; expression of antigen of another lineage (e.g., CD19, CD7 lymphatic markers on myeloid cells)7; asynchronous expression of antigens (e.g., CD15 mature neutrophil marker expression on CD34 blasts) 7; and abnormal intensity of normal antigen expression (e.g., low CD3 expression in T-ALL, low or no CD20 expression in B-ALL)5
Acute leukemia
Under homeostatic conditions, blast counts in the bone marrow or peripheral blood account for less than 5% of cells. Increased blast count is a cause for concern and warrants further investigation. In AML, aberrations can be found in counts and phenotype of blasts.
Diagnosing chronic lymphocytic leukemia
The diagnosis of CLL relies on morphological analysis and immunophenotyping. Based on WHO diagnostic criteria, immunophenotyping of typical CLL demonstrates weak expression of CD20 with co-expression of CD5, CD19 and CD23 on the neoplastic B cells (with consistent counts of 5,000 circulating neoblasts/mL in the peripheral blood during a 3-month period).8 Some markers, such as CD5, CD19, CD23, weak CD20 and immunoglobulin kappa/lambda, are used with consensus in routine diagnostics and are considered required in screening panels. Other markers are recommended as they can provide additional information to better characterize neoplastic B cells or are of prognostic value e.g., weak CD79b, CD81 or CD38, CD45.8
Monitoring residual disease in chronic leukemia
Flow cytometry has been reported to be a sensitive method with which to detect residual disease in chronic leukaemia. Using sequential gating strategies, multiple markers, such as CD5+, CD19+ CLL, can be monitored simultaneously. Marker expression between normal and neoplastic cells can also be discriminated.8
Discrimination between myeloid and lymphoid blasts:
The classification criteria from both the WHO and European Group for the Immunological Characterization of Leukaemias (EGIL) offer specific lineage markers that can be used to identify the types of leukaemia by flow cytometry.8 Myeloid blasts are positive for cytoplasmic myeloperoxidase (cyMPO) and CD117 while lymphoid blasts can be confirmed with cytoplasmic CD79a (cyCD79a) or cytoplasmic CD3 positivity.7
Sub-grouping of leukemia
Flow cytometry panels used for sub-grouping of leukaemia depend on the cell lineages to be investigated.7
- B cell lineage anomalies: To investigate B cell lineage, an antibody panel combining CD5, CD10, CD19, CD20, CD45, and immunoglobulin kappa and lambda is usually used.8,9
- T cell linaege and NK cell anomalies: T cells and NK cells make up 14% of lymphoproliferative disorders. These are overlaps in T cell and NK cell antigen expression. To investigate T/NK cell neoplasms, antibody panels containing CD2, CD3, CD4, CD5, CD7 and CD8 markers can be used.9
- Elevated preipheral blood counts: When peripheral blood counts are elevated, a CD19 flow cytometry screen is recommended.
- Evaluation of myelomonocytic lineages: Examples of myelomonocytic markers include CD13, CD33, CD16, CD10, CD11b, CD14 and CD64 are included in the evaluation of myelomonocytic lineages.10
- Antibody anomalies: In cases where abnormal antibody levels are observed, a plasma cell lineage screening panel including CD19 and CD56 markers is used.11 These anomalies include arrested maturation; expression of antigen of another lineage (e.g., CD19, CD7 lymphatic markers on myeloid cells); asynchronous expression of antigens (e.g., CD15 mature neutrophil marker expression on CD34 blasts); and abnormal intensity of normal antigen expression (e.g., low CD3 expression in T-ALL, low or no CD20 expression in B-ALL). Aberrant CD5 expression is seen in CLL and aberrant CD10 expression can be observed in follicular lymphoma.12
- Flow cytometry also proves immensely valuable in disease monitoring and the evaluation of minimal residual disease.8
Flow cytometry–based assessment of minimal residual disease (MRD)
Minimal residual disease (MRD) is a state where patients still carry small counts of leukaemic cells during and after therapy. These cells can be the source of future recurrence, and monitoring MRD is essential for therapy adjustments, such as to escalate or decrease therapy doses, to determine qualification for transplantation, and to inform the therapy outcome. The prognostic utility of multicolor flow cytometry for assessing MRD in patients with acute lymphoblastic leukaemia and in acute myeloid leukaemia has been established.13 MRD negativity assessment is considered a positive predictor of good long-term prognosis in multiple myeloma as well.14
Flow cytometry facilitates the phenotypic characterization of leukaemic cells and allows the monitoring of MRD. Sets of fluorescently labeled monoclonal antibodies can be used in polychromatic flow cytometry to run panels of markers identifying MRD.5,11 Flow cytometry is used as a tool to monitor MRD in AML patients as PCR monitoring of fusion transcript amplification is only relevant in a fraction of patients. Standardized quantitative flow cytometry protocols can be used during MRD assessment with panels ranging from three to eight colors.13, 14 MRD values greater than 0.01% (1 MRD cell/10,000 bone marrow cells in a bone marrow aspirate) are associated with high risk of relapse and poor survival.15 Flow cytometry can concurrently elucidate the phenotype and functionality of morphologically ambiguous cells.
BD Biosciences offers flow cytometry instruments and reagents for the measurement of immune populations involved in leukaemia.
Our large portfolio of single-color antibody CE-IVD reagents span across a range of specificities and dyes to help in the characterization of hematologic neoplasia. These panels are to be verified and validated by the lab.
Furthermore, BD Biosciences provides the BD OneFlow™ Solution, comprising a comprehensive set of reagents, setup beads, protocols and assay templates, to help standardize leukaemia and lymphoma immunophenotyping. This can improve laboratory efficiency and enable reliability and accuracy of results.16,17
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [published correction appears in CA Cancer J Clin. 2020 Jul;70(4):313]. CA Cancer J Clin. 2018;68(6):394-424. doi:10.3322/caac.21492
- Filippini T, Heck JE, Malagoli C, Del Giovane C, Vinceti M. A review and meta-analysis of outdoor air pollution and risk of childhood leukaemia. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2015;33(1):36-66. doi:10.1080/10590501.2015.100299
- Thapa B, Fazal S, Parsi M, Rogers HJ. Myeloproliferative Neoplasms. In: StatPearls. Treasure Island (FL): StatPearls Publishing; August 13, 2020.
- Hao T, Li-Talley M, Buck A, Chen W. An emerging trend of rapid increase of leukaemia but not all cancers in the aging population in the United States. Sci Rep. 2019;9(1):12070. Published 2019 Aug 19. doi:10.1038/s41598-019-48445-1
- Wang XM. Advances and issues in flow cytometric detection of immunophenotypic changes and genomic rearrangements in acute pediatric leukaemia. Transl Pediatr. 2014;3(2):149-155. doi:10.3978/j.issn.2224-4336.2014.03.06
- Chauhan AF, Viruni N, Alvin Liu TY, Arevalo JF, Cheson BD. Successful treatment of ocular chronic lymphocytic leukaemia with Ibrutinib: case report and review of the literature. Leuk Res Rep. 2020;14:100200. doi:10.1016/j.lrr.2020.100200
- Matarraz S, Almeida J, Flores-Montero J, et al. Introduction to the diagnosis and classification of monocytic-lineage leukaemias by flow cytometry. Cytometry B Clin Cytom. 2017;92(3):218-227. doi:10.1002/cyto.b.21219
- Del Giudice I, Raponi S, Della Starza I, et al. Minimal residual disease in chronic lymphocytic leukaemia: a new goal? Front Oncol. 2019;9:689. doi:10.3389/fonc.2019.00689
- Seegmiller AC, Hsi ED, Craig FE. The current role of clinical flow cytometry in the evaluation of mature B-cell neoplasms. Cytometry B Clin Cytom. 2019;96(1):20-29. doi:10.1002/cyto.b.21756
- Bento LC, Correia RP, Pitangueiras Mangueira CL, et al. The use of flow cytometry in myelodysplastic syndromes: A review. Front Oncol. 2017;7:270. doi:10.3389/fonc.2017.00270
- Kumar S, Kimlinger T, Morice W. Immunophenotyping in multiple myeloma and related plasma cell disorders. Best Pract Res Clin Haematol. 2010;23(3):433-451. doi:10.1016/j.beha.2010.09.002
- Jaseb K, Purrahman D, Shahrabi S, Ghanavat M, Rezaeean H, Saki N. Prognostic significance of aberrant CD5 expression in B-cell leukaemia. Oncol Rev. 2019;13(1):400. doi:10.4081/oncol.2019.400
- Kern W, Bacher U, Haferlach C, Schnittger S, Haferlach T. The role of multiparameter flow cytometry for disease monitoring in AML. Best Pract Res Clin Haematol. 2010;23(3):379-390. doi: 10.1016/j.beha.2010.06.007
- Perrot A, Lauwers-Cances V, Corre J, et al. Minimal residual disease negativity using deep sequencing is a major prognostic factor in multiple myeloma. Blood. 2018;132(23):2456-2464. doi:10.1182/blood-2018-06-858613
- Kruse A, Abdel-Azim N, Kim HN, et al. Minimal residual disease detection in acute lymphoblastic leukaemia. Int J Mol Sci. 2020;21(3):1054. doi:10.3390/ijms21031054
- van der Velden VHJ, Flores-Montero J, Perez-Andres M, et al. Optimization and testing of dried antibody tube: The EuroFlow LST and PIDOT tubes as examples. J Immunol Methods. 2019;475:112287. doi: 10.1016/j.jim.2017.03.011
- Moloney E, Watson H, Barge D, et al. Efficiency and health economic evaluations of BD OneFlow™ Flow Cytometry Reagents for diagnosing chronic lymphoid leukaemia. Cytometry B Clin Cytom. 2019;96(6):514-520. doi: 10.1002/cyto.b.21779
BD Biosciences clinical flow cytometry solutions, including instrumentation, software and reagents, offer the building blocks for laboratory-developed tests used in the identification of markers associated with leukaemias / lymphomas.
These solutions are ordered as Analyte Specific Reagents (ASR) and their analytical and performance characteristics are not established.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.