-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Multicolor Flow Cytometry
For Professionals in Research
Multicolor flow cytometry provides rapid, multiparametric analysis of various cellular subpopulations of interest. Its applications include immunology, immuno-oncology, virology, immune monitoring and many others. BD Biosciences has developed innovative dyes across every laser line for providing panel design flexibility and a comprehensive portfolio of conjugated antibodies to help deep characterization of surface, intracellular or secreted markers. From flow cytometers and sorters for simple to complex research applications to an extensive selection of reagents and a comprehensive collection of educational resources, tools such as the Interactive Human Cell Map, Clone Comparison, BD® Spectrum Viewer, and protocols, we support you in efficiently navigating your multicolor flow cytometry workflow journey.
Principles of multicolor flow cytometry
Flow cytometry analyzes a population of cells on a cell-by-cell basis, a critical capability for today’s researchers and clinicians who are looking for the very few cells among the many cells in a sample (like finding needles in a haystack) that will enable them to study a disease state or biological process. Second, flow cytometry is extraordinarily rapid. Routine sample analysis rates can range up to 10,000 cells per second—an incredible advancement over historical methods of visually examining and counting cells. Finally, flow cytometry has the capacity to simultaneously measure multiple parameters (multiplexing) of single cells. Multiplexing allows researchers and clinicians to gather more information from a single sample faster than ever before. These capabilities have made flow cytometry a powerful tool with multiple applications for researchers and clinicians alike.
How flow cytometry works
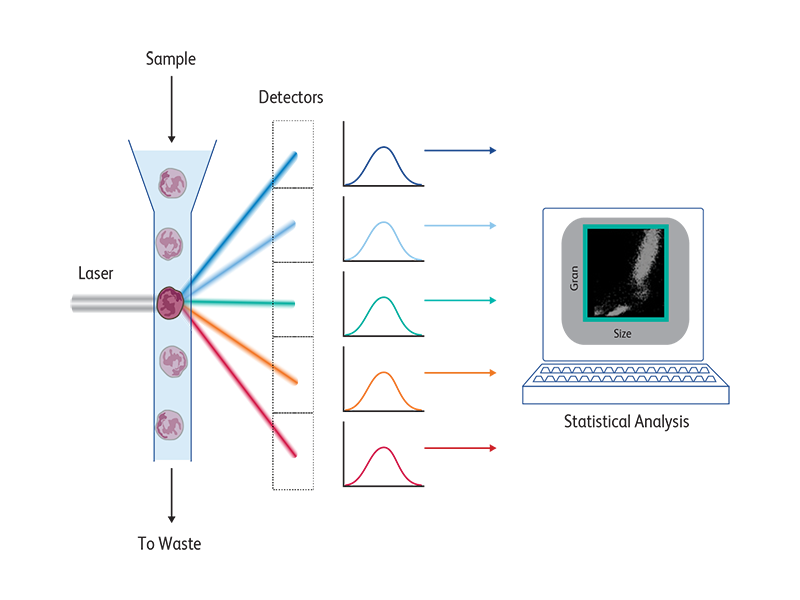
Flow cytometers contain three main systems—fluidics, optics and electronics.
The fluidics system funnels a sample of cells (e.g., a sample of human blood) into a single stream so that the cells pass through a laser beam one at a time. As each cell passes through the beam, it scatters light and may emit fluorescent light. These light signals are collected by the optics system and routed to various detectors. The signals received by the detectors are then converted into numerical values by the electronics system. Results can be displayed on the screen or saved for future analysis using specially designed software.
As each cell moves through the beam, its parameters (characteristics) are measured and recorded, along with the time that it passed through the beam. Typically, data are collected for at least 10,000 cells per sample.
Sorting
First the parameters are set so that the flow cytometer can identify cells of interest. The stream of fluid containing the sample is funneled at high pressure by the fluidics system into a single stream so that cells pass one at a time through the laser beam where information about the cells is detected. If a cell matches the parameters specified, the cytometer applies an electrical charge to it. This causes the charged cell to be deflected toward a collection tube. The researcher can then take the cells of interest and grow them in culture or study them in other assays. The droplets that are not charged continue in the stream to a waste container. The process is rapid and can be performed at speeds of up to 20,000 cells per second.
Find the most appropriate cell sorter for your experiment. BD Biosciences offers a range of research cell sorters for various research applications, from simple, benchtop systems to advanced, fully configurable systems.
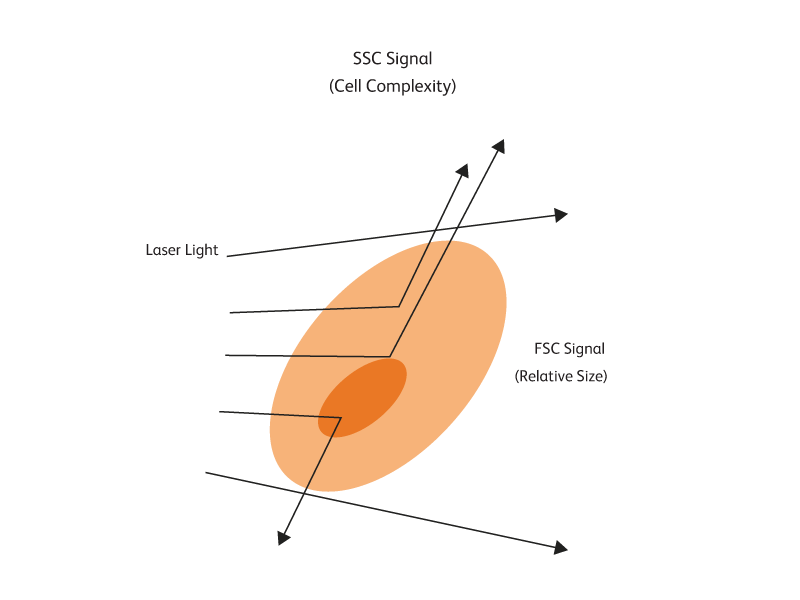
Measuring cell characteristics
As a cell passes through a laser beam, the light is scattered in different directions. Measuring how much and in which direction light is scattered helps researchers determine both the size and internal complexity of a cell. Using flow cytometry, more than one population of cells can be studied at a time and information beyond size and complexity can be obtained.
Fluorescence as a marker of cells and cellular activities
Monoclonal antibodies are often linked to a fluorescent dye (fluorochrome) that will emit fluorescent light when excited by a laser. The fluorochrome allows cells that have bound to the antibody or marker to be tracked. Most cells do not naturally emit fluorescent light. However, if a fluorochrome-tagged monoclonal antibody is bound to an antigen present on a cell, a fluorescent light signal will be picked up when that cell passes through the laser beam of the flow cytometer.
Fluorochromes differ from each other with respect to the color of light that they will emit. For example, FITC, one of the fluorochromes BD offers, will emit green light, whereas PE, another one of the fluorochromes BD offers, will emit orange light when it is exposed to the laser used in our flow cytometers. The use of different fluorochromes helps researchers distinguish between cell populations or different cellular activities.
Below is the list of fluorochromes currently available for custom conjugation. Our team of experienced chemists can address any additional configuration or special requirements to deliver the product you need.
| Excitation Laser Line | Fluorochrome | Ex-max (nm) | Em-max (nm) | Relative Brightness |
|---|---|---|---|---|
| UV 355 nm |
BD Horizon™ BUV395 | 348 | 395 | |
| BD Horizon™ BUV496 | 350 | 496 | ||
| BD Horizon™ BUV563 | 350 | 564 | ||
| BD Horizon™ BUV615 | 350 | 615 | ||
| BD Horizon™ BUV661 | 350 | 660 | ||
| BD Horizon™ BUV737 | 350 | 735 | ||
| BD Horizon™ BUV805 | 351 | 803 | ||
| Violet 405 nm |
BD Horizon™ BV421 | 407 | 423 | |
| BD Horizon™ V450 | 405 | 450 | ||
| Pacific Blue™ | 404 | 455 | ||
| BD Horizon™ BV480 | 440 | 479 | ||
| BD Horizon™ V500 | 415 | 499 | ||
| BD Horizon™ BV510 | 405 | 512 | ||
| BD Horizon™ BV570** | 407 | 573 | ||
| BD Horizon™ BV605 | 407 | 605 | ||
| BD Horizon™ BV650 | 406 | 649 | ||
| BD Horizon™ BV711 | 407 | 713 | ||
| BD Horizon™ BV75O | 409 | 754 | ||
| BD Horizon™ BV786 | 407 | 786 | ||
| BD Horizon™ RV828 | 412 | 828 | ||
| Blue 488 nm |
BD Horizon™ BB515 | 490 | 515 | |
| Alexa Fluor™ 488 | 494 | 517 | ||
| FITC | 494 | 518 | ||
| BD Horizon™ RB545 | 496 | 545 | ||
| BD Horizon™ RB613 | 492 | 613 | ||
| BD Horizon™ RB670 | 492 | 670 | ||
| PerCP | 481 | 675 | ||
| PerCP-Cy5.5 | 482 | 676 | ||
| BD Horizon™ BB700 | 476 | 695 | ||
| BD Horizon™ RB705 | 498 | 707 | ||
| BD Horizon™ RB744 | 498 | 746 | ||
| BD Horizon™ RB780 | 498 | 781 | ||
| BD Horizon™ RB824 | 492 | 824 | ||
| Yellow-Green 561 nm |
PE* | 566 | 576 | |
| BD Horizon™ RY586 | 564 | 586 | ||
| BD Horizon™ RY610 | 557 | 610 | ||
| BD Horizon™ PE-CF594* | 566 | 615 | ||
| BD Horizon™ RY655 | 557 | 654 | ||
| PE-Cy5* | 566 | 670 | ||
| BD Horizon™ RY703 | 557 | 703 | ||
| BD Horizon™ RY743 | 557 | 743 | ||
| BD Horizon™ RY775 | 557 | 775 | ||
| PE-Cy7* | 566 | 781 | ||
| Red 640 nm |
APC | 651 | 660 | |
| Alexa Fluor™ 647 | 653 | 669 | ||
| BD Horizon™ RR688 | 669 | 688 | ||
| BD Horizon™ APC-R700 | 651 | 706 | ||
| BD Horizon™ Red 718 | 695 | 718 | ||
| Alexa Fluor™ 700 | 697 | 719 | ||
| APC-Cy7 | 651 | 779 | ||
| BD® APC H7 | 659 | 782 |
BUV: BD Horizon Brilliant™ UV; BV: BD Horizon Brilliant™ Violet; V: BD Horizon™ Violet; RV: BD Horizon RealViolet™; BB: BD Horizon Brilliant™ Blue; RB: BD Horizon RealBlue™; RY: BD Horizon RealYellow™; RR: BD Horizon RealRed™.
*Excited by 488-nm, 532-nm and 561-nm lasers.
**Available as custom.
For additional fluorochrome selection support, launch the BD® Spectrum Viewer Tool.
Multicolor flow cytometry
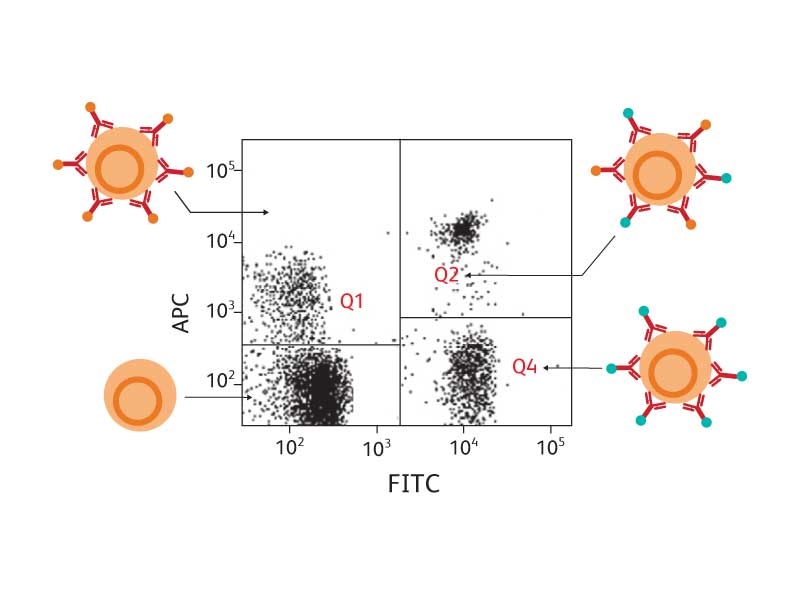
Cells of different types bear different combinations of antigens and will bind with different combinations of antibodies. If each monoclonal antibody used in testing a sample of cells is linked to a different fluorochrome, the cell types can be distinguished from each other by the combinations of colors that they emit when they pass through the laser beam.
The use of combinations of multiple fluorochromes simultaneously further enhances the ability to distinguish between different populations of cells. The figure below shows an example in which two different monoclonal antibodies—each linked to a different fluorochrome—were used in testing a cell sample to look for T lymphocytes. Four different types of cells were evident.
Flow cytometry combines the flexibility and sensitivity of fluorescence technology with high speed and data integration capabilities. It has become the gold standard in cell analysis and now is being used as an analytic tool across numerous sectors of life sciences.
Intracellular flow cytometry
Intracellular flow cytometry is a powerful technique for the identification of cell types and the analysis of signaling and functional responses within cell lines and heterogeneous cell samples. BD Biosciences provides a variety of reagents, kits and protocols to facilitate intracellular flow cytometry. Find the tools and techniques, including BD fluorochrome-conjugated antibodies, buffers, kits and protocols that support intracellular cytokine staining, phosphoprotein and transcription factor detection by intracellular flow cytometry.
Spectral flow cytometry
Spectral flow cytometry has transformed the field of cytometry to meet the analytical needs for multiparameter immunophenotyping of immune cells in basic and translational research. By measuring the entire fluorochrome emission spectrum, spectral flow cytometry can resolve individual spectral signatures of highly overlapping fluorochromes and increases the number of fluorochromes that can be used simultaneously for larger and more complex panels.
BD Biosciences tools for multicolor flow cytometry
Flow cytometry Instruments
As pioneers in flow cytometry, we have been at the forefront of flow cytometry instrumentation development. We offer a comprehensive line of research cell analyzers and cell sorters to support you through your research continuum.
Lasers and dyes
Lasers provide information on the size and complexity of cells and excite fluorochromes conjugated to antigen-specific antibodies that enable the identification and understanding of the function of cells. To fully leverage the power of multicolor flow cytometry, it is critical to understand which fluorochrome to use, compare the spectral properties of different fluorochromes and determine the right instrument based on the number of parameters needed to be analyzed. We offer a wide variety of fluorochromes across every laser line for multicolor flow cytometry. To help you choose the right fluorochromes for your experiments, we have developed a fluorochrome atlas that allows you to view and compare dyes with attributes such as excitation/emission spectra, filter recommendations and relative resolution information.
BD OptiBuild™ On-Demand Reagents
BD OptiBuild™ On-Demand Reagents make it easy for you to gain access to the antibodies you need on an ever-growing catalog of dye combinations. BD OptiBuild™ Reagents provide flexibility to evaluate and add new fluorochromes and simplify your panel design.
Protocols and resources
Protocols
To help you find the right protocols for your experiments, we have developed a protocol repository. Here you will find step-by-step guides, helpful recommendations and other resources to support your flow cytometry workflow.
Panel design resources and tools
Find helpful tools and resources such as the Interactive Cell Map, Clone Comparison, and BD® Spectrum Viewer, to support multicolor panel design, workflow and experimental analysis.
BD Horizon Brilliant™ Dyes
BD Horizon Brilliant™ Dyes were developed from pioneering polymer dye technology acquired from Sirigen Ltd. These unique dyes can be brighter than conventional dyes, with equivalent background—a breakthrough in the field.
For cells that have few receptors on the surface, bright reagents are essential in resolving these dim cells from others in a sample. The characteristics of the BD Horizon Brilliant™ Polymer Dyes enable them to achieve, in some cases, much brighter fluorescence signals than traditional organic fluorescent dyes or even phycobiliproteins such as PE or APC. This means that they are effective for identifying cell populations with a broader range of receptor density than previously possible.
Signal Amplification
BD Horizon Brilliant™ Dye polymer chains can be considered as a collection of optical segments. Each segment in the polymer chain is capable of absorbing light, resulting in materials that can have extremely large extinction coefficients (or probability of absorbing photons of light).
As a result of the electronic delocalization inherent to these materials, energy has the ability to move or migrate along these extended chains. Thus, all the light harvested by the polymers can either be channeled to a closely associated fluorescent acceptor of lower energy through a fluorescence resonance energy transfer (FRET) process, or emitted as fluorescence by the polymer itself if no dyes are used. It is this collective molecular antenna–like behavior that forms the foundation of the technology and intellectual property base.
Conjugated to a reporter dye, these polymers can also serve as donors to create tandem dyes with exceptional Stokes shifts.
Base Polymer Dyes and Polymer Tandem Dyes
BD Horizon Brilliant™ Dyes come in two basic formats—base polymer dyes and polymer tandem dyes.
The base polymer dyes are the simplest embodiment of the technology, are very photostable and are bright compared to conventional fluorescent dyes. They are very effective at collecting excitation light (like molecular antennae) and efficiently converting it to emitted light at a longer wavelength.
Polymer tandem dyes are composed of two dye molecules—the base polymer (donor) dye and a second conventional fluorescent (acceptor) dye. The molecules are chemically linked together to form a tandem dye that is excited at the excitation wavelength of the donor and emits at the emission wavelength of the acceptor. These tandem dyes are very bright compared to conventional tandem dyes.
One of the future goals for this technology is to design base polymer dyes and polymer tandem dyes that can be easily multiplexed to make multicolor flow cytometry easier.
View sample data from our newest BD Horizon Brilliant™ Dyes.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Alexa Fluor and Pacific Blue are trademarks of Life Technologies Corporation.
Cy is a trademark of Global Life Sciences Solutions Germany GmbH or an affiliate doing business as Cytiva.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.