-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Natural Killer (NK) Cells
For Professionals in Research
NK cells are generally categorized as innate immune cells despite the fact that they bear features of lymphocytes such as morphological similarity and expression of lymphoid markers. NK cells mount a frontline defense against invading pathogens and tumors but, unlike T lymphocytes, do not require specific antigen recognition or costimulation. They are the counterparts of T cells and are a part of the innate lymphoid cells (ILCs), which are grouped into three groups (Group 1–3) based on their functions. NK cells are part of Group I ILCs and exert cytotoxic functions similar to CD8+ T cells.1
Types of NK cells
NK cells are derived from the bone marrow but can mature both in the bone marrow and in secondary lymphoid tissues. In humans, different types of NK cells (NK cytotoxic, NK regulatory and NK memory) have been described based on the functions they carry out—cytotoxicity, regulatory profile and tolerance, respectively.2 Each type of NK cell can be discriminated by sets of surface/intracellular NK cell markers along with secretory molecules. Expression of these markers is conditioned by the environmental context and stimuli that recruit and activate NK cells. While surface markers alone cannot segregate the type of NK cells, metabolic markers can give a direct clue about what NK cells are involved in. NK cytotoxic cells are characterized by glycolysis and oxidative phosphorylation; NK regulatory cells utilize sources other than glucose for energy and have low levels of glycolysis and oxidative phosphorylation; and NK memory cells have low levels of reactive oxygen species (ROS), high glucose and lipid metabolism.3
The functions of NK cells
Viral control
NK cells expand, undergo specific differentiation and acquire different sets of receptors and signaling molecules when they encounter certain types of viruses such as HIV (NKG2C, NK p10, NK p46), HCV (NKG2D, perforin, granzyme B, 2B4), CMV (NKG2C, CD57, KIRs), and EBV (NKG2A, NKG2D, CD16, 2B4). NK cells also produce high amounts of IFN-gamma and have memory-like properties if they re-encounter a known viral pathogen.4
Tumor control
In conjunction with cytotoxic T lymphocytes, NK cells are at the forefront of anti-tumor immune responses.5 NK cells secrete cytolytic granules containing the pore-forming protein perforin to opsonize target cells via induction of nonselective transport across the plasma membranes.6 NK secreted granules also contain granzymes A/B, which trigger apoptotic cascades in target cells. NK cells can also induce tumor cell death through death receptor pathways and cytokine secretion.7 As tumor cells acquire escape mechanisms to blunt and re-orient immune responses, NK cells may lose their efficacy against them. In immuno-oncology research, the potential of NK cells is being exploited as they can be expanded and manipulated (CAR-NK cells) for cell therapy transplant.8, 9
Homeostasis
Besides their cytotoxic role in viral and tumor control, NK cells are capable of homeostatic functions, balancing clearance and tolerance. Killer immunoglobulin-like receptors (KIR) allow NK cells to recognize the self through the binding of HLA molecules.10 During pregnancy, NK cells partner with M2 polarized macrophages. NK cells also promote tolerance during pregnancy and participate in trophoblast invasion and spiral artery development, benefiting both the mother and child.11
Learn more about multicolor flow cytometry panels for the assessment of activating and inhibitory receptor expression in NK cells.
Listen to the NK cell webinar
References
- Vivier E, Artis D, Colonna M, et al. Innate lymphoid cells: 10 years on. Cell. 2018;174(5):1054-1066. doi:10.1016/j.cell.2018.07.017
- Abel AM, Yang C, Thakar MS, Malarkannan S. Natural killer cells: development, maturation, and clinical utilization. Front Immunol. 2018;9:1869. doi:10.3389/fimmu.2018.01869
- Poznanski SM, Ashkar AA. What defines NK cell functional fate: phenotype or metabolism? Front Immunol. 2019;10:1414. doi:10.3389/fimmu.2019.01414
- Wilk AJ, Blish CA. Diversification of human NK cells: Lessons from deep profiling. J Leukoc Biol. 2018;103(4):629-641. doi:10.1002/JLB.6RI0917-390R
- Fu B, Tian Z, Wei H. Subsets of human natural killer cells and their regulatory effects. Immunology. 2014;141(4):483-489. doi:10.1111/imm.12224
- Hodgins JJ, Khan ST, Park MM, Auer RC, Ardolino M. Killers 2.0: NK cell therapies at the forefront of cancer control. J Clin Invest. 2019;129(9):3499-3510. doi:10.1172/JCI129338
- Osińska I, Popko K, Demkow U. Perforin: an important player in immune response. Cent Eur J Immunol. 2014;39(1):109-115. doi:10.5114/ceji.2014.42135
- Marcus A, Gowen BG, Thompson TW, et al. Recognition of tumors by the innate immune system and natural killer cells. Adv Immunol. 2014;122:91-128. doi:10.1016/B978-0-12-800267-4.00003-1
- Gonzalez-Rodriguez AP, Villa-Álvarez M, Sordo-Bahamonde C, Lorenzo-Herrero S, Gonzalez S. NK cells in the treatment of hematological malignancies. J Clin Med. 2019;8(10):1557. doi:10.3390/jcm8101557
- Boudreau JE, Hsu KC. Natural killer cell education in human health and disease. Curr Opin Immunol. 2018;50:102-111. doi:10.1016/j.coi.2017.11.003
- Faas MM, de Vos P. Uterine NK cells and macrophages in pregnancy. Placenta. 2017;56:44-52. doi:10.1016/j.placenta.2017.03.001
BD Biosciences provides several tools for characterizing NK cells and their functions
NK cell enrichment
BD Biosciences tools allow both positive and negative selection of your target cells using magnetic separation methods. The BD IMag™ Human NK Cell Enrichment Set - DM is used for the negative selection of NK cells from peripheral blood. The Biotinylated Human NK Cell Enrichment Cocktail contains monoclonal antibodies that recognize antigens expressed on erythrocytes, platelets and peripheral leukocytes (including NK-T cells) that are not NK cells. The BD IMag™ Anti-Human CD56 Magnetic Particles have been reported to effectively separate CD56-bearing cells of human and rhesus macaque blood. These particles are optimized for the positive selection or depletion of the CD56-bearing leukocytes using the BD IMag™ Cell Separation Magnet. Enrichment of NK cells without NK-T cells can also be carried out using the BD IMag™ Anti-Human CD3 Magnetic Particles - DM to specifically deplete T lymphocytes before separation of CD56-positive leukocytes. Based on the same principles, enrichment of mouse NK cells can be conducted, for example, using the BD IMag™ Mouse NK Cell Enrichment Set - DM for the negative selection of NK cells from the mouse spleen or lymph node. This negative selection ensures no inadvertent activation of NK cells during isolation.
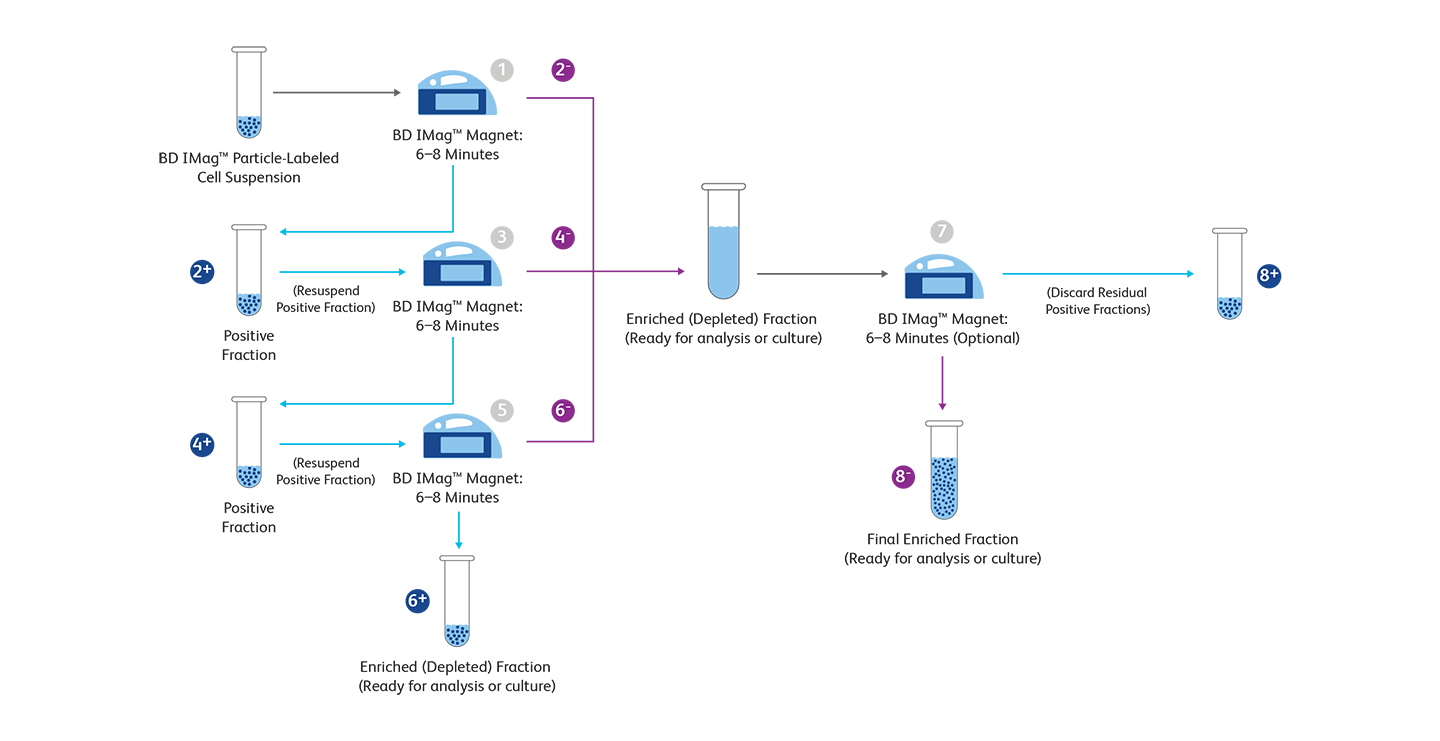
Magnetic separation workflow
+ and – refers to the steps in the positive and negative selection process, respectively. For more details, refer to our magnetic cell separation page.
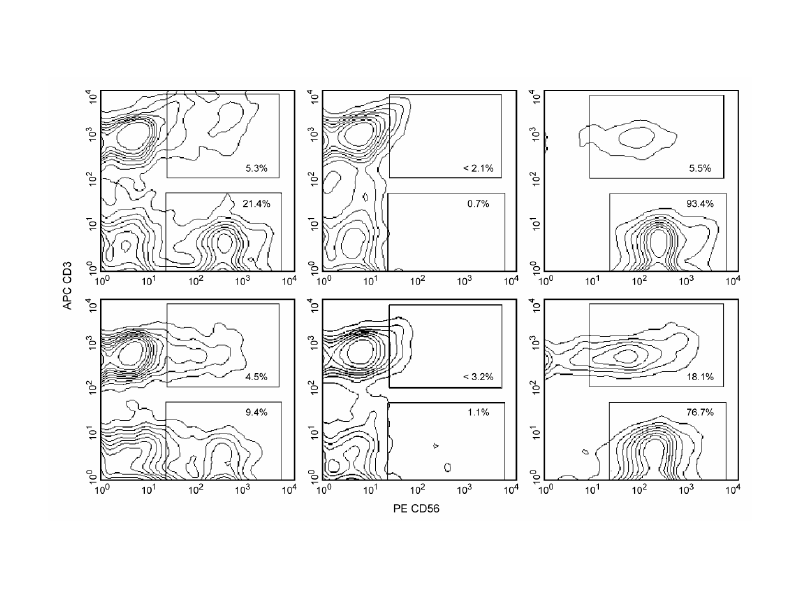
Positive selection of human CD56+ NK cells from PBMC derived from two different donors.
Leukocytes were labeled with BD IMag™ Anti-Human CD56 Magnetic Particles - DM (Cat. No. 557775), separated using the BD IMag™ Cell Separation Magnet (Cat. No. 552311), and the negative (CD56-) and positive (CD56+) fractions were collected. Fresh PBMC (left panels), the negative fractions (center panels) and the positive fractions (right panels) were stained with PE Mouse Anti-Human CD56 (Cat. No. 555516) and APC Mouse Anti-Human CD3 (Cat. No. 555335). Percentages of CD56+CD3- NK cells and CD56+CD3+ NK-T cells in each sample are displayed. Flow cytometry was performed on a BD FACSCalibur™ Flow Cytometry System. The dimmer staining observed in the right panels, compared to the respective left panels, indicates that BD IMag Particle-labeling partially blocks CD56 staining by the PE Mouse Anti-Human CD56 antibody. It has been reported that the relative percentage of CD56+CD3+ NK-T cells, among the total CD56+ cells, can vary among donors. Data from donors with relatively low (top panels) and high (bottom panels) levels of NK-T cells are depicted.
NK cell cytotoxicity
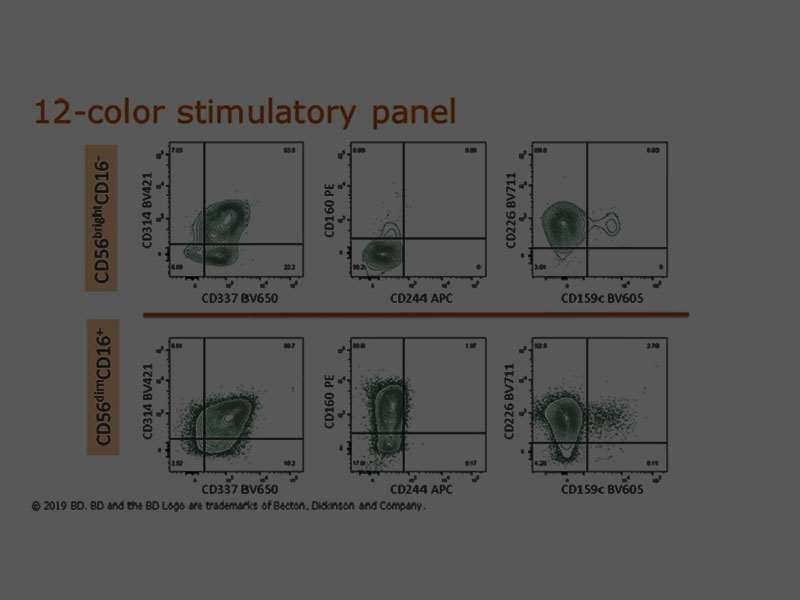
The share of NK cells in cytotoxicity responses can be monitored by a multicolor flow cytometry panel with a selection of antibodies including the pan NK marker CD56.
For analyzing natural cytotoxicity receptors (NCRs), CD335 (NKp46), CD336 (NKp44) and CD337 (NKp30) can be used. BD Pharmingen™ and BD Horizon™ Antibodies are available in an array of fluorescence formats to accommodate your detection panel needs.
Surface marker detection can also be combined with antibodies to cytotoxic cytokines such as interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α) and chemokines such as macrophage inflammatory protein-1β (MIP-1β).
Ready-to-use cocktails such as the BD Pharmingen™ NHP T/B/NK Cocktail with surface markers for T cells (CD3), B cells (CD20) and NK/monocyte/macrophages (CD16) can be combined with other NK markers to specifically profile their functionality.
In addition, BD Biosciences provides high-sensitivity ELISPOT assays to evaluate the cytotoxicity profile of enriched NK cells by interferon gamma secretion.
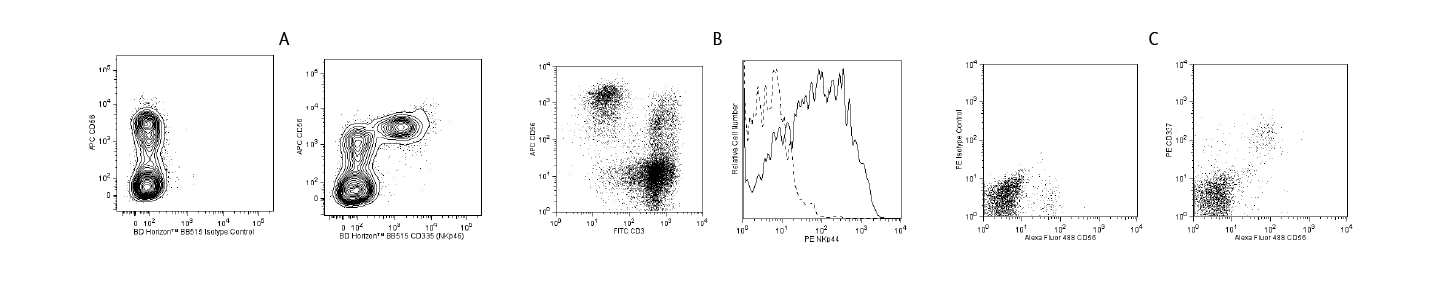
NK cell degranulation assay
Activated NK cells rich in cytoplasmic cytolytic granules containing granzymes and perforin can also be assessed by flow cytometry.1, 2 The type of cytolytic granules can be discriminated by flow cytometry by using BD Biosciences reagent set portfolio such as BD Pharmingen™ FITC Mouse Anti-Human Granzyme A Reagent Set and BD Pharmingen™ PE Perforin Reagent Set.
NK degranulation monitoring can be carried out by detecting surface expression of CD107a (LAMP-1) on NK cells. The content of granules in activated NK cell populations before their release can be monitored by blocking the secretory pathway with BD GolgiPlug™ Protein Transport Inhibitor containing brefeldin A or BD GolgiStop™ Inhibitor containing monensin followed by fixation and permeabilization with BD Cytofix/Cytoperm™ Solution. BD Cytofix/Cytoperm™ Plus Kits are also available with BD GolgiPlug™ or BD GolgiStop™ Inhibitors. BD antibodies with fluorescently labeled interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α) and chemokines such as macrophage inflammatory protein-1β (MIP-1β) are also available.
Find out more details on a multicolor flow cytometer panel designed to assess NK cell cytotoxicity.
References
- Tognarelli S, Jacobs B, Staiger N, Ullrich E. Flow Cytometry-based assay for the monitoring of NK cell functions. J Vis Exp. 2016;(116):54615. doi:10.3791/54615
- Shabrish S, Gupta M, Madkaikar M. A modified NK cell degranulation assay applicable for routine evaluation of NK cell function. J Immunol Res. 2016;2016:3769590. doi:10.1155/2016/3769590
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.