-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Clinical Brilliance Expanded
Improving and Expanding BD Horizon Brilliant™ ASRs to Enhance Performance
BD has expanded the portfolio of our BD Horizon Brilliant Violet™ Analyte Specific Reagents (ASRs). Since 2018 we have implemented a new manufacturing process for all new BD Horizon Brilliant™ ASRs that improves performance to enable confidence in results and added flexibility in multicolor design. Additional BD Horizon Brilliant™ ASRs using this manufacturing process are now available. BD Horizon™ BV605 and BV421 ASRs can be used on flow cytometers equipped with a violet laser and appropriate filters, such as high color configurations of the BD FACSLyric™ and BD FACSCanto™ Flow Cytometers.*
Clinical Brilliance Expanded
Lot-to-Lot Consistency
In 2018, BD improved lot-to-lot consistency and reduced the allowable manufacturing lot-to-lot variation in brightness by 50% for all new BD Horizon Brilliant™ ASRs. With robust reagent consistency, you are given confidence in the reproducibility of your results.
Illustrative examples from product development are shown in Figures 1, 2 and 3.
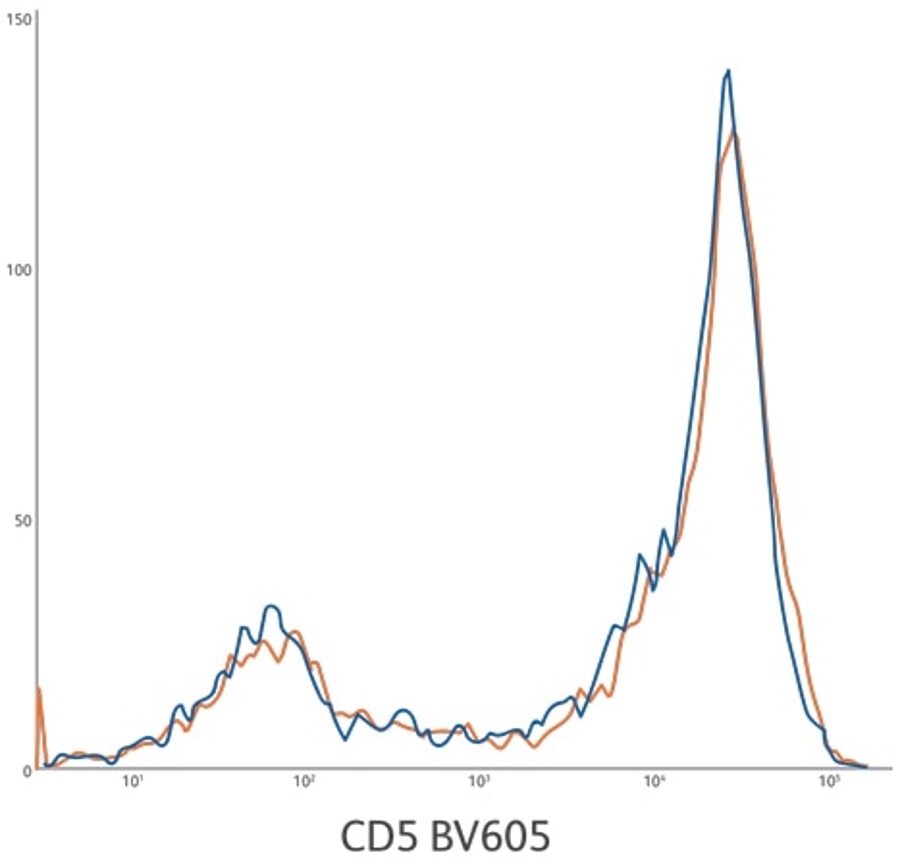
Lot-to-lot consistency for CD5 BV605 ASR development lots 1 and 2. Lysed whole blood gated on lymphocytes.
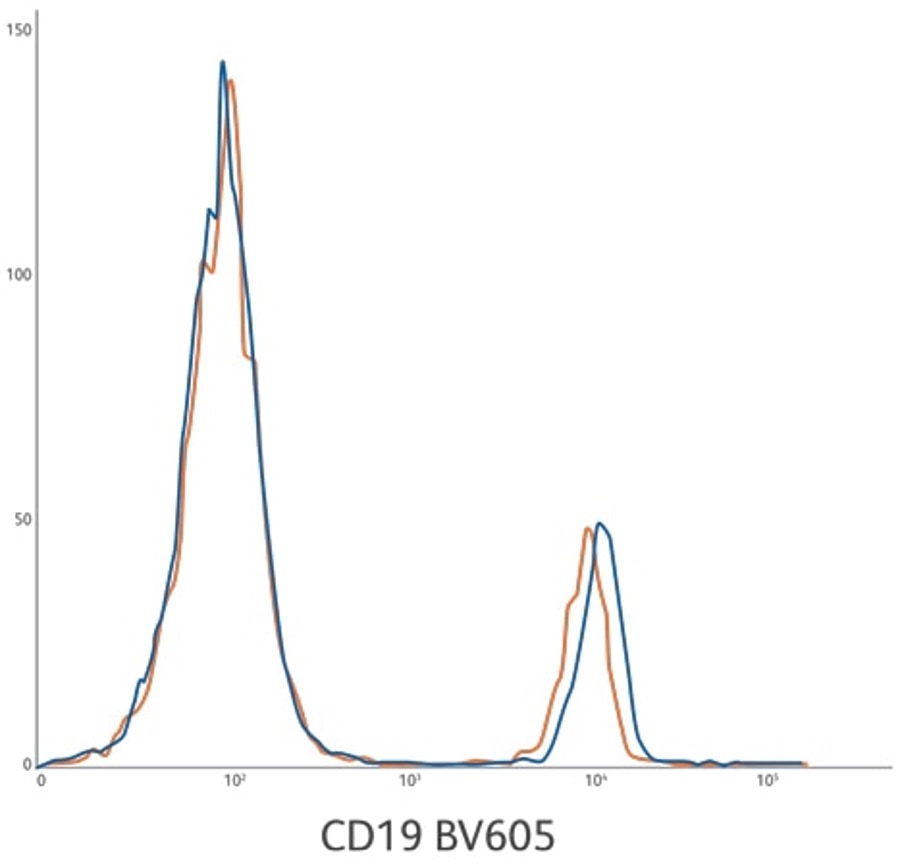
Lot-to-lot consistency for CD19 BV605 ASR development lots 1 and 2. Lysed whole blood gated on lymphocytes.
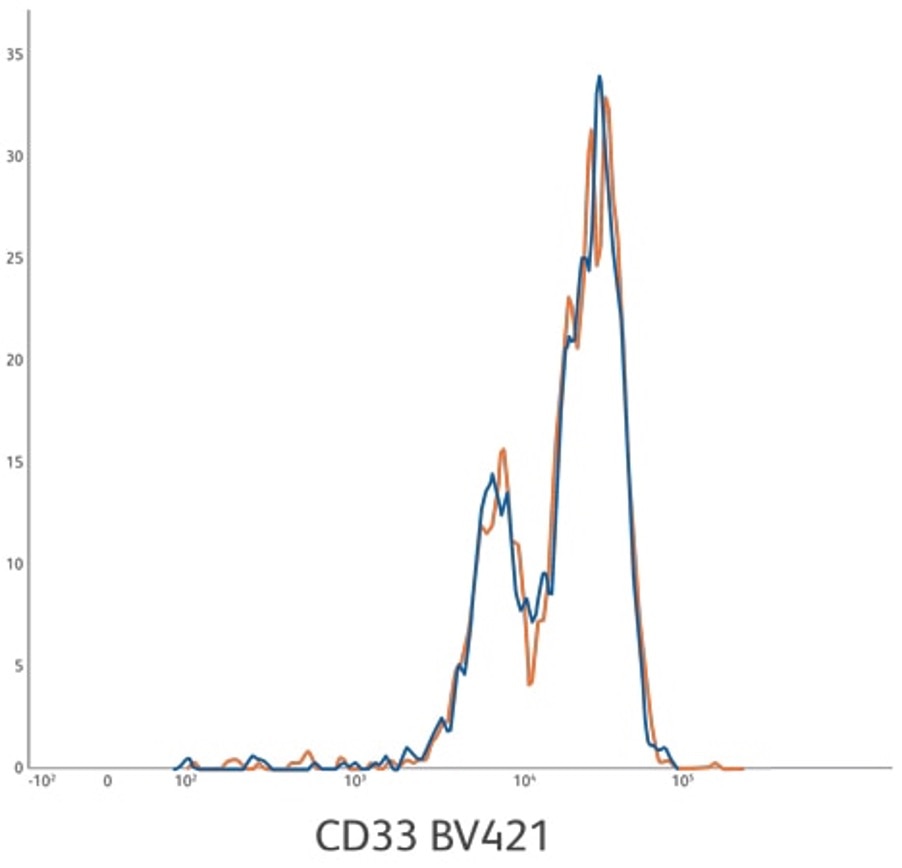
Lot-to-lot consistency for CD33 BV421 ASR development lots 1 and 2. Lysed whole blood gated on monocytes.
Brightness
ASRs were developed to be bright - typically as bright or brighter than our analogous research use only (RUO) reagents.
Illustrative examples from product development are shown in Figures 4 and 5.
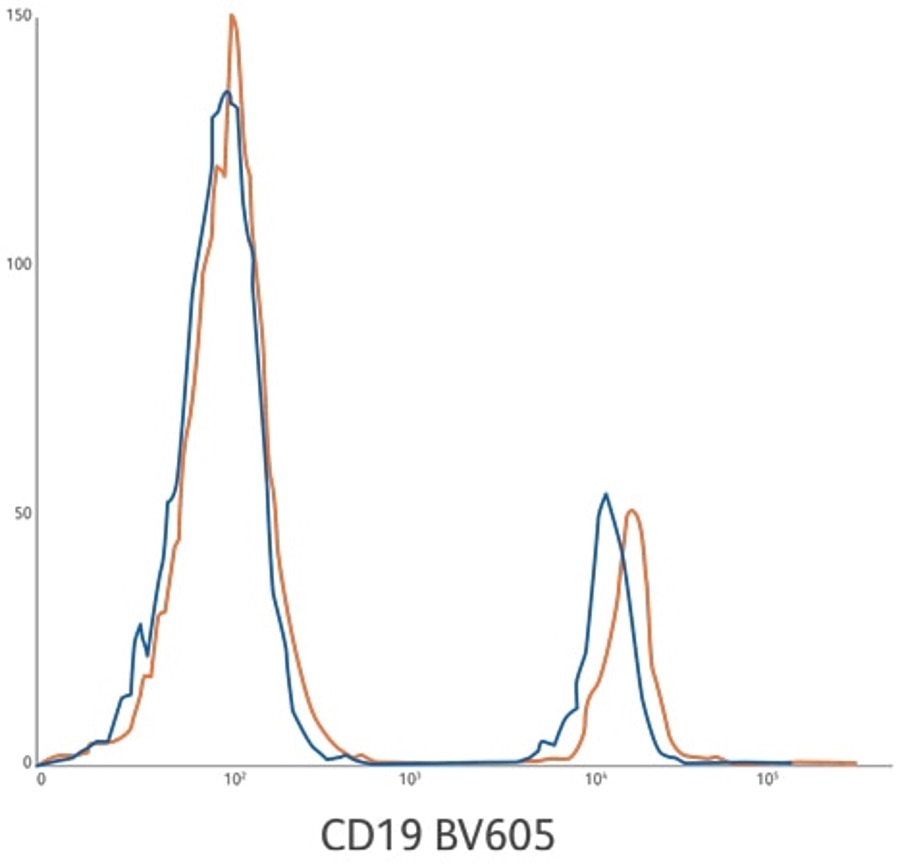
CD19 BV605 ASR (orange) compared to analogous CD19 BV605 RUO reagent (blue). The CD19 BV605 ASR development lots are on average 18% brighter based upon positive population MFI. Average stain index of 152 compared to 133. Lysed whole blood gated on lymphocytes.
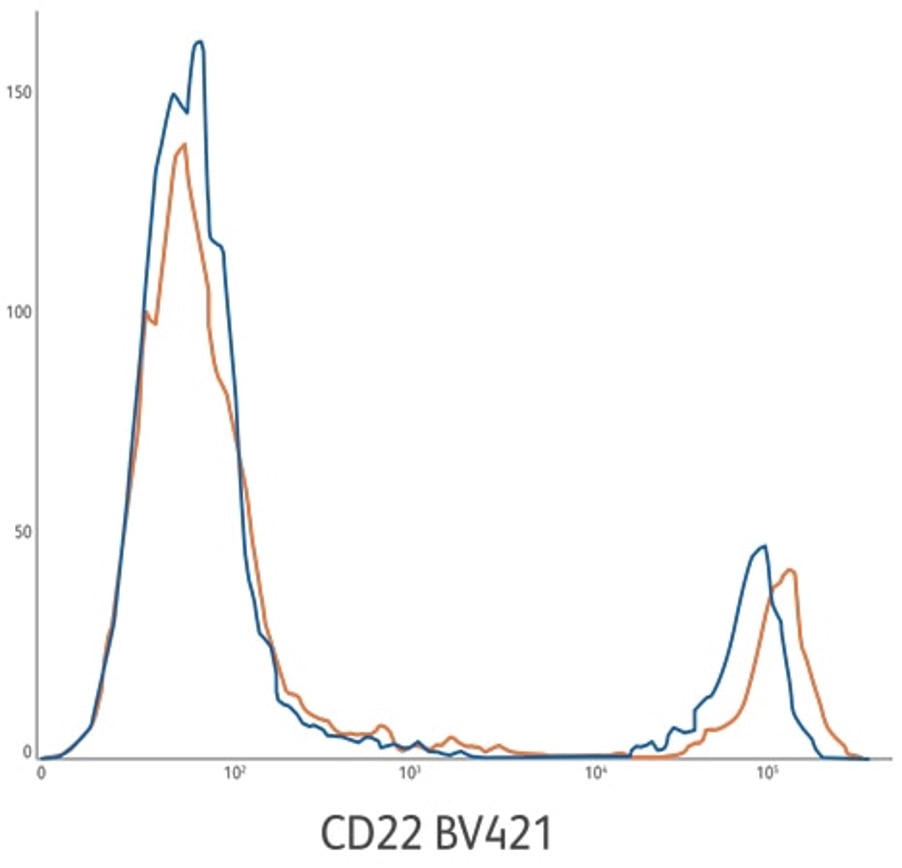
CD22 BV421 ASR (orange) compared to analogous CD22 BV421 RUO reagent (blue). The CD22 BV421 ASR development lots are on average 40% brighter based upon positive population MFI. Average stain index of 131 compared to 49. Lysed whole blood gated on lymphocytes.
Flexibility
With an expanded portfolio of ASRs for violet laser channels, you now have increased flexibility in
multicolor panel design. This allows more options to move panel markers to the violet laser channels
freeing up the blue and red laser channels.
Illustrative examples from product development are shown in Figures 6 and 7.
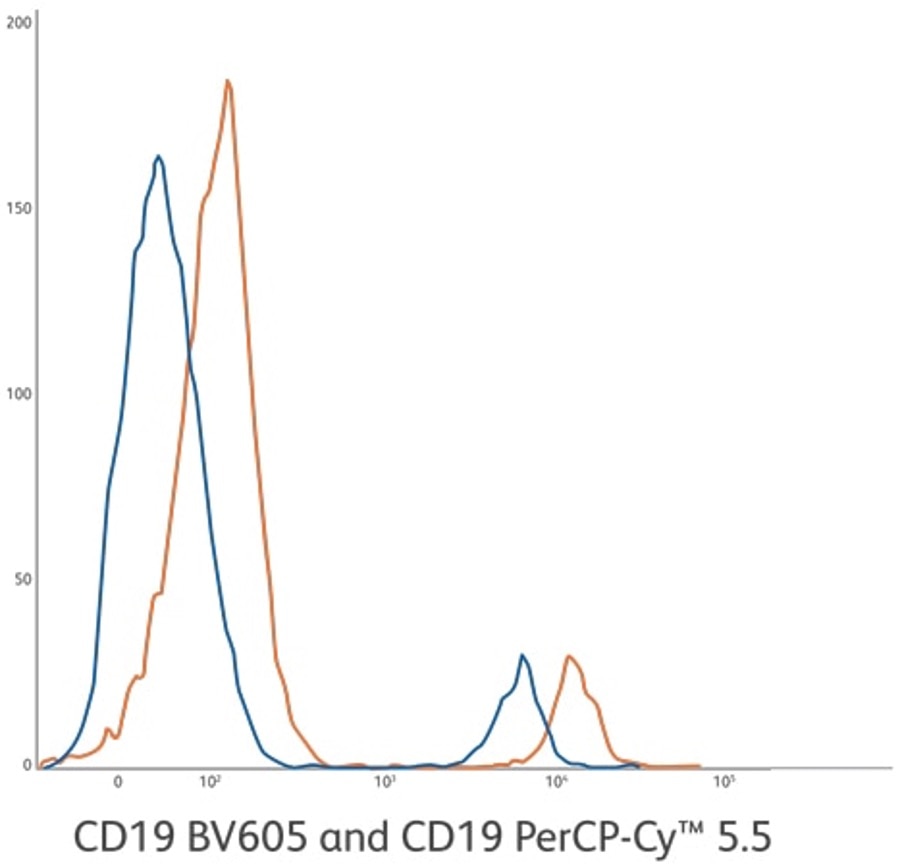
CD19 BV605 ASR (orange) compared to CD19 PerCP-Cy™5.5 ASR (blue). CD19 BV605 ASR development lot is 113% brighter based upon positive population MFI. Stain index of 132 compared to 69. Clone: SJ25C1. Lysed whole blood gated on lymphocytes.
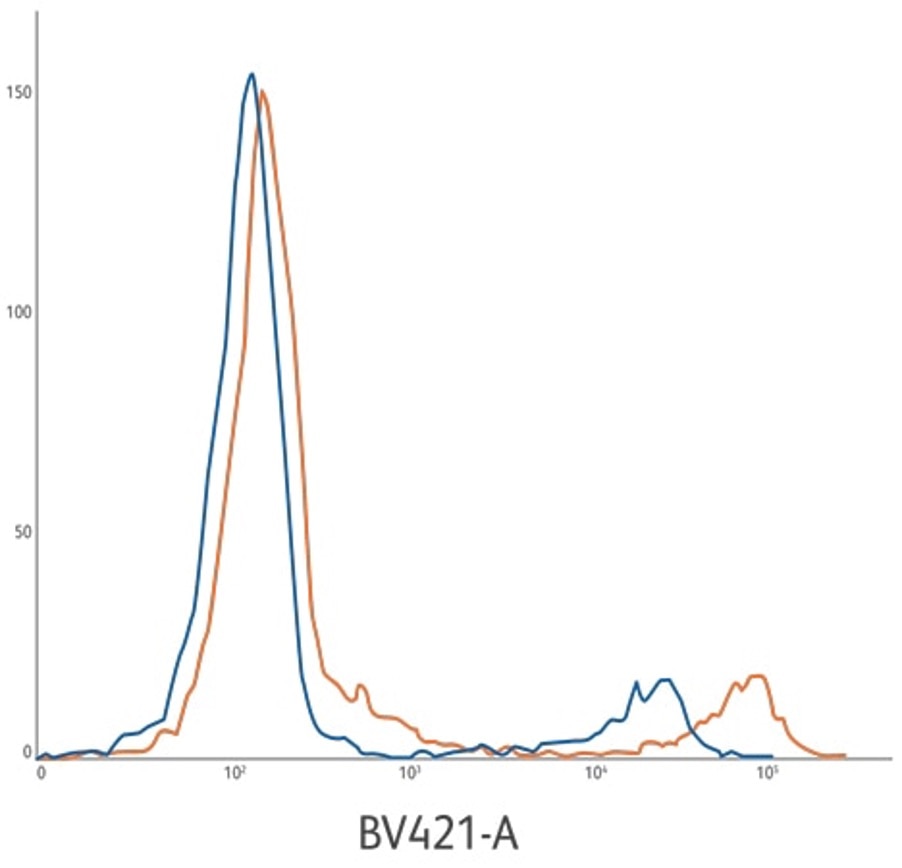
CD22 BV421 ASR (orange) compared to CD22 PE ASR (blue). CD22 B421 ASR development lot is 219% brighter based upon positive population MFI. Stain index of 499 compared to 196. Clone: HIB22. Lysed whole blood gated on lymphocytes.
New ASRs
The following BD Horizon Brilliant™ ASRs are now available for consistency, brightness and channel flexibility.
| Description | Clone | ASR Cat. No. | ASR with BD Horizon Brilliant Stain Buffer GPR Cat. No. |
|---|---|---|---|
| CD22 BV421 | HIB22 | 663979 | 663998 |
| CD33 BV421 | P67.6 | 663980 | 663999 |
| CD34 BV421 | 8G12 | 663981 | 664000 |
| CD117 BV421 | 104D2 | 663982 | 664001 |
| CD123 BV421 | 9F5 | 663983 | 664002 |
| CD5 BV605 | L17F12 | 663985 | 664004 |
| CD10 BV605 | HI10a | 663986 | 664005 |
| CD15 BV605 | HI98 | 663987 | 664006 |
| CD16 BV605 | 3G8 | 663988 | 664007 |
| CD19 BV605 | SJ25C1 | 663989 | 664008 |
| CD56 BV605 | NCAM16.2 | 663990 | 664009 |
| CD64 BV605 | 10.1 | 663991 | 664010 |
Further expanding the ASR portfolio beyond BD Horizon Brilliant™ ASRs, the following ASRs are now also available to increase panel flexibility.
* Seven to twelve color assays are for Research Use Only
For optimal results, use BD Horizon™ Brilliant Stain Buffer any time two or more BD Horizon Brilliant™ Reagents are used in the same multicolor staining cocktail.
Analyte Specific Reagent. Analytical and performance characteristics are not established.
GPR – General Purpose Reagent for Laboratory Use.
Cy is a trademark of GE Healthcare.
23-22810-00
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.


