Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

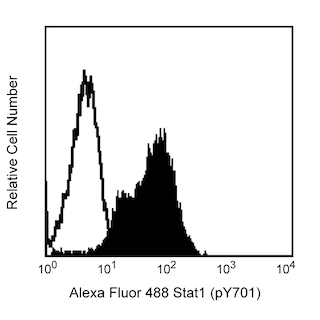
Analysis of Akt1 in human epithelioid carcinoma. HeLa S3 cells (ATCC CCL 2.2) were either transfected with Akt1 RNAi (open histogram) or untreated (shaded histogram). The cells were fixed (BD Cytofix™ buffer, Cat. No. 554655) for 10 minutes at 37°C, then permeabilized (BD Phosflow™ Perm Buffer III, Cat. No. 558050) on ice for 30 minutes, and then stained with Alexa Fluor® 488 Mouse anti- Akt1 (Cat. No. 560048). Down-regulation of Akt1 expression is evident in the RNAi-transfected cells. Flow cytometry was performed on a BD FACSCalibur™ flow cytometry system.
.png)

BD™ Phosflow Alexa Fluor® 488 Mouse anti-Akt1
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Either BD Cytofix™ fixation buffer or BD Phosflow™ Fix Buffer I may be used for cell fixation.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Alexa Fluor® 488 fluorochrome emission is collected at the same instrument settings as for fluorescein isothiocyanate (FITC).
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Species cross-reactivity detected in product development may not have been confirmed on every format and/or application.
- Alexa Fluor® is a registered trademark of Molecular Probes, Inc., Eugene, OR.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Companion Products




Akt [also known as PKB (Protein kinase B) or RAC-PK (Related to the A and C kinases] is a family of serine/threonine kinases that contains a pleckstrin homology (PH) domain. PH domains play important roles in signal transduction. There are three known isoforms of Akt in mammalian cells [Akt1 (α), Akt2 (β) and Akt3 (γ)]; they are thought to be regulated similarly. Akt is activated by insulin and growth factors by a mechanism involving phosphoinositide 3-OH kinase. Phosphoinositide 3-OH kinase products bind to the PH domain, resulting in translocation of Akt to the plasma membrane and activation of Akt to phospho-Akt by upstream kinases. Akt is phosphorylated within the activation loop at threonine 308 and the C-terminus at serine 473. Phospho-Akt promotes cell survival by inhibiting apoptosis. Specifically, phospho-Akt1 has been shown to phosphorylate Bad, a member of the Bcl-2 family that promotes cell death. This phosphorylation results in the inactivation of the proapoptotic function of Bad. The Akt molecule is thus considered to link extracellular survival signals (growth factors) with the apoptotic machinery (Bad). Akt is also a key mediator of the metabolic effects of insulin. Additionally, Akt has been referred to as an oncogene because it has increased activity in a number of tumors.
The 55/PKBa/Akt monoclonal antibody recognizes Akt1, regardless of phosphorylation status.
The specificity of this antibody conjugate for flow cytometric analysis was validated by confirming that RNA-mediated interference (RNAi) of the specific protein reduced the staining of the cells (see figure). Furthermore, the capacity of the RNAi to down-regulate the expression of the relevant protein was confirmed by western blot analysis.
Development References (5)
-
Alessi DR, Andjelkovic M, Caudwell B, et al. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J. 1996; 15(23):6541-6551. (Biology). View Reference
-
Cantley LC, Neel BG. New insights into tumor suppression: PTEN suppresses tumor formation by restraining the phosphoinositide 3-kinase/AKT pathway. Proc Natl Acad Sci U S A. 1999; 96(8):4240-4245. (Biology). View Reference
-
Datta SR, Dudek H, Tao X, et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell. 1997; 91:231-241. (Biology). View Reference
-
Ferrigno P, Silver PA. Regulated nuclear localization of stress-responsive factors: how the nuclear trafficking of protein kinases and transcription factors contributes to cell survival. Oncogene. 1999; 18(45):6129-6134. (Biology). View Reference
-
Kandel ES, Hay N. The regulation and activities of the multifunctional serine/threonine kinase Akt/PKB. Exp Cell Res. 1999; 253(1):210-229. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.