Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


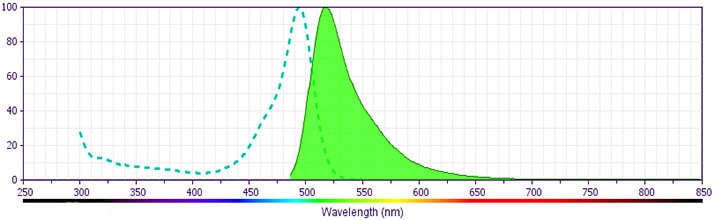
CD4 FITC
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
1. For in vitro diagnostic use.
2. When stored at 2° to 8°C, the antibody reagent is stable until the expiration date shown on the label. Do not use after the expiration date.
3. The antibody reagent should not be frozen or exposed to direct light during storage or during incubation with cells. Keep the reagent vial dry.
4. Alteration in the appearance of the reagent, such as precipitation or discoloration, indicates instability or deterioration. In such cases, the reagent should not be used.
5. The antibody reagents contain sodium azide as a preservative; however, care should be taken to avoid microbial contamination, which may cause erroneous results.
Becton Dickinson Immunocytometry Systems (BDIS) CD4 fluorescein isothiocyanate (FITC) reagent is a single-color direct immunofluorescence reagent for enumerating percentages of mature human helper/inducer (CD4+) lymphocytes in erythrocyte-lysed whole blood (LWB) or peripheral blood mononuclear cell (PBMC) suspensions.
Development References (25)
-
A National Committee for Clinical Laboratory Standards. Procedures for the Collection of Diagnostic Blood Specimens by Venipuncture (H3-A3). 1991. (Biology).
-
Angadi CV. Lack of Leu-3a epitope on T-helper (CD4) lymphocytes. J Clin Lab Anal. 1990; 4:193-195. (Biology).
-
Bernard A, Boumsell L, Hill C. Joint report of the first international workshop on human leucocyte differentiation antigens by the investigators of the participating laboratories. In: Bernard A, Boumsell L, Dausset J, Milstein C, Schlossman SF, ed. Leucocyte Typing. New York, NY: Springer-Verlag; 1984:9-108.
-
Centers for Disease Control. Guidelines for the performance of CD4+ T-cell determination in persons with human immunodeficiency virus infection. MMWR. 1992; 41:1-17. (Biology).
-
Dalgleish A, Beverly P, Clapham P, Crawford D, Greaves M, Weiss R. The CD4 (T4) antigen is an essential component of the receptor for the AIDS virus. Nature. 1984; 312:763-767. (Biology).
-
Engleman EG, Benike CJ, Glickman E, Evans RL. Antibodies to membrane structures that distinguish suppressor/cytotoxic and helper T lymphocyte subpopulations block the mixed leukocyte reaction in man. J Exp Med. 1981; 154(1):193-198. (Biology). View Reference
-
Evans RL, Wall DW, Platsoucas CD, et al. Thymus-dependent membrane antigens in man: inhibition of cell-mediated lympholysis by monoclonal antibodies to TH2 antigen. Proc Natl Acad Sci U S A. 1981; 78(1):544-548. (Biology). View Reference
-
Fourth International Workshop on Human Leucocyte Differentiation Antigens. Knapp W, Dörken B, Gilks W, et al, ed. Leucocyte Typing IV: White Cell Differentiation Antigens. 1989:1075.
-
Giorgi J, Hultin L. Lymphocyte subset alterations and immunophenotyping by flow cytometry in HIV disease. Clin Immunol Newslett. 1990; 10(4):55-61. (Biology).
-
Giorgi JV. Lymphocyte subset measurements: significance in clinical medicine. In: Rose NR, Friedman H, Fahey JL, ed. Manual of Clinical Laboratory Immunology. 3rd ed.. Washington, DC: American Society for Microbiology; 1986:236-246.
-
Jackson A. Basic phenotyping of lymphocytes: selection and testing of reagents and interpretation of data. Clin Immunol Newslett. 1990; 10:43-55. (Biology).
-
Jackson AL, Warner NL. Rose NR, Friedman H, Fahey JL, ed. Manual of Clincial Laboratory Immunology, Third Edition. Washington DC: American Society for Microbiology; 1986:226-235.
-
Kotzin BL, Benike CJ, Engleman EG. Induction of immunoglobulin-secreting cells in the allogeneic mixed leukocyte reaction: regulation by helper and suppressor lymphocyte subsets in man. J Immunol. 1981; 127(9):931-935. (Biology). View Reference
-
Landay A, Ohlsson-Wilhelm B, Giorgi JV. Application of flow cytometry to the study of HIV infection. AIDS. 1990; 4(6):479-497. (Biology). View Reference
-
Lewis DE, Puck JM, Babcock GF, Rich RR. Disproportionate expansion of a minor T cell subset in patients with lymphadenopathy syndrome and acquired immunodeficiency syndrome.. J Infect Dis. 1985; 151(3):555-9. (Biology). View Reference
-
Maddon PJ, Dalgleish AG, McDougal JS, Clapham PR, Weiss RA, Axel R. The T4 gene encodes the AIDS virus receptor and is expressed in the immune system and the brain.. Cell. 1986; 47(3):333-48. (Biology). View Reference
-
Mishell B, Shiigi S, Henry C, et al. Mishell B, Shiigi S, ed. Selected Methods in Cellular Immunology. New York: WH Freeman and Co; 1980:16-17.
-
National Committee for Clinical Laboratory Standards. Clinical Applications of Flow Cytometry: Quality Assurance and Immunophenotyping of Peripheral Blood Lymphocytes; Tentative Guideline (H42-T). NCCLS. 1992; 27395. (Biology).
-
Ohno T, Kanoh T, Suzuki T, et al. Comparative analysis of lymphocyte phenotypes between carriers of human immunodeficiency virus (HIV) and adult patients with primary immunodeficiency using two-color immunofluorescence flow cytometry.. Tohoku J Exp Med. 1988; 154(2):157-72. (Biology). View Reference
-
Prince H, Hirji K, Waldbeser L, Plaeger-Marshall S, Kleinman S, Lanier L. Influence of racial background on the distribution of T cell subsets and Leu-11-positive lymphocytes in healthy blood donors. Diag Immunol. 1985; 3:33-37. (Biology).
-
Reichert T, DeBruyere M, Deneys V, et al. Lymphocyte subset reference ranges in adult Caucasians. Clin Immunol Immunopathol. 1991; 60(2):190-208. (Biology). View Reference
-
Sattentau QJ, Dalgleish AG, Weiss RA, Beverley PC. Epitopes of the CD4 antigen and HIV infection. Science. 1986; 234(4780):1120-1123. (Biology). View Reference
-
Schmidt RE. Monoclonal antibodies for diagnosis of immunodeficiencies. Blut. 1989; 59:200-206. (Biology).
-
Stites DP, Casavant CH, McHugh TM, et al. Flow cytometric analysis of lymphocyte phenotypes in AIDS using monoclonal antibodies and simultaneous dual immunofluorescence.. Clin Immunol Immunopathol. 1986; 38(2):161-77. (Biology). View Reference
-
Wood GS, Warner NL, Warnke RA. Anti–Leu-3/T4 antibodies react with cells of monocyte/macrophage and Langerhans lineage. J Immunol. 1983; 131(1):212-216. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For In Vitro Diagnostics Use.
Documents are subject to revision without notice. Please verify you have the correct revision of the document, and always refer back to BD's eIFU website for the latest and most up to date information.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.