-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- BD OMICS-One™ WTA Next Assay
- BD OMICS-Guard™ CRYO Preservation Buffer
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- FACSAria Product Based Training
- FACSMelody Product-Based Training
- FACSLyric Product-Based Training
- FACSCanto Product-Based Training
- LSRFortessa Product-Based Training
- FACSymphony Product-Based Training
- FACSDuet Product-Based Training
- HTS Product-Based Training
- BD FACSDiscover™ S8 Cell Sorter Product Training
-
Advanced Training
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?
Waters Biosciences (formerly BD Biosciences) enables laboratories to design and build their own flow cytometry assays with a comprehensive portfolio of analyte-specific reagents (ASRs) for laboratory-developed tests (LDTs). Our flow cytometry-based immunophenotyping solutions feature an easy-to-use clinical cytometer, the BD FACSLyric™ Flow Cytometer, that includes standardized workflows, and powerful software for seamless data acquisition and analysis. We also offer IVD kits for accurate enumeration of lymphocytes, CD4+ T cells, and CD34+ stem cells. To further streamline lab operations, BD provides automated sample preparation instruments that boost efficiency and consistency—helping labs deliver reliable results, faster.
When no commercially available IVD assay meets a laboratory’s clinical or research needs, some laboratories choose to design and validate their own laboratory‑developed tests (LDTs). These lab‑defined assays are commonly used for applications such as cellular protein identification, cell population characterization, and the evaluation of biomarkers associated with cancer and other diseases.
We offer a broad portfolio of research‑use‑only (RUO) reagents and components that support laboratories in building and optimizing their own LDT workflows. These reagents are manufactured under GMP‑aligned quality systems to help ensure consistency and reliability during assay development and routine use. They come in a variety of single-color antibodies and pre-formulated ready-to-use cocktails – in liquid and dried format. In addition, we offer BD® CAR Detection Reagents, which identify CAR expressing cells and deliver workflow efficiency, high resolution and data quality.
Our portfolio includes a wide range of reagents and workflow components — such as staining reagents, lysing buffers, intracellular staining kits, and single‑antibody reagents — designed to provide flexibility when configuring lab‑defined panels. These solutions are complemented by the BD FACSLyric™ Flow Cytometer, equipped with a universal setup workflow to support reproducible, accurate, and standardized data acquisition across instruments.
Together, these RUO reagents and systems provide laboratories with the building blocks needed to design, standardize, and maintain lab‑defined assays, while retaining full control over assay validation and clinical decision‑making within their own laboratories.
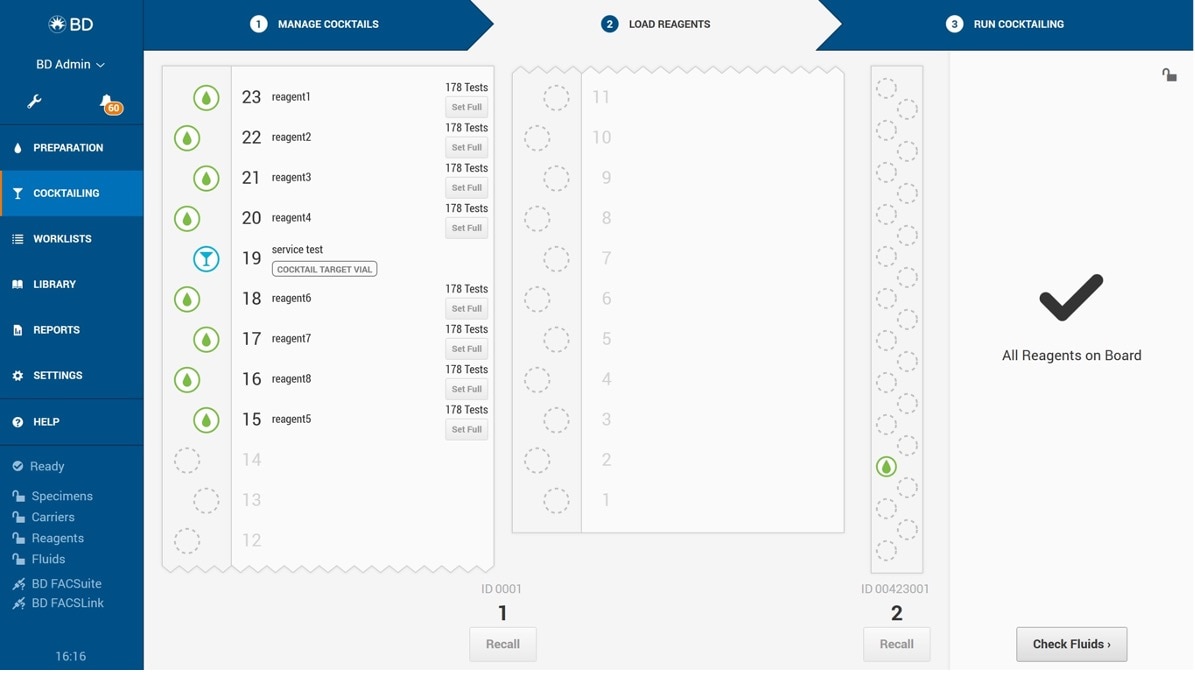
Streamline Accuracy with Automated Cocktail Preparation
When working with multiple single-color reagents, precision matters—especially in pipetting. BD offers automated flow cytometry solutions that enhance workflow efficiency and reduce manual errors. The BD FACSLyric™ Flow Cytometer, integrated with the BD FACSDuet™ Sample Preparation System, automates antibody cocktail preparation directly on board, eliminating pipetting mistakes and ensuring consistency.
The system digitally tracks every antibody used—including part number, lot number, and expiration date—making auditing and quality control seamless. Each cocktail can include up to 45 unique conjugates in a single vial, and user-defined recipes are stored in the software for easy reuse. This guarantees reproducibility and high-quality results, regardless of who prepares the assay.
The BD FACSLyric™ Flow Cytometer provides automated setup and QC for the acquisition and analysis of flow cytometry data. We also offer integrated connectivity and secure data solutions to optimize lab productivity.
Our IVD BD FACSLyric™ Flow Cytometer streamlines lab workflow through flexibility and automation, enabling efficiency and productivity. It is powered by the BD FACSuite™ Clinical Application for acquisition and analysis, supporting many functions and options relevant to U.S. FDA 21 CFR Part 11, such as password protection, audit trail, electronic signatures and IQ/OQ procedures. This lab-friendly system features a small footprint, quiet operation and the BD FACS™ Universal Loader. It offers a worklist driven workflow with user-friendly automated startup and shutdown. As part of daily Quality Control, Universal setup is an automated system that establishes and maintains instrument MFI target values and measure and maintain accurate spillover values on a single instrument using the BD FACSuite™ Clinical Application, BD® CS&T Beads, and BD® FC Beads, allowing for reproducible and consistent results within and between BD FACSLyric™ instruments over time. These settings are determined objectively by the system and are user-independent. Settings can be easily transferred from instrument to instrument via the BD FACSuite™ Clinical Application assay export and transfer feature automating standardization and enabling collaboration across sites.
The BD™ Stem Cell Enumeration (SCE) kit provides simultaneous enumeration of viable dual-positive CD45+/CD34+ hematopoietic stem cell populations in CD34+ absolute counts (cells/µL) as well as the percentage of the total viable leucocyte count that is CD34+ (%CD34). We also offer BD Multitest™ 6-Color TBNK Reagent with optional BD Trucount™ Tubes for assessing immune system reconstitution and status.
When no commercially available IVD assay meets a laboratory’s clinical or research needs, some laboratories choose to design and validate their own laboratory‑developed tests (LDTs). These lab‑defined assays are commonly used for applications such as cellular protein identification, cell population characterization, and the evaluation of biomarkers associated with cancer and other diseases.
We offer a broad portfolio of research‑use‑only (RUO) reagents and components that support laboratories in building and optimizing their own LDT workflows. These reagents are manufactured under GMP‑aligned quality systems to help ensure consistency and reliability during assay development and routine use. They come in a variety of single-color antibodies and pre-formulated ready-to-use cocktails – in liquid and dried format. In addition, we offer BD® CAR Detection Reagents, which identify CAR expressing cells and deliver workflow efficiency, high resolution and data quality.
Our portfolio includes a wide range of reagents and workflow components — such as staining reagents, lysing buffers, intracellular staining kits, and single‑antibody reagents — designed to provide flexibility when configuring lab‑defined panels. These solutions are complemented by the BD FACSLyric™ Flow Cytometer, equipped with a universal setup workflow to support reproducible, accurate, and standardized data acquisition across instruments.
Together, these RUO reagents and systems provide laboratories with the building blocks needed to design, standardize, and maintain lab‑defined assays, while retaining full control over assay validation and clinical decision‑making within their own laboratories.
Streamline Accuracy with Automated Cocktail Preparation
When working with multiple single-color reagents, precision matters—especially in pipetting. BD offers automated flow cytometry solutions that enhance workflow efficiency and reduce manual errors. The BD FACSLyric™ Flow Cytometer, integrated with the BD FACSDuet™ Sample Preparation System, automates antibody cocktail preparation directly on board, eliminating pipetting mistakes and ensuring consistency.
The system digitally tracks every antibody used—including part number, lot number, and expiration date—making auditing and quality control seamless. Each cocktail can include up to 45 unique conjugates in a single vial, and user-defined recipes are stored in the software for easy reuse. This guarantees reproducibility and high-quality results, regardless of who prepares the assay.

The BD FACSLyric™ Flow Cytometer provides automated setup and QC for the acquisition and analysis of flow cytometry data. We also offer integrated connectivity and secure data solutions to optimize lab productivity.
Our IVD BD FACSLyric™ Flow Cytometer streamlines lab workflow through flexibility and automation, enabling efficiency and productivity. It is powered by the BD FACSuite™ Clinical Application for acquisition and analysis, supporting many functions and options relevant to U.S. FDA 21 CFR Part 11, such as password protection, audit trail, electronic signatures and IQ/OQ procedures. This lab-friendly system features a small footprint, quiet operation and the BD FACS™ Universal Loader. It offers a worklist driven workflow with user-friendly automated startup and shutdown. As part of daily Quality Control, Universal setup is an automated system that establishes and maintains instrument MFI target values and measure and maintain accurate spillover values on a single instrument using the BD FACSuite™ Clinical Application, BD® CS&T Beads, and BD® FC Beads, allowing for reproducible and consistent results within and between BD FACSLyric™ instruments over time. These settings are determined objectively by the system and are user-independent. Settings can be easily transferred from instrument to instrument via the BD FACSuite™ Clinical Application assay export and transfer feature automating standardization and enabling collaboration across sites.
The BD™ Stem Cell Enumeration (SCE) kit provides simultaneous enumeration of viable dual-positive CD45+/CD34+ hematopoietic stem cell populations in CD34+ absolute counts (cells/µL) as well as the percentage of the total viable leucocyte count that is CD34+ (%CD34). We also offer BD Multitest™ 6-Color TBNK Reagent with optional BD Trucount™ Tubes for assessing immune system reconstitution and status.
Why BD?
From diagnosis to disease monitoring, BD Biosciences offers robust immunophenotyping solutions for clinical evaluation and surveillance.
- Streamlined workflow with flexibility and automation with the BD FACSLyric™ Flow Cytometer
- Full portfolio or ASR reagents supporting 12 channels on the BD FACSLyric™ Flow Cytometer for user defined testing
- Automated cocktailing with BD FACSDuet™ Sample Preparation System and BD FACSDuet™ Premium Sample Preparation System for LDTs
- IVD kits for stem cell enumeration and immune system reconstitution assessment
Analyte Specific Reagent. Analytical and performance characteristics are not established.
BD FACSLyric™ Flow Cytometer, BD FACSDuet™ Premium Sample Preparation System, and BD FACSDuet™ Sample Preparation System are Class 1 Laser Products.
The BD FACSLyric™ Flow Cytometer is for In Vitro Diagnostic Use with BD FACSuite™ Clinical Application for up to six colors. The BD FACSLyric™ Flow Cytometer is for Research Use Only with BD FACSuite™ Application for up to 12 colors. Not for use in diagnostic or therapeutic procedures.
The BD FACSDuet™ Sample Preparation System and BD FACSDuet™ Premium Sample Preparation System are for In Vitro Diagnostic Use. Sample preparation for user defined protocols and cocktailing functions are for Research Use Only. Not for use in diagnostic or therapeutic procedures.
The BD Multitest™ 6-Color TBNK with optional BD Trucount™ Tubes are for In Vitro Diagnostic Use with the BD FACSLyric™, BD FACSCanto™ II, and BD FACSCanto™ Flow Cytometer Systems.
The BD® Stem Cell Enumeration Kit is for In Vitro Diagnostic Use with the BD FACSLyric™ Flow Cytometer, BD FACSCanto™ II Flow Cytometer and the BD FACSCalibur™ Flow Cytometer.
BD® CS&T Beads are for In Vitro Diagnostic Use with BD FACSVia™ flow cytometer system with BD FACSVia™ clinical software or the BD FACSLyric™flow cytometer with BD FACSuite™ Clinical application.
The BD®FC Beads 7-Color Kit is for In Vitro Diagnostic Use on a BD FACSLyric™ flow cytometer with BD FACSuite™ Clinical application
