Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

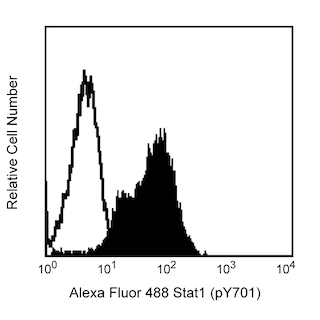
Analysis of eNOS in human endothelial cells. EA-hy 926 cells (Edgell, McDonald, Graham, 1983) were either transfected with eNOS RNAi (open histogram) or untreated (shaded histogram). The cells were fixed (BD Cytofix™ Fixation buffer, Cat. No. 554655) for 10 minutes at 37°C, then permeabilized (BD Phosflow™ Perm Buffer III, Cat. No. 558050) on ice for 30 minutes, and then stained with PE Mouse anti-eNOS (Cat. No. 560103). Down-regulation of eNOS expression is evident in the RNAi-transfected cells. Flow cytometry was performed on a BD™ LSR II flow cytometry system.
.png)

BD™ Phosflow PE Mouse anti-eNOS
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Either BD Cytofix™ fixation buffer or BD Phosflow™ Fix Buffer I may be used for cell fixation.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Companion Products



Nitric oxide synthase (NOS), a cell-type specific enzyme, catalyzes the synthesis of nitric oxide (NO). NO is a short-lived radical that transmits signals involved in vasorelaxation, neurotransmission, and cytotoxicity. In neurons and endothelial cells, constitutive NOS (cNOS) is activated by agonists that increase intracellular Ca[2+] levels and enhance calmodulin binding. Neuronal NOS (mNOS) and endothelial NOS (eNOS) have recognition sites for NADPH, FAD, FMN, and calmodulin and both are regulated in a similar manner. The human forms exhibit 52% amino acid identity. However, they are distinct gene products of about 155 kDa (mNOS) and 140 kDA (eNOS). The eNOS gene was cloned from human vascular endothelium as well as from bovine aortic endothelial cells (BAEC). eNOS protein has a unique N-myristylation consensus sequence that may explain its membrane localization.
The specificity of this antibody conjugate for flow cytometric analysis was validated by confirming that RNA-mediated interference (RNAi) of the specific protein reduced the staining of the cells (see figure). Furthermore, the capacity of the RNAi to down-regulate the expression of the relevant protein was confirmed by western blot analysis.
Development References (6)
-
Chen PF, Tsai AL, Wu KK. Cysteine 184 of endothelial nitric oxide synthase is involved in heme coordination and catalytic activity. J Biol Chem. 1994; 269(40):25062-25066. (Clone-specific: Western blot). View Reference
-
Dinerman JL, Dawson TM, Schell MJ, Snowman A, Snyder SH. Endothelial nitric oxide synthase localized to hippocampal pyramidal cells: implications for synaptic plasticity. Proc Natl Acad Sci U S A. 1994; 91(10):4214-4218. (Biology). View Reference
-
Edgell C-JS, McDonald CC, Graham JB. Permanent cell line expressing human factor VIII-related antigen established by hybridization. Proc Natl Acad Sci U S A. 1983; 80:3734-3737. (Methodology: Controls).
-
Nathan C, Xie QW. Regulation of biosynthesis of nitric oxide. J Biol Chem. 1994; 269(19):13725-13728. (Biology). View Reference
-
Shen YH, Zhang L, Utama B et al. Human cytomegalovirus inhibits Akt-mediated eNOS activation through upregulating PTEN (phosphatase and tensin homolog deleted on chromosome 10). Cardiovasc Res. 2006; 69(2):502-511. (Biology). View Reference
-
Varghese P, Harrison RW, Lofthouse RA, Georgakopoulos D, Berkowitz DE, Hare JM. β3-adrenoceptor deficiency blocks nitric oxide-dependent inhibition of myocardial contractility. J Clin Invest. 2000; 106(5):697-703. (Clone-specific: Western blot). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.