-
Your selected country is
Middle East / Africa
- Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
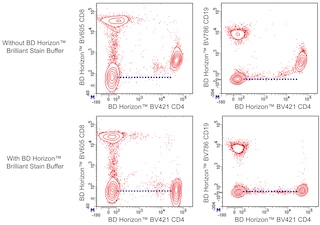
For optimal and reproducible results, BD Horizon Brilliant Stain Buffer should be used anytime two or more BD Horizon Brilliant dyes (including BD OptiBuild Brilliant reagents) are used in the same experiment. Fluorescent dye interactions may cause staining artifacts which may affect data interpretation. The BD Horizon Brilliant Stain Buffer was designed to minimize these interactions. More information can be found in the Technical Data Sheet of the BD Horizon Brilliant Stain Buffer (Cat. No. 563794).
Product Notices
- This antibody was developed for use in flow cytometry.
- The production process underwent stringent testing and validation to assure that it generates a high-quality conjugate with consistent performance and specific binding activity. However, verification testing has not been performed on all conjugate lots.
- Researchers should determine the optimal concentration of this reagent for their individual applications.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- BD Horizon Brilliant Stain Buffer is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,575,303; 8,354,239.
- BD Horizon Brilliant Ultraviolet 496 is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,227,187; 8,575,303; and 8,354,239.
Companion Products
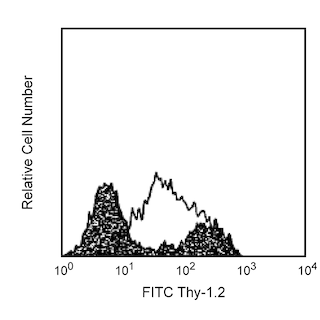



The B3B4 monoclonal antibody specifically binds to CD23, the low affinity IgE Fc receptor (FcεRII) expressed on mature resting conventional B lymphocytes, but not on B-1 cells (CD5+ B cells) or T lymphocytes. It does not react with high-affinity IgE receptors, as demonstrated on mouse mast cell lines. The regulation of CD23 surface expression on activated B cells appears to be complex, depending upon the mode of activation and the presence of cytokines. IgE synthesis is negatively regulated by CD23, and CD23 expression is upregulated on splenocytes in the presence of IgE. CD23 is also upregulated on follicular dendritic cells in the lymph nodes of immunized mice, and a subset of splenic dendritic cells expresses CD23. The B3B4 antibody abrogates antigen-specific IgE-dependent modulation of immune responses in normal mice. This monoclonal antibody also blocks IgE binding and eosinophil infiltration in the lung of immunized mice. Different in vivo results have been obtained when using the intact B3B4 antibody or the F(ab')2 fragments. B3B4 mAb does not cross-react with rat or human IgE Fc Receptor.
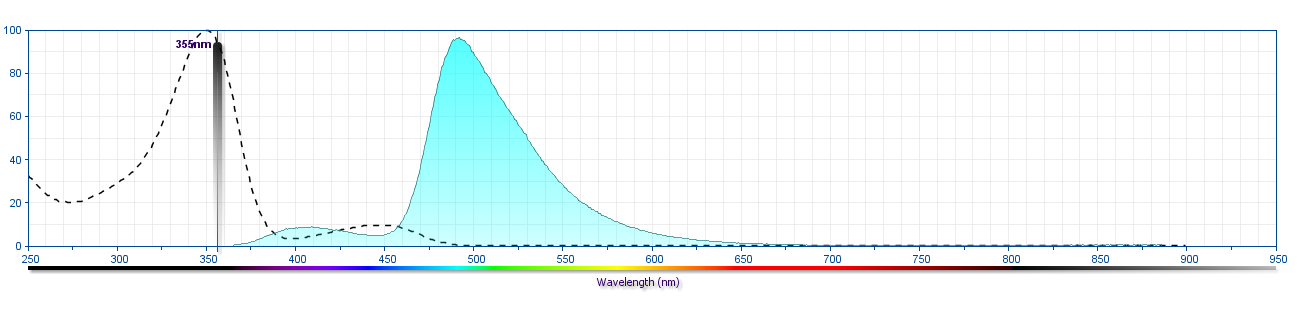
The antibody was conjugated to BD Horizon™ BUV496 which is part of the BD Horizon Brilliant™ Ultraviolet family of dyes. This dye is a tandem fluorochrome of BD Horizon BUV395 with an Ex Max of 348-nm and an acceptor dye with an Em Max at 496-nm. BD Horizon BUV496 can be excited by the ultraviolet laser (355 nm) and detected with a 515/30 nm filter with a 450LP. Due to the excitation of the acceptor dye by other laser lines, there may be significant spillover into the channel detecting BD Horizon V500 or BV510 (eg, 525/40-nm filter). However, the spillover can be corrected through compensation as with any other dye combination.
Development References (14)
-
Conrad DH, Waldschmidt TJ, Lee WT, et al. Effect of B cell stimulatory factor-1 (interleukin 4) on Fc epsilon and Fc gamma receptor expression on murine B lymphocytes and B cell lines. J Immunol. 1987; 139(7):2290-2296. (Clone-specific: Flow cytometry, Functional assay, Immunoaffinity chromatography, Immunoprecipitation, Radioimmunoassay). View Reference
-
Coyle AJ, Wagner K, Bertrand C, Tsuyuki S, Bews J, Heusser C. Central role of immunoglobulin (Ig) E in the induction of lung eosinophil infiltration and T helper 2 cell cytokine production: inhibition by a non-anaphylactogenic anti-IgE antibody. J Exp Med. 1996; 183(4):1303-1310. (Clone-specific: Blocking). View Reference
-
Dasic G, Juillard P, Graber P, et al. Critical role of CD23 in allergen-induced bronchoconstriction in a murine model of allergic asthma. Eur J Immunol. 1999; 29(9):2957-2967. (Clone-specific: Blocking, In vivo exacerbation). View Reference
-
Kisselgof AB, Oettgen HC. The expression of murine B cell CD23, in vivo, is regulated by its ligand, IgE. Int Immunol. 1998; 10(9):1377-1384. (Clone-specific: Flow cytometry). View Reference
-
Maeda K, Burton GF, Padgett DA, et al. Murine follicular dendritic cells and low affinity Fc receptors for IgE (Fc epsilon RII). J Immunol. 1992; 148(8):2340-2347. (Clone-specific: Electron microscopy, Immunohistochemistry). View Reference
-
Oshiba A, Hamelmann E, Haczku A, et al. Modulation of antigen-induced B and T cell responses by antigen-specific IgE antibodies. J Immunol. 1997; 159(8):4056-4063. (Clone-specific: Flow cytometry). View Reference
-
Pulendran B, Lingappa J, Kennedy MK, et al. Developmental pathways of dendritic cells in vivo: distinct function, phenotype, and localization of dendritic cell subsets in FLT3 ligand-treated mice. J Immunol. 1997; 159(5):2222-2231. (Clone-specific: Flow cytometry). View Reference
-
Rabin E, Cong YZ, Wortis HH. Loss of CD23 is a consequence of B-cell activation. Implications for the analysis of B-cell lineages. Ann N Y Acad Sci. 1992; 651:130-142. (Biology: Flow cytometry). View Reference
-
Rao M, Lee WT, Conrad DH. Characterization of a monoclonal antibody directed against the murine B lymphocyte receptor for IgE. J Immunol. 1987; 138(6):1845-1851. (Immunogen: Blocking, Immunoprecipitation, Inhibition, Radioimmunoassay). View Reference
-
Stief A, Texido G, Sansig G, et al. Mice deficient in CD23 reveal its modulatory role in IgE production but no role in T and B cell development. J Immunol. 1994; 152(7):3378-3390. (Clone-specific: Flow cytometry, Immunohistochemistry). View Reference
-
Waldschmidt T, Snapp K, Foy T, Tygrett L, Carpenter C. B-cell subsets defined by the Fc epsilon R. Ann N Y Acad Sci. 1992; 651:84-98. (Biology). View Reference
-
Waldschmidt TJ, Conrad DH, Lynch RG. Expression of B cell surface receptors. II. IL-4 can accelerate the developmental expression of the murine B cell IgE Fc receptor. J Immunol. 1989; 143(9):2820-2827. (Clone-specific: Flow cytometry, Immunoaffinity chromatography). View Reference
-
Waldschmidt TJ, Conrad DH, Lynch RG. The expression of B cell surface receptors. I. The ontogeny and distribution of the murine B cell IgE Fc receptor. J Immunol. 1988; 140(7):2148-2154. (Clone-specific: Flow cytometry). View Reference
-
Yu P, Kosco-Vilbois M, Richards M, Kohler G, Lamers MC. Negative feedback regulation of IgE synthesis by murine CD23. Nature. 1994; 369(6483):753-756. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.