-
Your selected location is
Middle East / Africa
- Change location/language
-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- BD OMICS-One™ WTA Next Assay
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Leukemia
According to the Global Cancer Statistics 2020, cases of leukemia are on the rise worldwide: almost 500.000 new cases of leukemia were registered in 2020. It is estimated globally that these numbers will rise more than 35% in the coming 20 years.1
These statistics strongly encourage to better understanding the origin of these diseases, finding the best way to diagnose and treat them, using state-of-the-art techniques and reagents.
The hematopoietic system is responsible for hematopoiesis, which is the physiological, dynamic, and continuous process ensuring the production of cellular blood components. The structure of the system is complex, and involves different organs and tissues including bone marrow, spleen, thymus and lymph nodes. Hematopoietic stem cells play a central role in there, due to their unique ability to give rise to all the different mature blood cell types and tissues. More precisely, hematopoietic stem cells will develop into either myeloid or lymphoid stem cells with different functions. Understanding the myeloid or lymphoid origin of pathological cells in the context of hematological disorders will be crucial for diagnosis, prognosis and patient therapy.
Myeloid stem cells will give rise to granulocytes, monocytes, red blood cells or platelets. While granulocytes and monocytes are fighting infections and killing bacteria, red blood cells are responsible to supply oxygen to all tissues of the body, and platelets are important to form blood clots in case of bleeding or injuries. Lymphoid stem cells develop into various types of fully differentiated cells which are crucial in fighting against infections and attacking abnormal cells which can enter the body. Three main cell types can be considered and constitute our immune system: B-cell lymphocytes, T-cell lymphocytes, and NK cells, all with specific characteristics and functions.
Many different stages can be identified during the normal development of myeloid and lymphoid stem cells, all the way to their fully differentiated and functional cellular state in blood or tissues. Transformation from a normal to a malignant state can occur at any stage of cell differentiation. Moreover, proliferation of malignant cells may become uncontrolled, and they can invade organs or tissue like bone marrow, blood, spleen, or lymph nodes. This is the origin of a hematopoietic disorder, and more specifically, of a leukemia and/or a lymphoma. Leukemia characterization and classification can be a challenge, due to their complexity and heterogeneity in term of severity, treatment, and outcome. Also, while some forms of leukemia are more common in children, others occur more frequently in adults.
Most types of leukemia usually involve white blood cells and are divided into two main groups, in relation to the type of cells they derive from: lymphocytic leukemias developing from lymphoid cells, and myeloid leukemias developing from myeloid cells.
Further classification is based on how quickly the disease develops: chronic leukemia is usually a slow-growing disease; however, some indolent forms of leukemia may become more aggressive over time. On the opposite, acute leukemia is fast-growing, progressing quickly without treatment. Acute leukemias are of major concern due to their often-poorer prognostic profiles at diagnosis; drastic therapies, like bone marrow transplantation, may be required.
What causes leukemia?
The exact cause of leukemia is yet unknown and may involve a combination of genetic (e.g.: chromosomal alterations in lymphocytes or myeloid progenitors in the bone marrow) and environmental factors (e.g.: radiations).2
Types of leukemia
Characterization and classification of leukemia is based on multiple parameters like the type of cells they derive from or the progression of the disease.
For example, Lymphocytic leukemia or Myeloid leukemia are two types of leukemia which involve lymphoid cells (lymphocytes) or myeloid cells (granulocytes, monocytes, …), respectively.
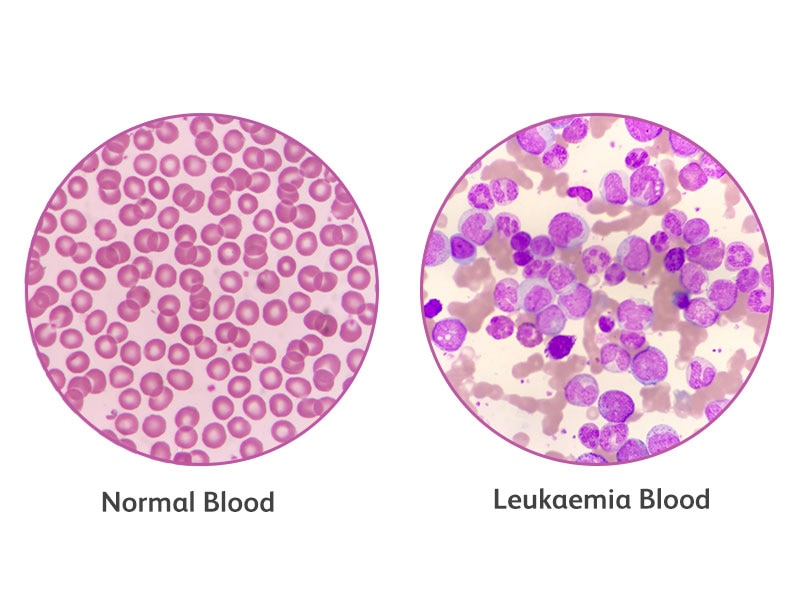
In term of disease progression, Acute Leukemia often shows fast-growing immature abnormal cells which is often associated with a bad diagnostic and prognostic outcome. On the opposite, chronic leukemia shows a slow-growing disease with a more indolent disease progression associated with mature cells. However, also indolent diseases may become more aggressive over time.
To develop a common language for health information across the world, the World Health Organization (WHO) has published a reference book on the classification of hematological malignancies.3 Although this classification is very complex and detailed, four (4) main types of leukemia exist, based on whether they are acute or chronic, and myeloid or lymphocytic: Acute myeloid leukemia (AML), Chronic lymphocytic leukemia (CLL) and Chronic myeloid leukemia (CML) are the three most frequent types of leukemia found in adults. Acute lymphoblastic leukemia (ALL) is the most frequent type of leukemia in children.
Of course, other types of leukemia exist, including hairy cell leukemia, myelodysplastic syndrome and myeloproliferative disorders. For an overview of all types please consider the World Health Organization publications and books.
How is leukemia diagnosed?
Leukemia is a complex multifactorial disease and diagnosis requires an interdisciplinary approach, including review of symptoms and physical examination, blood testing, bone marrow biopsy, morphology, cerebrospinal fluid (CSF) evaluation, radiology, and genetic testing.
Flow cytometry proves to be an important tool integral to leukemia diagnosis.4
Flow-cytometric immunophenotyping of Acute Leukemia
Flow cytometry is a powerful technique to identify cells and to study individual characteristics which can represent specific disease types. Flow cytometry is often used as an additional discipline besides morphology, histology, and cytology. For instance, stem cells, B or T lymphocytes can be hardly discriminated just based on their shape or granularity. Moreover, flow cytometry offers a commonly used method to discriminate between myeloid and lymphoid blasts.
- Identification of blasts (normal immature cells): blasts and immature cells can be identified by using specific expression profiles of membrane or intracellular molecules (CD markers) such as CD34 or CD45 in combination with cell characteristics like size and cytoplasmic complexity. Additional markers can be used for further differentiation between cell populations.
- Identification of aberrant blasts: Expression of abnormal antigens can be an indication for abnormal cells and justify further investigation. These abnormalities can include maturation arrest; expression of antigen of other lineage (e.g., lymphoid markers expressed on myeloid cells and vice versa)-; asynchronous expression of antigens (e.g., mature marker expressed on immature cells usually negative for the molecule)5 and abnormal expression of membrane or intracellular molecules (c.f.., CD10 expression in B-ALL, and CD34 expression in AML).4
Under homeostatic conditions, blast counts in the bone marrow or peripheral blood account for less than 5% of cells. Increased blast count is a cause for concern and warrants further investigation. Various treatment protocols define a threshold of blast counts in bone marrow above 20% from which leukemia can be suspected, investigated and if needed treated.5
Flow-cytometric immunophenotyping of Chronic Lymphocytic Leukemia
The diagnosis of Chronic Lymphocytic Leukemia (CLL) relies on cell counting, morphological analysis and immunophenotyping. Based on WHO diagnostic classification criteria, immunophenotyping of typical CLL demonstrates weak expression of CD20 with co-expression of CD5, CD19 and CD23 on the neoplastic B cells (with consistent counts of 5,000 circulating abnormal cells/mL in the peripheral blood during a 3-month period).6,7 Altogether, study of those markers associated with a systematic detection of kappa or lambda immunoglobulin light chain monoclonality (signature of the malignant expansion of abnormal B cells), are used with consensus in routine diagnostics and are considered required in screening panels.
Of course, this list is not exhaustive and other markers are recommended as they can provide additional information to better characterize neoplastic B cells or are of prognostic value e.g., weak CD79b, CD200, CD81 or CD38, CD45.4,6,7
Discrimination between blasts: myeloid versus lymphoid
Identifying the lymphoid or myeloid origin of malignant blasts is crucial, not only to be able to classify the leukemia but also to start as soon as possible the correct therapy. Treatments of AML and ALL are very different.
The classification criteria from both the WHO and European Group for the Immunological Characterization of Leukemias (EGIL) offer specific lineage markers that can be used to identify the types of leukemia by flow cytometry.4,8,9 For instance, normal myeloid blasts can be identified by using markers such as CD34, CD117,CD45 and Myeloperoxidase (MPO), while normal B-lymphoid blasts can be identified by using markers such as CD34, CD45, CD19 and cyCD79a. Normal T-lymphoid cells can be identified by using markers such as CD3, CD5 and cytoplasmatic CD3 (cyCD3).
Sub-grouping of leukemia
Classification used for sub-grouping of leukemia based on the WHO Guidelines
- Myeloproliferative Diseases/Myelodysplastic Syndromes
o Chronic Myelogenous Leukemia (CML)
o Myelodysplastic Syndrome (MDS)
- Acute Myeloid Leukemia (AML)
o Acute Promyelocytic Leukemia
o Acute Monocytic Leukemia
o Acute Erythroid Leukemia
o Acute Megakaryocytic Leukemia
o AML with maturation
- Precursor B- and T-cell Neoplasms
o Precursor B-lymphoblastic leukemia
o Precursor T-lymphoblastic leukemia
- Mature B-Cell Neoplasms
o Chronic lymphocytic leukemia
o Plasma cell neoplasms/myeloma
o Hairy cell leukemia
o Burkitt leukemia
- Mature T-Cell and NK-Cell Neoplasms
o T-cell prolymphocytic leukemia
o T-cell large granular lymphocytic leukemia
o NK cell leukemia
o Sezary syndrome
- Histiocytic and Dendritic Cell Neoplasms
- Mastocytosis
- Immunodeficiency-Associated Lymphoproliferative Disorders
Phenotypic evaluation
- Evaluation of mature B-cell lineage anomalies: to investigate B cell lineage, markers such as CD5, CD10, CD19, CD20, CD45, and immunoglobulin kappa and lambda are often used to characterize the B cells.6,7,9
- Evaluation of mature T-cell lineage and NK-cell anomalies: T cells and NK cells make up 15% of lymphoproliferative disorders. Interestingly, there are overlaps in T cell and NK cell antigen expression, allowing to use the same antibody panels to screen those two lineages. To investigate T/NK cell neoplasms, markers such as CD2, CD3, CD4, CD5, CD7 and CD8 markers can be evaluated, as well as CD16 and CD56.10
- Evaluation of myelomonocytic lineages: examples of myelomonocytic markers include CD13, CD33, CD16, CD10, CD11b, CD14 and CD64. Antibodies directed against those molecules can be used in the evaluation of myelomonocytic lineages.9
- Evaluation of plasma cell anomalies: in cases where abnormal antibody serum levels are observed, a plasma cell lineage screening panel including CD38, CD138, CD19 and CD56 markers can be used for further characterization.10
- Evaluation of abnormal expression profiles: evaluation of expression patterns of the various CD markers can be used to discriminate abnormal cells from their normal counterpart. Higher or lower expression of markers is often observed in abnormal cells.
Characterization of these anomalies is important not only for diagnosis, but also to track residual disease after therapy. These anomalies include arrested maturation, expression of antigen of another lineage (e.g., CD19, CD7 lymphatic markers on myeloid cells), asynchronous expression of antigens (e.g., CD15 mature neutrophil marker expression on CD34+ immature blasts), abnormal intensity of normal antigen expression (e.g., low CD3 expression in T-ALL, low or no CD20 expression in B-ALL). Also, aberrant CD5 expression is seen in CLL and aberrant CD10 expression can be observed in follicular lymphoma.11
Interestingly, these abnormal expression profiles can offer a specific marker expression which can be used to track residual pathological cells after therapy.
In summary, the knowledge of normal and aberrant expression patterns of specific molecules drives the choice of antibody used in flow cytometry panels in the context of hematological disorders. Based on that, flow cytometric immunophenotyping proves to be highly valuable in disease diagnosis, prognosis and monitoring of minimal residual disease.6,12,13
Flow cytometry–based assessment of minimal residual disease (MRD)
Minimal residual disease (MRD) is a term indicating that patients after therapy can still carry small amounts of pathological cells. Quantification of MRD has been reported to be helpful for therapy adjustment, such as to increase or decrease the therapy doses, or to determine if a patient is qualified for transplantation. Moreover, MRD follow up has been shown to be predictive for disease outcome, remission, or relapse: the prognostic utility of multicolor flow cytometry for assessing MRD in patients with Acute Lymphoblastic leukemia (ALL)14 and Acute Myeloid Leukemia (AML) has been established.15 MRD negativity assessment is often considered a positive predictor of good long-term prognosis.16
Historically, molecular biology has been commonly used to track residual disease in patients after treatment. Flow cytometry has been reported to be a sensitive method with which to detect minimal residual disease. Using sequential gating strategies and multiple markers it is possible to detect low numbers of cells which can be led back to the original phenotype of the diagnosis. In case of differences in marker expression between normal and residual abnormal cells, these differences can be used to monitor the disease progression over time.17 Flow cytometry nowadays can provide sensitivity of 1 x 10^6, comparably to molecular tests.12 However, since the ability to reach such high sensitivity and efficient detection has always been a challenge for flow cytometry, the availability of sensitive multicolor instruments has clearly resolved this with the possibility to increase the number of antibodies used to target a specific pathological population.
Flow cytometry facilitates the phenotypic characterization of leukemic cells at diagnosis, gives prognostic information relative to disease severity and allows monitoring of MRD. Sets of fluorescently labeled monoclonal antibodies can be used in multicolor flow cytometry to run panels of markers identifying MRD.14,15, 18, 19, 20
Flow cytometry is also used as a tool to monitor MRD in AML patients since PCR monitoring of fusion transcript amplification is only relevant in a fraction of patients. Standardized quantitative flow cytometry protocols can be used during MRD assessment with panels ranging from three to eight colors.20 MRD values greater than 0.01% (1 MRD cell/10,000 bone marrow cells in a bone marrow aspirate) are associated with high risk of relapse and poor survival.20
BD Biosciences offers reagents that can help in the diagnosis of hematological disorders as well as flow cytometry instruments
Our large portfolio of single-color antibody reagents spans across a range of specificities and dyes to help in the characterization of hematologic neoplasia. When analyzed separately, single reagents can provide only limited information in the analysis of leukemias and lymphomas while multicolor analysis using reagent combinations can provide more compelling and clinically relevant information.4
Multicolor panels, by definition, strongly increase the amount of information “per tube”, and it is helpful to not only characterize the abnormal cells, but also remaining normal cells in the same biological sample if they are still present. The remaining normal cells are often used for discrimination between normal and abnormal cells in the context of expression patterns.
Furthermore, BD Biosciences provides the BD OneFlow™ Solution, comprising a comprehensive set of reagents, setup beads, protocols and assay templates, to help support and standardize the diagnosis of hematological disorders, such as leukemia, lymphoma and myeloma. This can improve laboratory efficiency and enable reliability and accuracy of results.21,22
References
1. Sung H, Ferlay J, Siegel, RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 0:1-41. doi: 10.3322/caac.21660. Available online at cacancerjournal.com
2. Filippini T, Heck JE, Malagoli C, Del Giovane C, Vinceti M. A review and meta-analysis of outdoor air pollution and risk of childhood leukemia. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2015;33(1):36-66. doi:10.1080/10590501.2015.1002999
3. Swerdlow SH, Campo E, Harris NL, Jae ES, Pileri SA, Stein H, Thiele J (Eds.) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised 4th ed.; IARC: Lyon, France, 2017.
4. van Dongen JJM, Lhermitte L, Böttcher S, et al. EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia 2012;26:1908–1975. doi.org/10.1038/leu.2012.120
5. Bento LC, Correia RP, Pitangueiras Mangueira CL, et al. The use of flow cytometry in myelodysplastic syndromes: A review. Front Oncol. 2017;7:270. doi:10.3389/fonc.2017.00270
6. Jaseb K, Purrahman D, Shahrabi S, Ghanavat M, Rezaeean H, Saki N. Prognostic significance of aberrant CD5 expression in B-cell leukemia. Oncol Rev. 2019;13(1):400. doi:10.4081/oncol.2019.400
7. Rawstron et al. Reproducible Diagnosis of Chronic Lymphocytic Leukemia by Flow Cytometry. Cytometry B Clin Cytom. 2018; 94(1):121–128.
8. Bene M C , Castoldi G, Knapp W, Ludwig W D, Matutes E, Orfao A, van't Veer MB Proposals for the immunological classification of acute leukemias. European Group for the Immunological Characterization of Leukemias (EGIL) Leukemia 1995;9(10):1783-6.
9. Matarraz S, Almeida J, Flores-Montero J, et al. Introduction to the diagnosis and classification of monocytic-lineage leukemias by flow cytometry. Cytometry B Clin Cytom. 2017;92(3):218-227. doi:10.1002/cyto.b.21219
10. Jevremovic D, and Olteanu H. Flow Cytometry Applications in the Diagnosis of T/NK-Cell Lymphoproliferative Disorders. Cytometry Part B 2019; 96B: 99–115.
11. Seegmiller AC, Hsi ED, Craig FE. The current role of clinical flow cytometry in the evaluation of mature B-cell neoplasms. Cytometry B Clin Cytom. 2019;96(1):20-29. doi:10.1002/cyto.b.21756
12. Flores-Montero, J., Sanoja-Flores, L., Paiva, B. et al. Next Generation Flow for highly sensitive and standardized detection of minimal residual disease in multiple myeloma. Leukemia.2017; 31:2094–2103 https://doi.org/10.1038/leu.2017.29
13. Kruse A, Abdel-Azim N, Kim HN, et al. Minimal residual disease detection in acute lymphoblastic leukemia. Int J Mol Sci. 2020;21(3):1054. doi:10.3390/ijms21031054
14. Theunissen et al., Standardized Flow Cytometry for Highly Sensitive MRD Measurements in B-Cell Acute Lymphoblastic Leukemia. Blood.2017;129(3):347–357. https://doi.org/10.1182/blood-2016-07-726307
15. Schuurhuis et al., Minimal/Measurable Residual Disease in AML. Blood. 2018;131(12): 1275–1291. http://doi.org/10.1182/blood-2017-09-801498
16. Short NJ, Rafei H, Daver N, Hwang H, Ning J, Jorgensen JL, Kadia TM, DiNardo CD, Wang SA, Jabbour E, Popat U, Oran B, Cortes J, Konopleva M, Yilmaz M, Issa GC, Kantarjian H, Ravandi F. Prognostic impact of complete remission with MRD negativity in patients with relapsed or refractory AML. Blood Advances. 2020; 4(24):6117- 6126 doi:10.1182/bloodadvances.2020002811.
17. Kern W, Bacher U, Haferlach C, Schnittger S, Haferlach T. The role of multiparameter flow cytometry for disease monitoring in AML. Best Pract Res Clin Haematol. 2010;23(3):379-390. doi: 10.1016/j.beha.2010.06.007
18. Böttcher S. (2019) Minimal Residual Disease Quantification in Chronic Lymphocytic Leukemia: Clinical Significance and Flow Cytometric Methods. In: Malek S. (eds) Chronic Lymphocytic Leukemia. Methods in Molecular Biology, vol 1881. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8876-1_17
19. van Dongen JJ, van der Velden VH, Brüggemann M, Orfao A. Minimal residual disease diagnostics in acute lymphoblastic leukemia: need for sensitive, fast, and standardized technologies. Blood. 2015;125(26):3996-4009.
20. Perrot A, Lauwers-Cances V, Corre J, et al. Minimal residual disease negativity using deep sequencing is a major prognostic factor in multiple myeloma. Blood. 2018;132(23):2456-2464. doi:10.1182/blood-2018-06-858613
21. van der Velden VHJ, Flores-Montero J, Perez-Andres M, et al. Optimization and testing of dried antibody tube: The EuroFlow LST and PIDOT tubes as examples. J Immunol Methods. 2019;475:112287. doi: 10.1016/j.jim.2017.03.011
22. Moloney E, Watson H, Barge D, et al. Efficiency and health economic evaluations of BD OneFlow™ Flow Cytometry Reagents for diagnosing chronic lymphoid leukemia. Cytometry B Clin Cytom. 2019;96(6):514-520. doi: 10.1002/cyto.b.21779
BD Single-Color Antibody reagents, as intended on this page, and BD OneFlow™ Reagents are in vitro diagnostic medical devices bearing a CE mark.