-
Your selected location is
Middle East / Africa
- Change location/language
-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- BD OMICS-One™ WTA Next Assay
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?
Built on the research and validation work of the EuroFlow™ Consortium for the characterization of hematological malignancies for improved diagnostic outcomes1, the BD OneFlow™ Solution is a standardized, ready-to-use oncohaematological flow cytometry testing solution that streamlines both screening/ orientation and classification—empowering clinicians with next-level diagnostic insights for patient care. This solution can help maximize lab efficiency and reduce testing variability to deliver reliable results clinicians can trust.2, 3 The BD OneFlow™ Solution is a comprehensive set of reagents (BD OneFlow™ LST, B-CLPD T1, PCST, PCD , ALOT, AML T1, AML T2, AML T3, AML T4, and BCP-ALL T1), setup beads, protocols, and assay acquisition and analysis templates to reproducibly set up the flow cytometer and stain, acquire and analyze patient specimens for immunophenotyping of normal and aberrant cell populations.
Learn more from the BD OneFlow™ Solution brochure.
- Efficient workflows to reduce manual workload
- Ready-to-use, single-test tube format for direct specimen staining
- Convenient dried format
- Comprehensive solution of reagents, beads, protocols and assay templates
Our innovative BD OneFlow™ Solution brings the standardization of leukemia and lymphoma immunophenotyping one step forward.
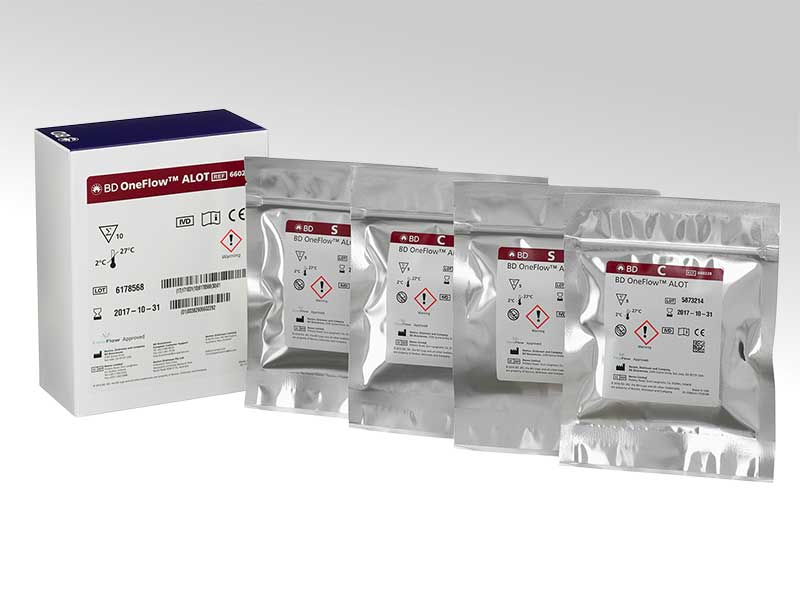
BD OneFlow™ Acute Leukemia Orientation Tube (ALOT)
The BD OneFlow™ ALOT is a preconfigured, ready-to-use 8-color reagent comprising two tubes—one containing the cytoplasmic markers (C tube) and the other containing the surface markers (S tube). The tube is intended for flow cytometric immunophenotyping of immature populations of hematopoietic cells (lymphoid and non-lymphoid lineage) in bone marrow and peripheral blood as an aid in the diagnosis of acute lymphoblastic leukemia and non-lymphoid acute leukemia.
BD OneFlow™ B-cell Precursor Acute Lymphoblastic Leukemia Tube 1 (BCP-ALL T1)
The BD OneFlow™ BCP-ALL T1 is a preconfigured, ready-to-use 8-color reagent provided in a single-test tube format. It can be used for qualitative flow-cytometric immunophenotyping of mature and immature hematopoietic cells populations (lymphoid lineage) in bone marrow and peripheral blood as an aid in the differential diagnosis of hematologically abnormal patients, including pediatric patients, having or suspected of having, B-cell acute lymphoblastic leukemia.
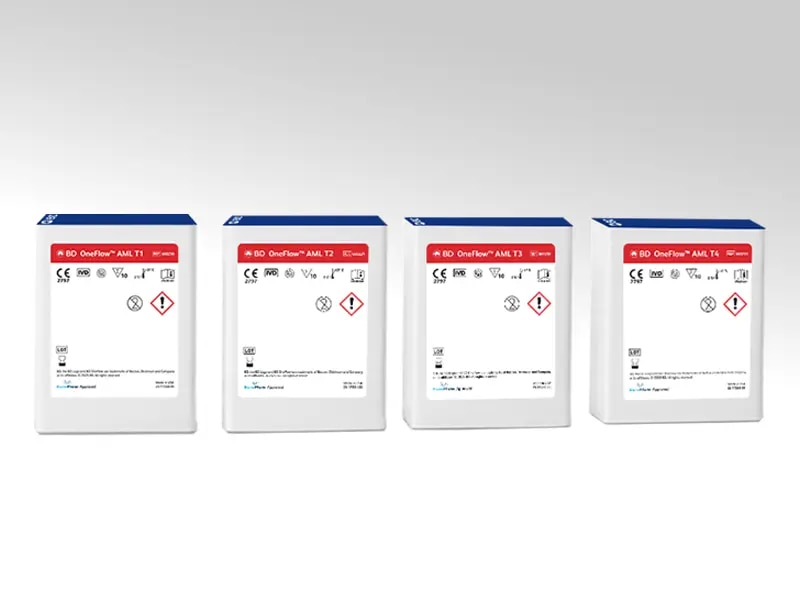
BD OneFlow™ Acute Myeloid Leukemia/MDS Panel (Acute Myeloid Leukemia Tubes 1 – 4; AML T1 – T4)
The BD OneFlow™ AML/MDS Panel comprises four (4) preconfigured, ready-to-use 8-color reagents, available separately, provided in a single-test tube format. The 4-tube panel can be used for qualitative flow-cytometric immunophenotyping of mature and immature hematopoietic cells populations (lymphoid and myeloid lineages) in bone marrow and peripheral blood, as an aid in the differential diagnosis of hematologically abnormal patients, having or suspected of having, acute myeloid leukemia, acute leukemia of ambiguous lineage, mixed phenotype acute leukemia, myelodysplastic syndrome, or myelodysplastic/myeloproliferative neoplasms.
BD OneFlow™ Chronic Lymphoproliferative Diseases Limited Panel: BD OneFlow™ Lymphoid Screening Tube (LST) and BD OneFlow™ B-cell Chronic Lymphoproliferative Diseases Tube 1 (B-CLPD T1)
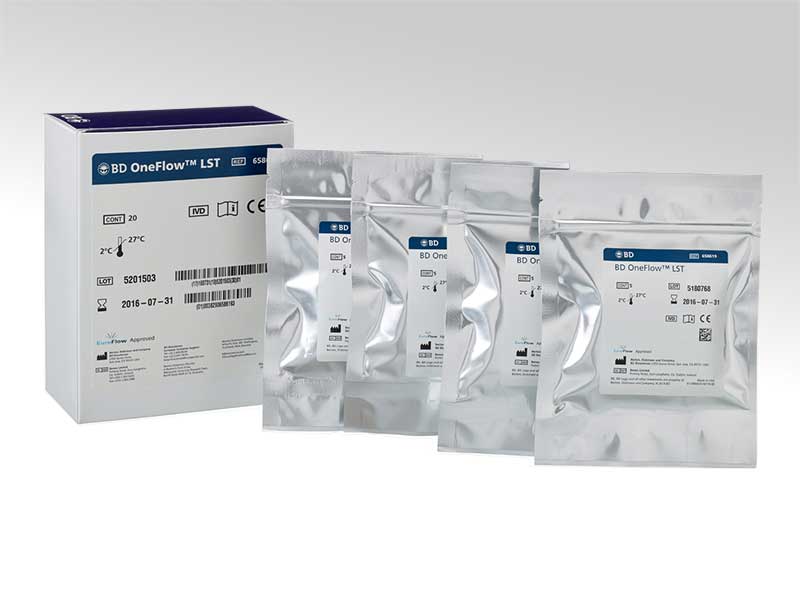
BD OneFlow™ Lymphoid Screening Tube (LST)
The BD OneFlow™ LST is a preconfigured, ready-to-use 8-color 12-antibodies reagent that is provided in a single-test tube format. It is intended for flow cytometric immunophenotyping of mature lymphocyte populations of B, T and NK cell lineages in peripheral blood, bone marrow and lymph nodes as an aid in diagnosis of hematological disorders.
BD OneFlow™ B-cell Chronic Lymphoproliferative Diseases Tube 1 (B-CLPD T1)
The BD OneFlow™ B-CLPD T1 is a preconfigured, ready-to-use 8-color reagent provided in a single-test tube format. It shall be used for specimens with B cell lineage populations needing further investigation in combination with the BD OneFlow™ LST. B-CLPD T1 is intended for flow cytometric immunophenotyping of B cells in peripheral blood and bone marrow as an aid in the diagnosis of chronic lymphocytic leukemia (CLL) and other B cell chronic lymphoproliferative diseases.
BD OneFlow™ Plasma Cell Disorders Panel: BD OneFlow™ Plasma Cell Screening Tube (PCST) and BD OneFlow™ Plasma Cell Disorders (PCD) Tube
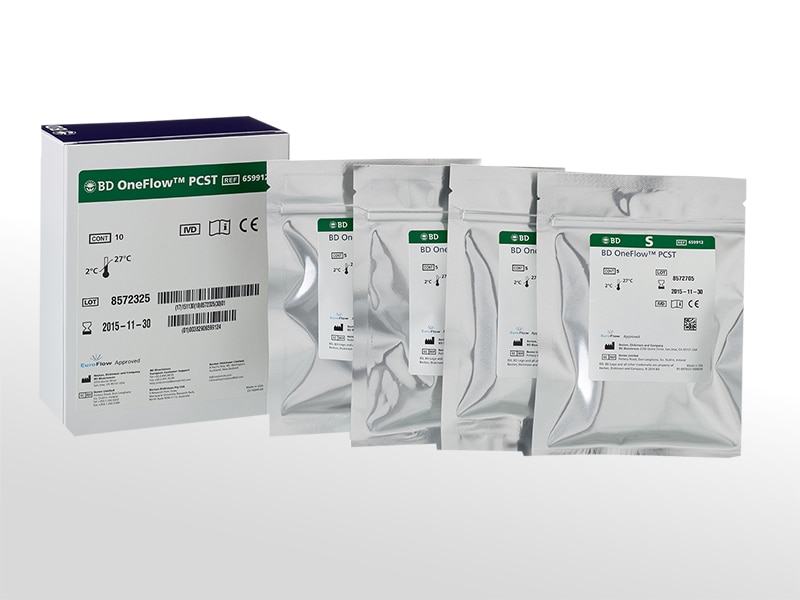
BD OneFlow™ Plasma Cell Screening Tube (PCST)
The BD OneFlow™ PCST is a preconfigured, ready-to-use 8-color reagent comprising two tubes—one containing the cytoplasmic markers (C tube) and the other containing the surface markers (S tube). It is intended for flow cytometric immunophenotyping of normal polyclonal and aberrant plasma cell populations in bone marrow as an aid in the diagnosis of plasma cell disorders.
BD OneFlow™ Plasma Cell Disorders (PCD) Tube
The BD OneFlow™ PCD Tube is a preconfigured, ready-to-use 8-color reagent provided in a single-test tube format. When run in parallel with the BD OneFlow™ PCST, it is intended for flow cytometric immunophenotyping of normal and aberrant plasma cells in bone marrow as an aid in the diagnosis of multiple myeloma or other plasma cell disorders.
References
- van Dongen JJM, Lhermitte L, Böttcher S, et al. on behalf of the EuroFlow Consortium (EU-FP6, LSHB-CT-2006-018708). EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia. 2012;26(9): 1908-1975. doi: 10.1038/leu.2012.120
- van der Velden VHJ, Flores-Montero J, Perez-Andres M, et al. Optimization and testing of dried antibody tube: The EuroFlow LST and PIDOT tubes as examples. J Immunol Methods. 2019;475:112287. doi: 10.1016/j.jim.2017.03.011
- Moloney E, Watson H, Barge D, et al. Efficiency and health economic evaluations of BD OneFlow™ Flow Cytometry Reagents for diagnosing chronic lymphoid leukemia. Cytometry B Clin Cytom. 2019;96(6):514-520. doi: 10.1002/cyto.b.21779
![]() The BD FACSLyric™ Flow Cytometer with the BD FACSuite™ Clinical and BD FACSuite™ Applications are an in vitro diagnostic medical device bearing a CE mark.
The BD FACSLyric™ Flow Cytometer with the BD FACSuite™ Clinical and BD FACSuite™ Applications are an in vitro diagnostic medical device bearing a CE mark.
![]() The BD OneFlow™ LST , BD OneFlow™ B-CLPD T1, BD OneFlow™ PCST, BD OneFlow™ PCD, BD OneFlow™ ALOT, BD OneFlow™ AML T1, BD OneFlow™ AML T2, BD OneFlow™ AML T3, BD OneFlow™ AML T4, BD OneFlow™ BCP-ALL T1 are in vitro diagnostic medical device bearing a CE mark and are CE certified by BSI Group the Netherlands B.V.
The BD OneFlow™ LST , BD OneFlow™ B-CLPD T1, BD OneFlow™ PCST, BD OneFlow™ PCD, BD OneFlow™ ALOT, BD OneFlow™ AML T1, BD OneFlow™ AML T2, BD OneFlow™ AML T3, BD OneFlow™ AML T4, BD OneFlow™ BCP-ALL T1 are in vitro diagnostic medical device bearing a CE mark and are CE certified by BSI Group the Netherlands B.V.
(Notified Body Number = 2797).
BD Flow Cytometers are Class 1 Laser Products.
The EuroFlow trademark is the property of the EuroFlow Consortium and cannot be reproduced or published without prior written permission from the EuroFlow coordinator (www.euroflow.org).
*This publication refers to EuroFlow and is relevant to the BD OneFlow™ LST, BD OneFlow™ B-CLPD T1, BD OneFlow™ ALOT, BD OneFlow™ PCST and BD OneFlow™ PCD as these were demonstrated to be equivalent to EuroFlow through a clinical trial.