-
Your selected location is
Middle East / Africa
- Change location/language
-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- BD OMICS-One™ WTA Next Assay
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?
Overview
The BD® Stem Cell Enumeration (SCE) Kit on the BD FACSLyric™ Flow Cytometer is an IVD solution with a simplified, acquisition-to-reporting and a standardized workflow. It follows ISHAGE guidelines that enables reliable enumeration of CD34+ stem cells for hematopoietic stem cell transplants while enhancing lab efficiency.
Learn more from the BD® Stem Cell Enumeration Kit brochure.
Why BD® Stem Cell Enumeration Kit
- Minimize errors by automatically calculating relevant results.
- Use trusted BD Trucount™ Tube technology for determining absolute CD34+ and CD45+ counts and two-level clinically relevant process controls, providing accurate and reproducible results on a single platform.
- Enhance workflow efficiency by reducing compensation frequency and minimizing hands-on time through an intuitive, guided workflow and faster, simpler assay setup.
- Work with a proven IVD solution that simplifies acquisition and gating following the International Society of Hematotherapy and Graft Engineering (ISHAGE) guidelines for bone marrow, peripheral blood, cord blood and leukapheresis products.

The BD® Stem Cell Enumeration Kit Enhances Lab Efficiency
Faster than BD FACSCanto II Flow Cytometer
Daily setup of the BD FACSLyric™ Flow Cytometer is 3.5x faster than the BD FACSCanto™ II Flow Cytometer with 39% less manual operator steps required.
Streamlined workflow
A streamlined workflow on BD FACSLyric™ Flow Cytometer removes the need for separate SCE optimization on the BD FACSCanto™ II Flow Cytometer thereby reducing the cost of daily assay setup and allowing you to get more tests out of the BD® SCE Kit.
Intuitive user interface
Intuitive user-interface, predefined assay template and automatic gating within the BD FACSuite™ Clinical Application simplifies analysis. Lab report includes multiple electronic signatures and calculated results.
Applications
The BD® Stem Cell Enumeration Kits for accurate and reliable enumeration of CD34+ stem cells
A single-tube, single platform IVD assay utilizing BD Trucount™ Tube technology
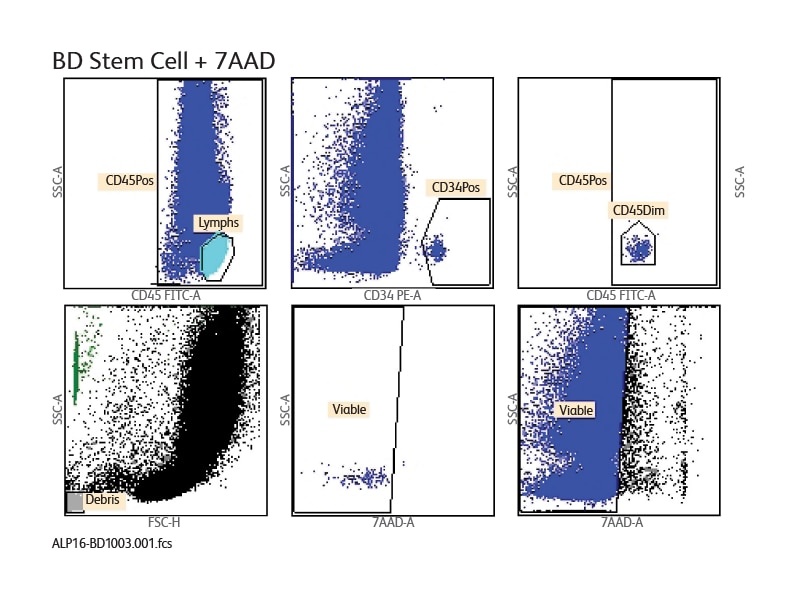
Results for fresh bone marrow are displayed in the report. Gating strategy follows the ISHAGE protocol.
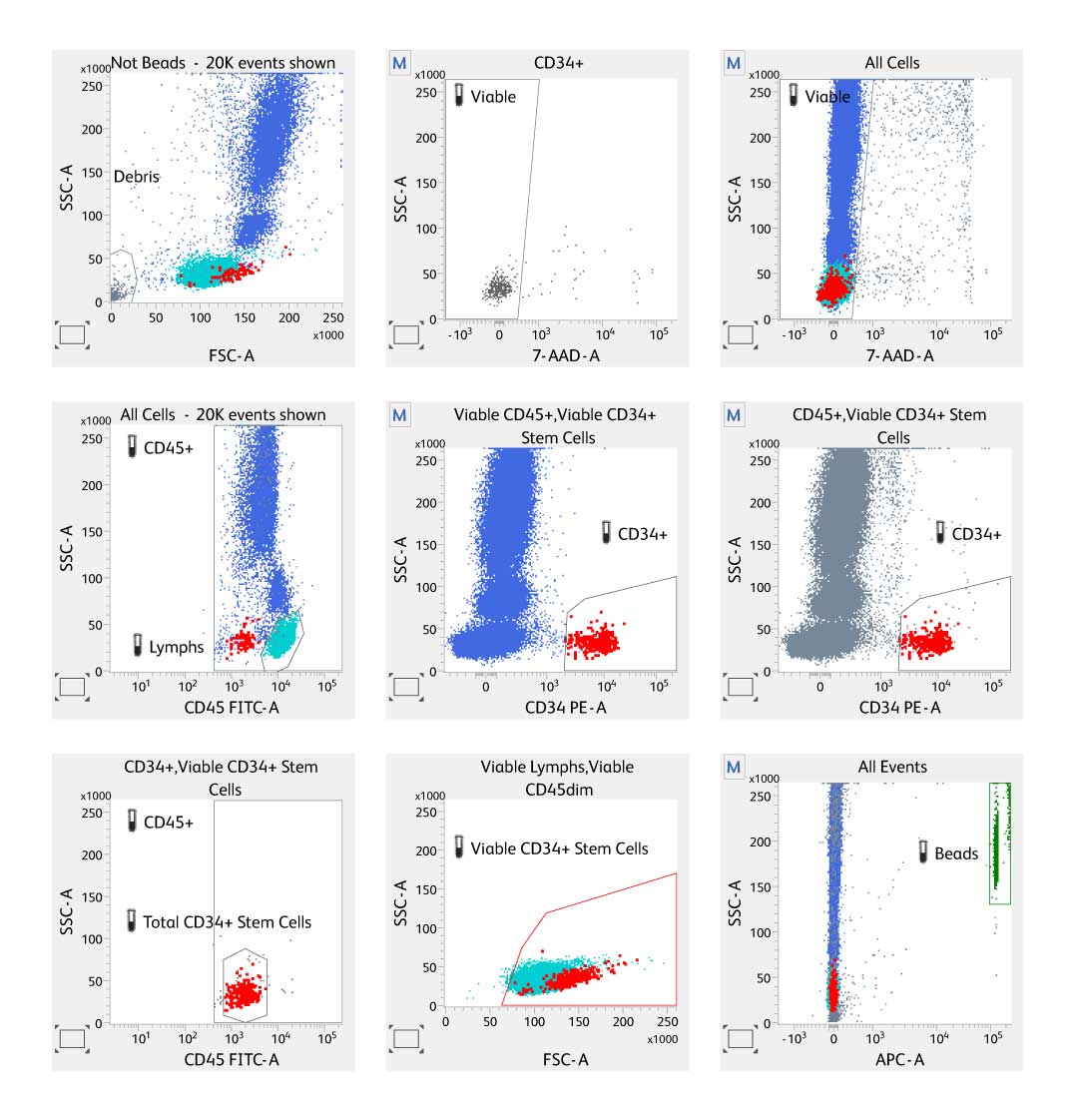
Results from the BD® Stem Cell Enumeration Assay in the BD FACSCanto™ Software.
-
Brochures
-
Posters
-
BD Stem Cell Enumeration Kit on FACSLyric Interference Evaluation Poster
-
Accurate enumeration of CD34+ cells with the BD® Stem Cell Enumeration Kit on the BD FACSLyric™ System
-
Evaluation of viable dual-positive CS45+/ CD34+ stem cells on BD FACSLyric™ System using BD® Stem Cell Enumeration Kit
-
Improved efficiency of BD® Stem Cell Enumeration (SCE) Kit on the BD FACSLyric™ Flow Cytometer as compared to BD FACSCanto™ II Flow Cytometers
-
Stability of fresh leukapheresis, fresh cord blood and fresh bone marrow specimens using the BD® Stem Cell Enumeration Kit on the BD FACSLyric™ Flow Cytometer
-
Multi-site evaluation of the BD® Stem Cell Enumeration Kit for CD34 Cell enumeration on BD FACSLyric™ and BD FACSCanto™ II Flow Cytometers.
-
CD34+ cell analysis on the BD FACSLyric™ Flow Cytometer using UK NEQAS samples and the BD®Stem Cell Enumeration (SCE) Kit
-
Instruction for Use
-
Application Guide
-
Protocol
The BD® Stem Cell Enumeration Kit is intended for use with the BD FACSLyric™ Flow Cytometer, BD FACSCanto™ II Flow Cytometer and the BD FACSCalibur™ Flow Cytometer.
|
BD® Stem Cell Enumeration Kit, BD® Stem Cell Control and BD FACSCanto™ II Flow Cytometer are in vitro diagnostic medical devices bearing a CE mark. BD Flow Cytometers are Class 1 Laser Products. BD FACSCalibur™ Flow Cytometer is discontinued. |
