Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
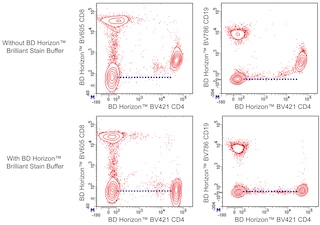
For optimal and reproducible results, BD Horizon Brilliant Stain Buffer should be used anytime two or more BD Horizon Brilliant dyes (including BD OptiBuild Brilliant reagents) are used in the same experiment. Fluorescent dye interactions may cause staining artifacts which may affect data interpretation. The BD Horizon Brilliant Stain Buffer was designed to minimize these interactions. More information can be found in the Technical Data Sheet of the BD Horizon Brilliant Stain Buffer (Cat. No. 563794).
Product Notices
- This antibody was developed for use in flow cytometry.
- The production process underwent stringent testing and validation to assure that it generates a high-quality conjugate with consistent performance and specific binding activity. However, verification testing has not been performed on all conjugate lots.
- Researchers should determine the optimal concentration of this reagent for their individual applications.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- BD Horizon Brilliant Stain Buffer is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,575,303; 8,354,239.
- BD Horizon Brilliant Ultraviolet 661 is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,227,187; 8,575,303; 8,354,239.
Companion Products
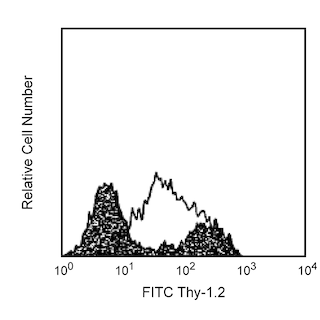


The H1.2F3 monoclonal antibody specifically binds to CD69 (Very Early Activation antigen), an 85 kDa disulfide-linked homodimer of differentially glycosylated subunits. CD69 is a C-type lectin, most closely related to the NKR-P1 and Ly-49 NK cell-activation molecules. Its expression is rapidly induced upon activation of lymphocytes (T, B, NK, and NK-T cells), neutrophils, and macrophages. CD69 is expressed also on thymocytes that are undergoing positive selection; its role in that process is unclear. H1.2F3 mAb augments PMA-induced T-cell stimulation and IFN-γ-induced macrophage stimulation. IL-2-activated NK cells express CD69, and H1.2F3 mAb induces redirected lysis of FcR-bearing target cells by NK cells.
The antibody was conjugated to BD Horizon™ BUV661 which is part of the BD Horizon Brilliant™ Ultraviolet family of dyes. This dye is a tandem fluorochrome of BD Horizon BUV395 with an Ex Max of 348-nm and an acceptor dye with an Em Max at 661-nm. BD Horizon Brilliant BUV661 can be excited by the ultraviolet laser (355 nm) and detected with a 670/25 filter and a 630 nm LP. Due to cross laser excitation of this dye, there may be significant spillover into channels detecting APC-like emissions (eg, 670/25-nm filter).
Due to spectral differences between labeled cells and beads, using BD™ CompBeads can result in incorrect spillover values when used with BD Horizon BUV661 reagents. Therefore, the use of BD CompBeads or BD CompBeads Plus to determine spillover values for these reagents is not recommended. Different BUV661 reagents (eg, CD4 vs. CD45) can have slightly different fluorescence spillover therefore, it may also be necessary to use clone-specific compensation controls when using these reagents.
Development References (15)
-
Bendelac A, Matzinger P, Seder RA, Paul WE, Schwartz RH. Activation events during thymic selection. J Exp Med. 1992; 175(3):731-742. (Clone-specific: Flow cytometry, Fluorescence activated cell sorting). View Reference
-
Gabor MJ, Godfrey DI, Scollay R. Recent thymic emigrants are distinct from most medullary thymocytes. Eur J Immunol. 1997; 27(8):2010-2050. (Clone-specific: Flow cytometry). View Reference
-
Karlhofer FM, Yokoyama WM. Stimulation of murine natural killer (NK) cells by a monoclonal antibody specific for the NK1.1 antigen. IL-2-activated NK cells possess additional specific stimulation pathways. J Immunol. 1991; 146(10):3662-3673. (Clone-specific: Induction). View Reference
-
Keefe R, Dave V, Allman D, Wiest D, Kappes DJ. Regulation of lineage commitment distinct from positive selection. Science. 1999; 286(5442):1149-1153. (Biology). View Reference
-
Lauzurica P, Sancho D, Torres M, et al. Phenotypic and functional characteristics of hematopoietic cell lineages in CD69-deficient mice. Blood. 2000; 95(7):2312-2320. (Clone-specific: Flow cytometry). View Reference
-
Marzio R, Jirillo E, Ransijn A, Mauel J, Corradin SB. Expression and function of the early activation antigen CD69 in murine macrophages. J Leukoc Biol. 1997; 62(3):349-355. (Clone-specific: Activation). View Reference
-
Merkenschlager M, Graf D, Lovatt M, Bommhardt U, Zamoyska R, Fisher AG. How many thymocytes audition for selection. J Exp Med. 1997; 186(7):1149-1158. (Clone-specific: Flow cytometry). View Reference
-
Nishimura T, Kitamura H, Iwakabe K, et al. The interface between innate and acquired immunity: glycolipid antigen presentation by CD1d-expressing dendritic cells to NKT cells induces the differentiation of antigen-specific cytotoxic T lymphocytes. Int Immunol. 2000; 12(7):987-994. (Clone-specific: Flow cytometry, Fluorescence activated cell sorting). View Reference
-
Punt JA, Suzuki H, Granger LG, Sharrow SO, Singer A. Lineage commitment in the thymus: only the most differentiated (TCRhibcl-2hi) subset of CD4+CD8+ thymocytes has selectively terminated CD4 or CD8 synthesis. J Exp Med. 1996; 184(6):2091-2099. (Clone-specific: Flow cytometry, Fluorescence activated cell sorting). View Reference
-
Sobel ES, Yokoyama WM, Shevach EM, Eisenberg RA, Cohen PL. Aberrant expression of the very early activation antigen on MRL/Mp-lpr/lpr lymphocytes. J Immunol. 1993; 150(2):673-682. (Clone-specific: Stimulation). View Reference
-
Wilkinson RW, Anderson G, Owen JJ, Jenkinson EJ. Positive selection of thymocytes involves sustained interactions with the thymic microenvironment. J Immunol. 1995; 155(11):5234-5240. (Clone-specific: Cell separation, Flow cytometry). View Reference
-
Yokoyama WM, Koning F, Kehn PJ, et al. Characterization of a cell surface-expressed disulfide-linked dimer involved in murine T cell activation. J Immunol. 1988; 141(2):369-376. (Immunogen: Flow cytometry, Immunoprecipitation, Stimulation). View Reference
-
Yokoyama WM, Maxfield SR, Shevach EM. Very early (VEA) and very late (VLA) activation antigens have distinct functions in T lymphocyte activation. Immunol Rev. 1989; 109:153-176. (Clone-specific: Flow cytometry, Stimulation). View Reference
-
Ziegler SF, Levin SD, Johnson L, et al. The mouse CD69 gene. Structure, expression, and mapping to the NK gene complex. J Immunol. 1994; 152(3):1228-1236. (Clone-specific: Flow cytometry). View Reference
-
Ziegler SF, Ramsdell F, Alderson MR. The activation antigen CD69. Stem Cells. 1994; 12(5):456-465. (Biology: Flow cytometry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.