-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- FACSAria Product Based Training
- FACSMelody Product-Based Training
- FACSLyric Product-Based Training
- FACSCanto Product-Based Training
- LSRFortessa Product-Based Training
- FACSymphony Product-Based Training
- FACSDuet Product-Based Training
- HTS Product-Based Training
- BD FACSDiscover™ S8 Cell Sorter Product Training
-
Advanced Training
-
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
- FACSAria Product Based Training
- FACSMelody Product-Based Training
- FACSLyric Product-Based Training
- FACSCanto Product-Based Training
- LSRFortessa Product-Based Training
- FACSymphony Product-Based Training
- FACSDuet Product-Based Training
- HTS Product-Based Training
- BD FACSDiscover™ S8 Cell Sorter Product Training
- Singapore (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from United States.
Would you like to stay on the current country site or be switched to your country?
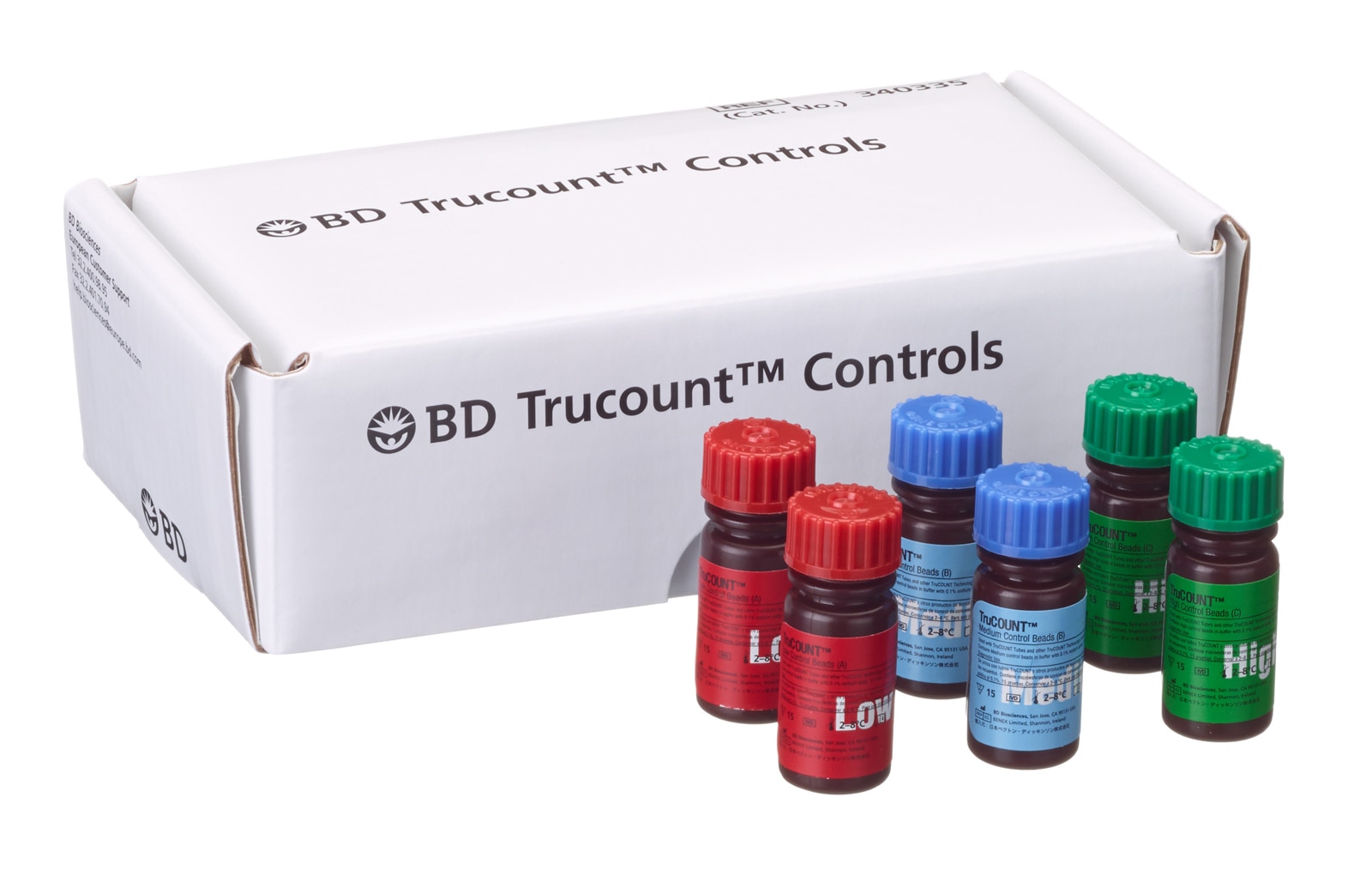
BD Trucount™ Controls

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Product Details
Description
BD Trucount™ Controls are designed for use with BD Trucount™ Tubes and the BD FACSLyric™ flow cytometer as a control for the absolute counting process. Specifically, a control bead value that is outside the expected range could indicate an error in pipetting or a problem with the bead count value from the BD Trucount™ Tubes. BD Trucount™ Controls can be used with the BD FACS™ Universal Loader.
Preparation And Storage
Store at 2–8 °C. Reagent in opened or unopened vials is stable until the expiration date shown on the vial label. Do not use after this expiration date.
Development References (4)
-
Centers for Disease Control and Prevention. 2007 Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings. Available: https://www.cdc.gov/infectioncontrol/guidelines/isolation/index.html March 12, 2019. View Reference
-
Clinical and Laboratory Standards Institute. Collection of Diagnostic Venous Blood Specimens, 7th ed. In: CLSI. CLSI, ed. CLSI document GP41-A7. Wayne, PA: Clinical and Laboratory Standards Institute; 2017:1-85. View Reference
-
Clinical and Laboratory Standards Institute. Protection of Laboratory Workers from Occupationally Acquired Infections. In: CLSI. CLSI, ed. CLSI document M29-A4. Wayne, PA: Clinical and Laboratory Standards Institute; 2014:1-133. View Reference
-
Jackson AL, Warner NL. Rose NR, Friedman H, Fahey JL, ed. Manual of Clinical Laboratory Immunology 3rd ed. Washington, DC: American Society for Microbiology; 1986:226-235.
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.