Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

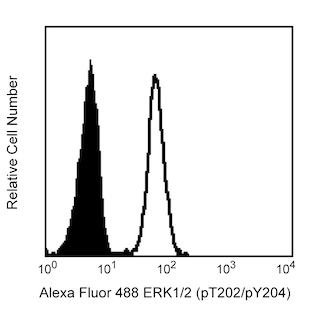
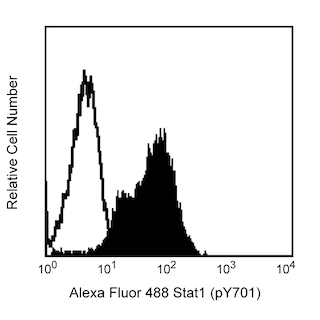
Flow cytometric analysis of Stat6 (pY641) expression in IL-4 stimulated human peripheral blood lymphocytes. Whole blood was either left untreated (dashed line histogram) or treated (solid line histogram) with 100 ng/ml (final concentration) of Recombinant Human IL-4 protein (Cat. No. 554605) for 15 minutes at 37°C. The samples were then treated with BD Phosflow™ Lyse/Fix Buffer (Cat. No. 558049) to lyse erythrocytes and fix leucocytes for 10 minutes at 37°C. The cells were permeabilized with BD Phosflow™ Perm Buffer III (Cat. No. 558050) on ice for 30 minutes, washed twice with BD Pharmingen™ Stain Buffer (FBS) (Cat. No. 554656), and then stained with BD Horizon™ R718 Mouse Anti-Human Stat6 (pY641) antibody (Cat No. 567081). The fluorescence histograms showing the expressed levels of Stat6 (pY641) were derived from gated events with the forward and side light-scatter characteristics of intact lymphocytes. Flow cytometry and data analysis were performed using a BD LSRFortessa™ X-20 Cell Analyzer System and FlowJo™ software.
.png)

BD Phosflow™ R718 Mouse Anti-Stat6 (pY641)
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
BD™ CompBeads can be used as surrogates to assess fluorescence spillover (Compensation). When fluorochrome conjugated antibodies are bound to BD CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cells and BD CompBead to ensure that BD CompBeads are appropriate for your specific cellular application.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- This product is provided under an Agreement between BIOTIUM and BD Biosciences. This product, and only in the amount purchased by buyer, may be used solely for buyer’s own internal research, in a manner consistent with the accompanying product literature. No other right to use, sell or otherwise transfer (a) this product, or (b) its components is hereby granted expressly, by implication or by estoppel. This product is for research use only. Diagnostic uses require a separate license from Biotium, Inc. For information on purchasing a license to this product including for purposes other than research, contact Biotium, Inc., 3159 Corporate Place, Hayward, CA 94545, Tel: (510) 265-1027. Fax: (510) 265-1352. Email: btinfo@biotium.com.
- Alexa Fluor™ is a trademark of Life Technologies Corporation.
Companion Products


.png?imwidth=320)
Interleukin-4 (IL-4), a major immunoregulatory cytokine, is secreted by activated T lymphocytes, basophils, and mast cells and plays an important role in modulating T helper cell lineage development. It induces specific gene expression via the tyrosine phosphorylation of Stat6 at tyrosine 641 (Y641). Stat6, a member of the signal transducers and activators of transcription protein family, mediates signals for IL-4 and, possibly, IL-13. While Stat6 is widely expressed in human tissues, it exhibits elevated expression in peripheral blood lymphocytes, colon, intestine, ovary, prostate, thymus, spleen, kidney, liver, lung, and placenta. Following cytokine receptor ligation, Jak kinases are activated and phosphorylate the cytoplasmic tails of the oligomerized receptors. The SH3:SH2 domain of Stat6 associates with tyrosine-phosphorylated IL-4 receptor and the proximal Jak kinase phosphorylates Stat6 at Y641 on the C-terminal side of the SH2 domain. Stat6 is then released from the receptor, dimerizes, and is thought to contact the basal transcription machinery by binding to p300/CBP. Thus, Stat6 mediates the IL-4 signal and is essential for the proper development of adaptive immunity.
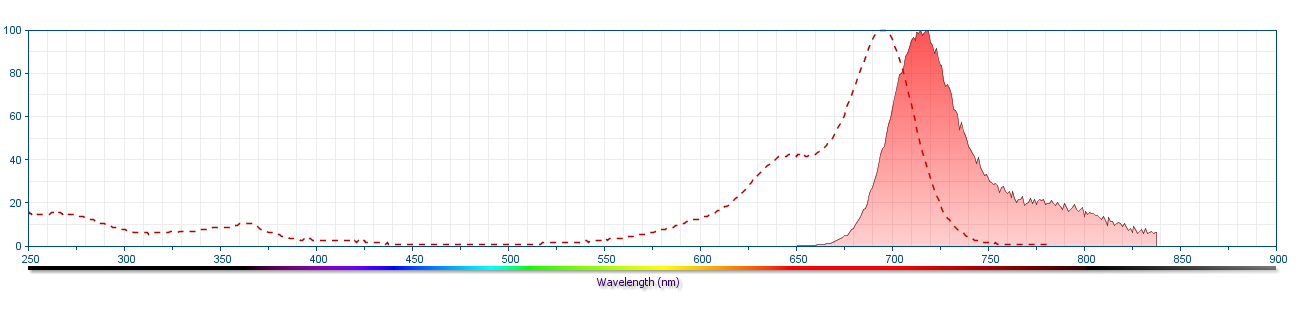
The antibody was conjugated to BD Horizon™ Red 718, which has been developed exclusively by for BD Biosciences as a better alternative to Alexa Fluor™ 700. BD Horizon™ Red 718 can be excited by the red laser (628–640 nm) and, with an Em Max around 718 nm, it can be detected using a 730/45 nm filter. Due to similar excitation and emission properties, we do not recommend using R718 in combination with APC-R700 or Alexa Fluor™ 700.
Development References (10)
-
Bromberg J, Darnell JE. The role of STATs in transcriptional control and their impact on cellular function. Oncogene. 2000; 19(21):2468-2473. (Biology). View Reference
-
Christophi GP, Panos M, Hudson CA, et al. Macrophages of multiple sclerosis patients display deficient SHP-1 expression and enhanced inflammatory phenotype.. Lab Invest. 2009; 89(7):742-59. (Clone-specific: Flow cytometry). View Reference
-
Dent AL, Hu-Li J, Paul WE, Staudt LM. T helper type 2 inflammatory disease in the absence of interleukin 4 and transcription factor STAT6. Proc Natl Acad Sci U S A. 1998; 95(23):13823-13828. (Biology). View Reference
-
Heim MH. The Jak-STAT pathway: specific signal transduction from the cell membrane to the nucleus. Eur J Clin Invest. 1996; 26(1):1-12. (Biology). View Reference
-
Hou J, Schindler U, Henzel WJ, Ho TC, Brasseur M, McKnight SL. An interleukin-4-induced transcription factor: IL-4 Stat. Science. 1994; 265(5179):1701-1706. (Biology). View Reference
-
Irish JM, Myklebust JH, Alizadeh AA, et al. B-cell signaling networks reveal a negative prognostic human lymphoma cell subset that emerges during tumor progression.. Proc Natl Acad Sci U S A. 2010; 107(29):12747-12754. (Clone-specific: Flow cytometry). View Reference
-
Krutzik PO, Nolan GP. Intracellular phospho-protein staining techniques for flow cytometry: monitoring single cell signaling events. Cytometry A. 2003; 55(2):61-70. (Clone-specific: Flow cytometry). View Reference
-
Mikita T, Campbell D, Wu P, Williamson K, Schindler U. Requirements for interleukin-4-induced gene expression and functional characterization of Stat6. Mol Cell Biol. 1996; 16(10):5811-5820. (Biology). View Reference
-
Quelle FW, Shimoda K, Thierfelder W, et al.. Cloning of murine Stat6 and human Stat6, Stat proteins that are tyrosine phosphorylated in responses to IL-4 and IL-3 but are not required for mitogenesis. Mol Cell Biol. 1995; 15(6):3336-3343. (Biology). View Reference
-
Suni MA, Maino VC. Flow cytometric analysis of cell signaling proteins. Methods Mol Biol. 2011; 717:155-169. (Clone-specific: Flow cytometry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.