-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Thought Leadership
- Product News
- Blogs
-
Scientific Publications
-
Events
- Expanding PARADIGM to Infectious Disease Modeling: HIV & Tuberculosis
- CYTO 2023: Advancing the World of Cytometry
- Advances in Immune Monitoring Series
- Validating Flow Cytometry Assays for Cell Therapy
- Enhancing Cell Analysis with a New Set of Eyes
- BD Biosciences at International Clinical Cytometry Society 2025
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
BD Pharmingen™ Purified Mouse Anti-Human TRADD
Clone B36-2 (RUO)
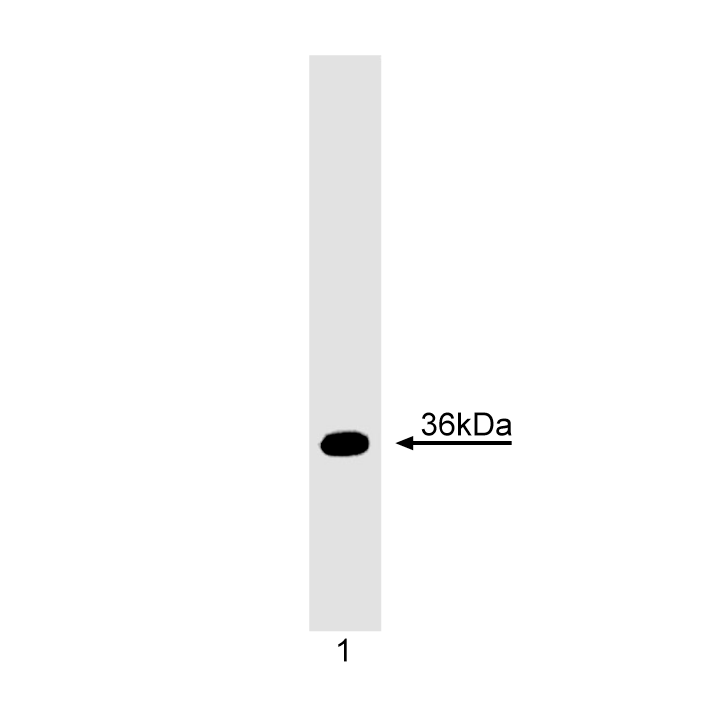



Western blot analysis of TRADD. Daudi lymphoma cell lysates were probed with anti-human TRADD (clone B36-2). TRADD is identified as a 36 kDa band.



Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Daudi B lymphoma cells (ATCC CRL-213) and Jurkat T cells (ATCC TIB-152) are suggested as positive controls.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
TRADD (TNF Receptor Associated Death domain) is an ~36 kDa protein that interacts specifically with the cytoplasmic domain of the type I TNF Receptor. Overexpression of the C-terminal 118 amino acids of TRADD, the "death domain," is sufficient to induce two major TNF-induced responses, apoptosis and activation of nuclear transcription factor, NF-κB. Evidence suggests that TRADD interacts with other signal molecules including TRAF1, TRAF2 and FADD, allowing recruitment of these molecules to the TNF receptor complex. A similar interaction has been demonstrated for TRADD and the serine/threonine kinase RIP. Coexpression of the ICE-specific protease inhibitor, CrmA, inhibits TRADD-induced apoptosis, but does not affect induction of NF-κB, suggesting that TRADD may serve to initiate distinct signal pathways.
The B36-2 antibody recognizes an ~36 kDa band corresponding to human TRADD. A recombinant human TRADD protein fragment corresponding to amino acids 14-62 was used as immunogen.
Development References (3)
-
Hsu H, Shu HB, Pan MG, Goeddel DV. TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal transduction pathways. Cell. 1996; 84(2):299-308. (Biology). View Reference
-
Hsu H, Xiong J, Goeddel DV. The TNF receptor 1-associated protein TRADD signals cell death and NF-kappa B activation. Cell. 1995; 81(4):495-504. (Biology). View Reference
-
Varfolomeev EE, Boldin MP, Goncharov TM, Wallach D. A potential mechanism of "cross-talk" between the p55 tumor necrosis factor receptor and Fas/APO1: proteins binding to the death domains of the two receptors also bind to each other. J Exp Med. 1996; 183(3):1271-1275. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.