-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD OMICS-Guard™ Sample Preservation Buffer
- BD® AbSeq Assay
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- BD OMICS-Guard™ CRYO Preservation Buffer
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Thought Leadership
- News
- Blogs
-
Scientific Publications
-
Events
- Expanding PARADIGM to Infectious Disease Modeling: HIV & Tuberculosis
- CYTO 2023: Advancing the World of Cytometry
- Advances in Immune Monitoring Series
- Validating Flow Cytometry Assays for Cell Therapy
- Enhancing Cell Analysis with a New Set of Eyes
- BD Biosciences at International Clinical Cytometry Society 2025
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

HIV
Human immunodeficiency virus (HIV) targets the immune system, and if untreated, causes acquired immune deficiency syndrome (AIDS). Around the world, 38 million individuals are currently living with HIV. Of those, 1.7 million people were newly infected in 2019 and 690,000 have died of AIDS-related illnesses.1 Two main types of HIV exist—HIV-1 and HIV-2. They are very similar structurally and both ultimately lead to AIDS. Globally, HIV-1 is more widespread while HIV-2 is restricted to Western and Central Africa. HIV-2 is less virulent with a longer progression into AIDS but induces diseases of the central nervous system more frequently.
What are the stages of HIV infection?
Assuming no treatment is taken, there are three phases in the progression of HIV infection:2
- Acute — usually occurs about 3 weeks post-exposure. During this phase, the virus replicates rapidly in CD4 T-cells, which is indicated by high viral load and high rate of CD4 T-cell destruction. This is when the host is the most infectious.
- Chronic — also known as clinical latency or asymptomatic HIV infection, the chronic phase is the second stage of infection. Viral replication is low and without treatment, those infected will progress to AIDS within 10+ years.
- AIDS — at this point, the virus has tremendously damaged the immune system and the host can no longer fight opportunistic infections.
Definition of advanced HIV disease
The World Health Organization (WHO) defines four clinical stages of HIV disease—stage 1 through 4, based on the severity of symptoms and coinfections and progression to AIDS.
For adults, adolescents and children older than five years, advanced HIV disease is defined as having a CD4 T-cell count <200 cells/mm3 or WHO stage 3 or 4 event. All children younger than 5 years old with HIV are considered as having advanced HIV disease.3
How is HIV diagnosed?
Antibody tests are the most widely used diagnostic tests to detect HIV infection. It can take about 28 days post-exposure (window period) for HIV-specific antibodies to be detected. Nucleic acid tests (NAT) help determine the viral load in patients. The 2015 World Health Organization (WHO) “Clinical Guidelines: HIV Diagnosis” provides diagnostic test recommendations for different patient groups.4
Biology of HIV infection
HIV is a retrovirus with a genome of 9.8 kilobases coding for a very small number of proteins and with a high mutation rate. The lipid envelope surrounding the core is derived from host cells and is studded with glycoproteins, which are of paramount importance during infection and for eliciting immunogenicity. The envelope protein gp120 binds to CD4 to fuse with T-cells and macrophages. Upon entry into the host cell, the viral RNA is reverse transcribed to DNA, which then integrates into the host genome and gets replicated using the hijacked host machinery. This results in the activation of immune responses instantly, resulting ultimately in depletion of the CD4+ T-cell population through various mechanisms.5
HIV-mediated CD4+ T-cell depletion
HIV-mediated CD4+ T-cell depletion is believed to occur through several stages—(i) enhanced T-cell production upon infection, (ii) accelerated destruction of T-cells through immune responses, (iii) accelerated T-cell production as a response to T-cell depletion through cytokine signaling in the lymph nodes, (iv) accelerated viral replication resulting in the destruction of progenitor cells in the bone marrow, thymus and peripheral lymphoid systems.6
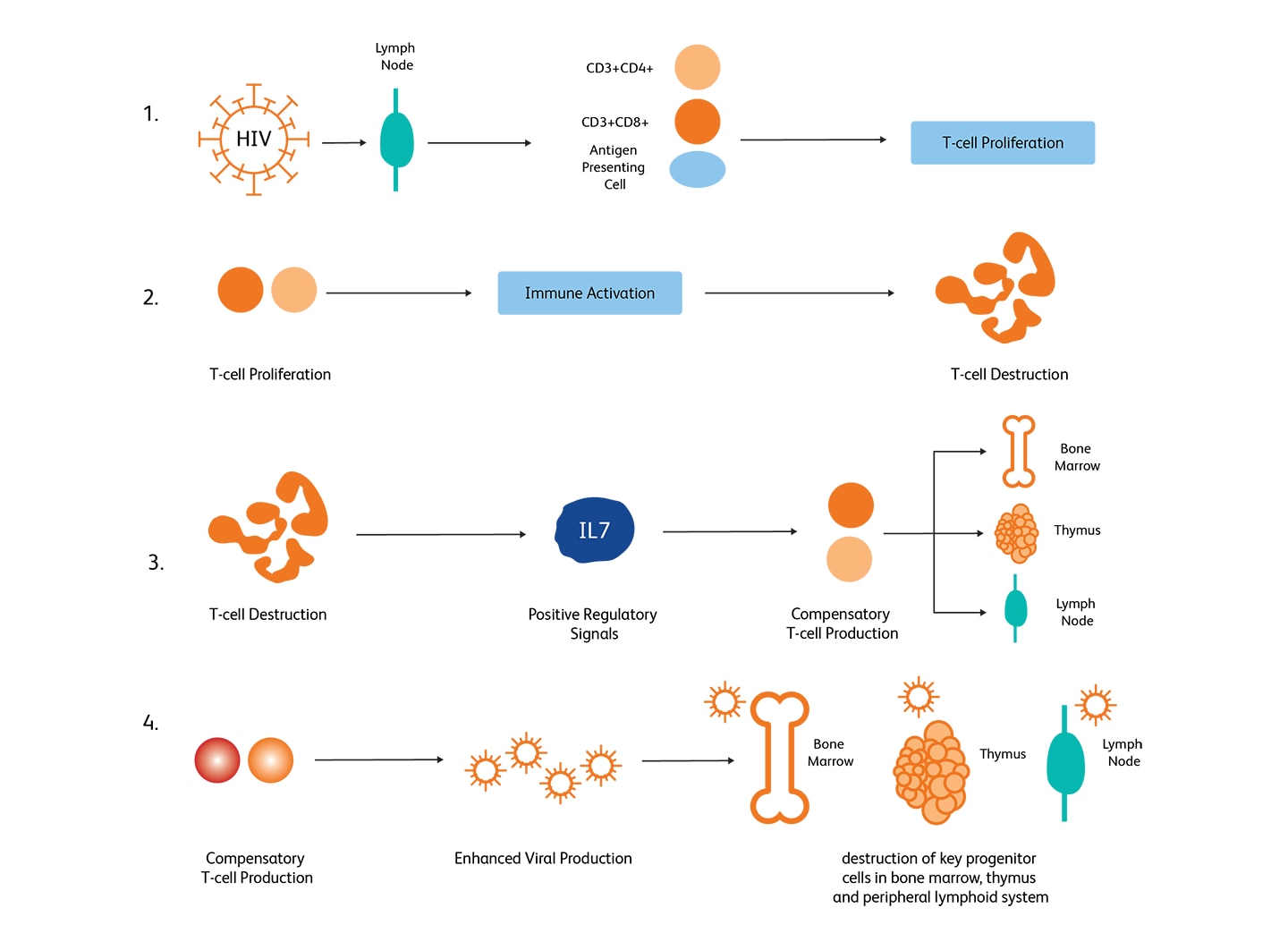
Mechanisms of HIV-mediated CD4+ T-cell depletion. (Image adapted from McCune, 2001).6
References
- World Health Organization. HIV/AIDS Fact Sheet. https://www.who.int/news-room/fact-sheets/detail/hiv-aids. Published July 6, 2020. Accessed September 30, 2020.
- Naif HM. Pathogenesis of HIV Infection. Infect Dis Rep. 2013;5(Suppl 1):e6. Published 2013 Jun 6. doi:10.4081/idr.2013.s1.e6
- World Health Organization. Guidelines for managing advanced HIV disease and rapid initiation of antiretroviral therapy. https://www.who.int/hiv/pub/guidelines/advanced-HIV-disease/en/. Published July 2017. Accessed September 30, 2020.
- World Health Organization. Clinical Guidelines: HIV Diagnosis. Chapter 2. https://www.who.int/hiv/pub/arv/chapter2.pdf?ua=1 Accessed October 4,2020.
- Cowley S. The biology of HIV infection. Lepr Rev. 2001;72(2):212-220. doi: 10.5935/0305-7518.20010028
- McCune JM. The dynamics of CD4+ T-cell depletion in HIV disease. Nature. 2001;410(6831):974-979. doi: 10.1038/35073648
How is HIV monitored?
Viral load monitoring and CD4 T-cell count are routinely used for HIV monitoring. CD4 T-cell count measurements are critical for gaining insights into HIV disease progression, for making clinical decisions on whether to initiate antiviral therapy (ART) and for monitoring the responses to ART.1,2
Determination of percent and absolute number of CD4+ T-lymphocytes using flow cytometry
Flow cytometry is a standard technique used for determining CD4 counts as it provides accuracy, precision and reproducibility. Flow cytometry also provides high-throughput capabilities. Both percentage CD4+ T-lymphocytes among lymphocytes and absolute lymphocyte counts can be determined using flow cytometry. Dual-platform approaches for determining CD4 counts use a flow cytometer for generating percentage CD4+ T-lymphocytes and a hematological analyzer for enumerating absolute lymphocyte counts. Single-platform approaches determine CD4 counts without using a hematological analyzer by either directly counting the number of CD4+ T-lymphocytes in a given volume of blood by CD45 gating3 or by adding microbeads to a known volume of CD4-stained blood samples.4 Determining percentages or counts of CD3+CD4+ lymphocytes can be useful in monitoring HIV-infected individuals.5 Individuals with HIV typically exhibit a steady decrease of CD3+CD4+ lymphocyte counts as the infection progresses.6 At the final stages of infection, CD4 counts go below 200 CD4/µL3, resulting in severe immunodeficiency. 7
Using adoptive transfer of costimulated CD4+ T-cells for reconstituting CD4+ helper T-cell activity has been demonstrated to be a possible method to augment natural immunity to HIV-1 infection.8 HIV-mediated immunosuppression could also result in patients becoming more prone to frequent and severe infections. Opportunistic infections leading to diseases such as Pneumocystis pneumonia (PCP), toxoplasma encephalitis, cytomegalovirus (CMV) infections and tuberculosis are hallmarks of AIDS.9
Role of CD4 cell count testing in identifying and managing people with advanced HIV disease
The 2016 World Health Organization (WHO) “Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection” recommend starting ART regardless of CD4 T-cell count and that the use of CD4 T-cell count for ART response monitoring can be stopped in settings where routine viral load monitoring is available and people are stable on ART.10 However, CD4 T-cell count testing at baseline for all people living with HIV remains important.11 Relying on clinical staging alone risks missing substantial numbers of people living with HIV with severe immune suppression.
BD Biosciences offers various clinical CD4 testing solutions to help manage HIV patients.
The solutions range from a small portable cytometry imaging and absorbance spectrometer system to a fully automated clinical flow cytometer.
The BD FACSLyric™ Flow Cytometer is a clinical cell analyzer that offers high sensitivity, automation and standardization.
BD Multitest™ Reagents, formulated to be used with BD Trucount™ Tubes, provide absolute counting capability.
BD FACSuite™ Clinical Application, with predefined templates for 4-color BD Multitest™ Reagent and 6-color TBNK analysis, provides reproducible and consistent results.
Each reagent includes a cocktail of multiple fluorescently labeled monoclonal antibodies, premixed at the appropriate titer to ensure quality staining.
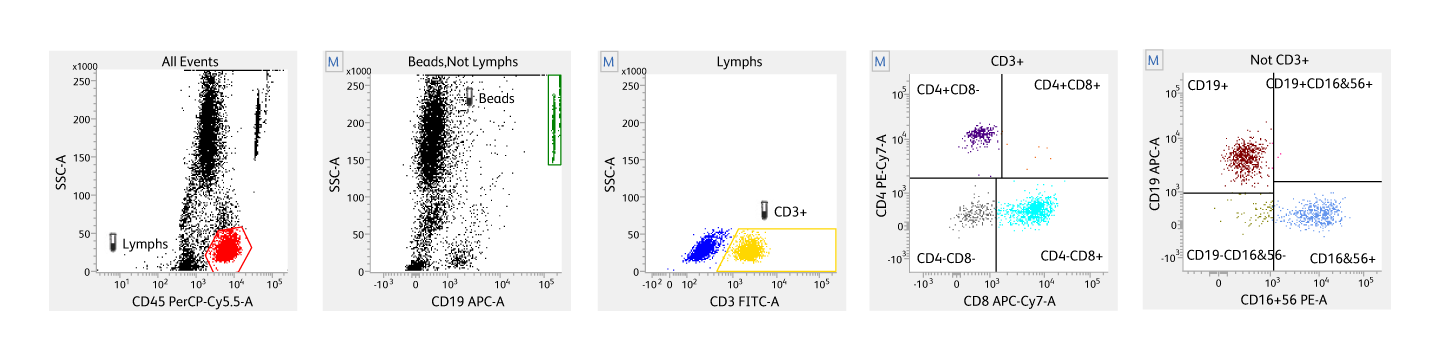
A streamlined methodology for CD4 cell analysis using the BD FACSLyric™ Flow Cytometry System.
References
- Bouteloup V, Sabin C, Mocroft A, et al. Reference curves for CD4 T-cell count response to combination antiretroviral therapy in HIV-1-infected treatment-naïve patients. HIV Med. 2017;18(1):33-44. doi:10.1111/hiv.12389
- Sabin CA, Lundgren JD. The natural history of HIV infection. Curr Opin HIV AIDS. 2013;8(4):311-317. doi:10.1097/COH.0b013e328361fa66
- Center for Disease Control and Prevention. Guidelines for performing single-platform absolute CD4+ T-cell determinations with CD45 gating for persons infected with human immunodeficiency virus. https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5202a1.htm. Accessed October 4,2020.
- Pattanapanyasat K. Immune status monitoring of HIV/AIDS patients in resource-limited settings: a review with an emphasis on CD4+ T-lymphocyte determination. Asian Pac J Allergy Immunol. 2012;30(1):11-25.
- Giorgi JV, Hultin LE. Lymphocyte subset alterations and immunophenotyping by flow cytometry in HIV disease. Clin Immunol Newslett. 1990;10(4):55-61. doi: 10.1016/0197-1859(90)90024-3
- Landay A, Ohlsson-Wilhelm B, Giorgi JV. Application of flow cytometry to the study of HIV infection. AIDS. 1990;4(6):479-497. doi: 10.1097/00002030-199006000-00001
- Streicher HZ, Reitz MS Jr, Gallo RC. Human immunodeficiency viruses. In Mandell GL, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Diseases. New York: Churchill Livingstone; 2000.
- Levine BL, Bernstein WB, Aronson NE, et al. Adoptive transfer of costimulated CD4(+) T cells induces expansion of peripheral T cells and decreased CCR5 expression in HIV infection. Nat Med. 2002;8(1):47-53. doi: 10.1038/nm0102-47
- National Institutes of Health. Guidelines for the prevention and treatment of opportunistic infections in adults and adolescents with HIV. https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-opportunistic-infection/introduction?view=full Accessed October 4,2020.
- World Health Organization. Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection: Recommendations for a public health approach – second edition. https://www.who.int/hiv/pub/arv/arv-2016/en. Published Jume 2016. Accessed September 23, 2020.
- Ford N, Meintjes G, Vitoria M, Greene G, Chiller T. The evolving role of CD4 cell counts in HIV care. Curr Opin HIV AIDS. 2017;12(2):123-128. doi: 10.1097/COH.0000000000000348
BD FACSLyric™ Flow Cytometers are Class 1 Laser Products.
The BD FACSLyric™ Flow Cytometer is for In Vitro Diagnostic Use with BD FACSuite™ Clinical Application for up to six colors. The BD FACSLyric™ Flow Cytometer is for Research Use Only with BD FACSuite™ Application for up to 12 colors. Not for use in diagnostic or therapeutic procedures.
The BD Multitest™ 6-Color TBNK Reagent with optional BD Trucount™ Tubes is intended for use with the BD FACSLyric™, BD FACSCanto™ II, and BD FACSCanto™ Flow Cytometers to determine the percentages and absolute counts of the following mature human lymphocyte subsets in peripheral whole blood for immunophenotyping:
• T-lymphocytes (CD3+)
• B-lymphocytes (CD19+)
• Natural killer (NK) lymphocytes (CD3–CD16+ and/or CD56+)
• Helper/inducer T-lymphocytes (CD3+CD4+)
• Suppressor/cytotoxic T-lymphocytes (CD3+CD8+)
This reagent is indicated for use in the immunological assessment of normal individuals and patients having, or suspected of having, immune deficiency.
BD Trucount™ Tubes are used for determining absolute counts of leucocytes in blood. BD Trucount™ Tubes are designed for use with in vitro diagnostic products such as BD Tritest™ Reagents and a suitably equipped flow cytometer. BD Trucount™ Tubes can be used with the BD FACS™ Loader.
The information provided herein is not meant to be used, nor should it be used, to diagnose or treat any medical condition. All content, including text, graphics, images and information etc., contained in or available through this literature is for general information purposes only. For diagnosis or treatment of any medical condition, please consult your physician/doctor. Becton Dickinson India Private Limited and or its affiliates, its employees are not liable for any damages/claims to any person in any manner whatsoever. BD-25874