Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




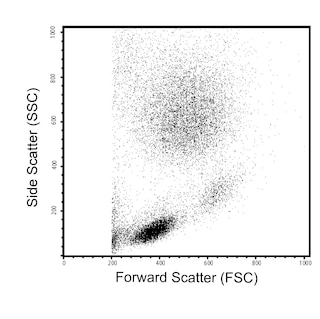
Multiparameter flow cytometric analysis of CD8 expression on human peripheral blood leucocyte populations. Human whole blood was stained with either BD Horizon™ RY586 Mouse IgG1, κ Isotype Control (Cat. No. 568097; Left Plot) or BD Horizon™ RY586 Mouse Anti-Human CD8 antibody (Cat. No. 568114/568115; Right Plot). The erythrocytes were lysed with BD FACS™ Lysing Solution (Cat. No. 349202). The bivariate pseudocolor density plot showing CD8 expression (or Ig Isotype control staining) versus side light scatter signals (SSC) was derived from events with the forward and side light-scatter characteristics of intact leucocytes. Flow cytometric analysis was performed using a BD LSRFortessa™ Cell Analyzer and FlowJo™ software.


BD Horizon™ RY586 Mouse Anti-Human CD8

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
BD® CompBeads can be used as surrogates to assess fluorescence spillover (compensation). When fluorochrome conjugated antibodies are bound to BD® CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cells and BD® CompBeads to ensure that BD® CompBeads are appropriate for your specific cellular application.
Product Notices
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- CF™ is a trademark of Biotium, Inc.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- Human donor specific background has been observed in relation to the presence of anti-polyethylene glycol (PEG) antibodies, developed as a result of certain vaccines containing PEG, including some COVID-19 vaccines. We recommend use of BD Horizon Brilliant™ Stain Buffer in your experiments to help mitigate potential background. For more information visit https://www.bdbiosciences.com/en-us/support/product-notices.
- Species cross-reactivity detected in product development may not have been confirmed on every format and/or application.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
Companion Products




The RPA-T8 monoclonal antibody specifically binds to CD8 alpha (CD8α). CD8α is a type I transmembrane glycoprotein and a member of the immunoglobulin superfamily. CD8α is expressed by the majority of thymocytes, by subpopulations of αβ T cells and γδ T cells and by some NK cells. Cell surface CD8α is expressed either as a disulfide-linked homodimer (CD8αα) or as a heterodimer (CD8αβ) when disulfide-bonded to a CD8 beta chain (CD8β). CD8-positive αβ T cells coexpress both CD8αα homodimers and CD8αβ heterodimers whereas some γδ T cells and NK cells express CD8αα homodimers. CD8 plays important roles in T cell activation and selection. The extracellular IgSF domain of CD8α binds to a non-polymorphic determinant on HLA class I molecules (α3 domain) and enables CD8 to function as a co-receptor with MHC class I-restricted TCR during T cell recognition of antigen. The cytoplasmic domain of CD8α associates with Lck, a Src family protein tyrosine kinase that is involved in intracellular signaling. The RPA-T8 and HIT8a monoclonal antibodies are not cross-blocking. This clone has been reported to react with a subset of peripheral blood lymphocytes, but not monocytes nor granuloyctes, of baboon and both rhesus and cynomolgus macaque monkey. In general, a higher frequency of CD8+ and CD4+CD8+ lymphocytes are observed in non-human primates compared to normal human donors.
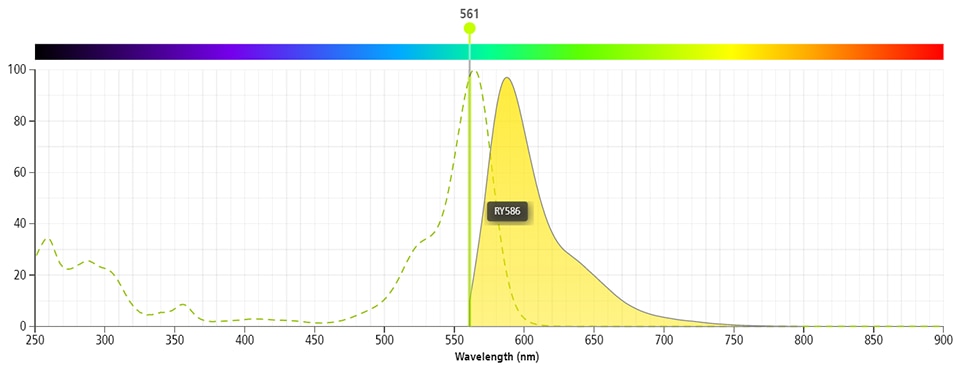
The BD Horizon RealYellow™ 586 (RY586) Dye is part of the BD family of yellow-green dyes. It is a small organic fluorochrome with an excitation maximum (Ex Max) at 565-nm and an emission maximum (Em Max) at 586-nm. Driven by BD innovation, RY586 can be used on both spectral and conventional cytometers and is designed to be excited by the Yellow-Green laser (561-nm) with minimal excitation by the 488-nm Blue laser. For conventional instruments equipped with a Yellow-Green laser (561-nm), RY586 can be used as an alternative to PE and we recommend using an optical filter centered near 586-nm (eg, a 586/15-nm bandpass filter). For spectral instruments equipped with a Yellow-Green laser (561-nm), it can be used in conjunction with PE. Compared to PE, RY586 is similar in brightness, minimal spillover into Blue detectors, and increased spillover into the 610/20-nm (PE-CF594) detector. Please ensure that your instrument configuration (lasers and optical filters) is appropriate for this dye.
Development References (8)
-
Garbrecht F, Loebel A, Disanto JP, Flomenberg N. Chatacterization of Workshop antiCD8 mAb using human CD8-expressing murine L-cell transfectants. In: Schlossman SF. Stuart F. Schlossman .. et al., ed. Leucocyte typing V : white cell differentiation antigens : proceedings of the fifth international workshop and conference held in Boston, USA, 3-7 November, 1993. Oxford: Oxford University Press; 1995:354-356.
-
Jeong SH, Qiao M, Nascimbeni M, et al. Immunization with hepatitis C virus-like particles induces humoral and cellular immune responses in nonhuman primates. J Virol. 2004; 78(13):6995-7003. (Clone-specific: Flow cytometry). View Reference
-
Kersh EN, Kersh GJ, Allen PM. Partially phosphorylated T cell receptor zeta molecules can inhibit T cell activation. J Exp Med. 1999; 190(11):1627-1636. (Clone-specific: Flow cytometry). View Reference
-
Moebius U. Cluster report: CD8. In: Knapp W. W. Knapp .. et al., ed. Leucocyte typing IV : white cell differentiation antigens. Oxford New York: Oxford University Press; 1989:342-343.
-
Qiu X, Audet J, Wong G, et al. Successful treatment of ebola virus-infected cynomolgus macaques with monoclonal antibodies.. Sci Transl Med. 2012; 4(138):138ra81. (Clone-specific: Flow cytometry). View Reference
-
Rabin RL, Park MK, Liao F, Swofford R, Stephany D, Farber JM. Chemokine receptor responses on T cells are achieved through regulation of both receptor expression and signaling. J Immunol. 1999; 162(7):3840-3850. (Clone-specific: Flow cytometry). View Reference
-
Sopper S, Stahl-Hennig C, Demuth M, Johnston IC, Dorries R, ter Meulen V. Lymphocyte subsets and expression of differentiation markers in blood and lymphoid organs of rhesus monkeys. Cytometry. 1997; 29(4):351-362. (Clone-specific: Flow cytometry). View Reference
-
Takami T, Saio M, Nakayama T, Tanaka Y-U, Liu Y. T-cell Blind Panel: Immunihistochemical analysis reporyt. In: Kishimoto T. Tadamitsu Kishimoto .. et al., ed. Leucocyte typing VI : white cell differentiation antigens : proceedings of the sixth international workshop and conference held in Kobe, Japan, 10-14 November 1996. New York: Garland Pub.; 1997:88-92.
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.