-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® OMICS-One Immune Profiler Protein Panel
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
Thought Leadership
- News
- Blogs
-
Scientific Publications
-
Events
- Expanding PARADIGM to Infectious Disease Modeling: HIV & Tuberculosis
- CYTO 2023: Advancing the World of Cytometry
- Advances in Immune Monitoring Series
- Validating Flow Cytometry Assays for Cell Therapy
- Enhancing Cell Analysis with a New Set of Eyes
- BD Biosciences at International Clinical Cytometry Society 2025
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current location site or be switched to your location?

Quality and Reproducibility
Rigorous standards to support reliable results.
Overview
Advance your research with confidence. Our trusted brand of BD flow cytometry instruments and reagents have been used by researchers for over 45 years and have enabled deep scientific discovery in multiple research fields in over 200 countries worldwide. We understand the value of consistency, and that’s reflected in the rigorous testing and quality control standards we place on our products. In particular, we recognize the importance of reagent lot-to-lot consistency; thus, over time is critical to you achieving reproducible results. For this reason, we continue to strive for the highest quality measures to help meet the data reproducibility needs of your research.
Download the BD reagents lot-to-lot consistency brochure
APPLICATIONS
BD Biosciences identifies key targets of interest in scientific research and develops its own specific antibodies but also collaborates with top research scientists around the world to license their antibodies.
We then transform these antibodies into flow cytometry reagents by conjugating them to a broad portfolio of high-performing dyes, including our vastly popular portfolio of BD Horizon Brilliant™ Dyes.
A world-class team of research scientists helps ensure that these reagents work reliably and consistently for flow cytometry applications.
The specificity is confirmed using multiple methodologies that may include a combination of flow cytometry, immunofluorescence, immunohistochemistry or western blot to test staining on a combination of primary cells, cell lines or transfectant models.
All flow cytometry reagents are titrated on the relevant positive or negative cells. To save time and cell samples for researchers, test size reagents are bottled at an optimal concentration with the best signal-to-noise ratio on relevant models during the product development. To ensure consistent performance from lot-to-lot, each reagent is bottled to match the previous lot MFI. You can look up the Certificate of Analysis and the concentration of test-size human reagents from specific lots via the Concentration Lookup page or BD Regulatory Documents
Technical data sheets provide data generated on the relevant primary model at this optimal concentration based on a titration curve. QC data on any lot of reagent can be requested through ResearchApplications@bd.com.

Our dedication to rigorous testing and high-quality control standards means that you can use our reagents in your research with the utmost confidence. All BD reagent facilities, including our California Design Center at San Diego, our manufacturing facility located at Tatabanya, Hungary and our California instrument facility (Manufacturing and Design Centers) at San Jose, are approved and registered to the internationally defined ISO 9001 standard.
Once our research and development (R&D) team completes evaluation of a new product, the developed process is transferred to our manufacturing teams, including Quality Control.
Our manufacturing process adheres to standard operating procedures (SOPs) and guidelines, which are based on ISO requirements, and is strictly followed, helping ensure that reagent builds provide consistent results to help give you assurance of experimental success and confidence in your research.
Quality control testing of newly manufactured lots is performed side-by-side with a previously accepted lot as a control, helping to assure that performance of the new lot is both reliable and consistent.
Our strict adherence to these guidelines helps ensure that different lots of conjugated reagents are performing consistently.
We understand the criticality of lot-to-lot consistency in helping you obtain experimental success and confidence in your research. We strive to ensure different production batches are consistent regardless of type of antigen or fluorochrome through exhaustive testing and strict adherence to quality control standards. Testing with prior batches as reference helps you obtain consistent results with the new batch relative to the previous batches.
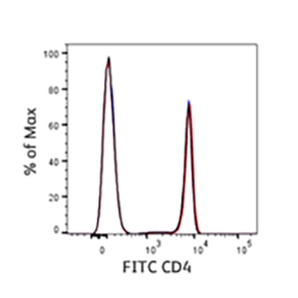
BD research cytometers represent quality, relentless innovation and tireless dedication to service. Our platforms continue to keep pace with the accelerating speed of discovery through ongoing innovation. BD FACSDiva™, BD FACSuite™ and BD FACSChorus™ Software provide one of the most complete and robust feature sets available for flow cytometry.
BD® CS&T Research Beads are specifically designed to take advantage of the automated cytometer setup and performance tracking features of BD FACSDiva™, BD FACSuite™ and BD FACSChorus™ Software to simplify setup with a single-vial reagent solution.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Alexa Fluor is a trademark of Life Technologies Corporation.
CF is a trademark of Biotium, Inc.
The information provided herein is not meant to be used, nor should it be used, to diagnose or treat any medical condition. All content, including text, graphics, images and information etc., contained in or available through this literature is for general information purposes only. For diagnosis or treatment of any medical condition, please consult your physician/doctor. Becton Dickinson India Private Limited and or its affiliates, its employees are not liable for any damages/claims to any person in any manner whatsoever.