Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Store vials at 2°C–8°C. Conjugated forms should not be frozen. Protect from exposure to light. Each reagent is stable until the expiration date shown on the bottle label when stored as directed.
The Anti-Cytokeratin antibody, clone CAM 5.2, is derived from the hybridization of P3/NS-1/1-Ag4-1 mouse myeloma cells with spleen cells from BALB/c mice immunized with the human colorectal carcinoma cell line HT29. The Anti-Cytokeratin antibody has a primary reactivity with human cytokeratin proteins that correspond to Moll’s peptide #7 (cytokeratin 7) and Moll’s peptide #8 (cytokeratin 8), 54 and 53 kilodaltons (kDa), respectively.
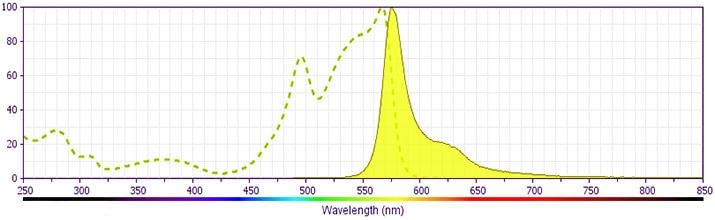
Development References (8)
-
Battifora H. Diagnostic uses of antibodies to keratins: A review and immunohistochemical comparison of seven monoclonal and three polyclonal antibodies. In: Fenoglio-Preiser C, Wolff M, Rilke F, ed. Progress in Surgical Pathology. 1988:1-15.
-
Centers for Disease Control. Update: universal precautions for prevention of transmission of human immunodeficiency virus, hepatitis B virus, and other bloodborne pathogens in healthcare settings. MMWR. 1988; 37:377-388. (Biology).
-
Clinical and Laboratory Standards Institute. 2005. (Biology).
-
Cooper D, Schermer A, Sun TT. Biology of disease classification of human epithelia and their neoplasms using monoclonal antibodies to keratins: strategies, applications and limitations. Lab Invest. 1985; 52:243-256. (Biology). View Reference
-
Johnson A, Cavallin-Stahl E, Akerman M. Flow cytometric light chain analysis of peripheral blood lymphocytes in patients with non-Hodgkin's lymphoma. Br J Cancer. 1985; 52(2):159-168. (Biology). View Reference
-
Makin C, Bobrow L, Bodmer W. Monoclonal antibody to cytokeratin for use in routine histopathology. J Clin Pathol. 1984; 37(9):975-983. (Biology). View Reference
-
Moll R, Franke W, Schiller D, Geiger B, Krepler R. The catalog of human cytokeratins: patterns of expression in normal epithelia, tumors, and cultured cells. Cell. 1982; 31:11-24. (Biology). View Reference
-
Smedts F, Ramaekers F, Robben H, et al. Changing patterns of keratin expression during progression of cervical intraepithelial neoplasia. Am J Pathol. 1990; 136(3):657-668. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Although not required, these products are manufactured in accordance with Good Manufacturing Practices.