3D Printing of Human Tissue Models
Interview with Dr. Nico Forraz

We are pleased to present an interview with Dr. Nico Forraz, CEO and Founding Director of CTIBiotech. Dr. Forraz has extensive experience in stem cells, regenerative medicine, and cell manipulation. He was a part of the team, headed by Prof. Colin McGuckin, that created the world’s first artificial hepatic-like liver tissue and neural cells from human umbilical cord blood. At CTIBiotech, the focus of the work of Dr. Forraz and Prof. McGuckin is on cell manipulation, drug testing, biobanking, and preclinical trials development. In this interview, Dr. Forraz shares with us the details of his work on 3D bioprinting of human tissues, his views on the use of flow cytometry, and the future perspectives of his company.
Could you present CTIBiotech in a few words?
CTIBiotech is a biotechnology company with expertise in human tissue bio-assay development. We use tissue engineering and 3D bioprinting to produce human 3D tissue models to support biomedical, pharmaceutical, and dermato-cosmetic development.
Can you tell us about your clients and your activities?
Our company is organised into three business units. One unit is called CTIPharma. I created this company with Prof. Colin McGuckin who is also our President and Chief Scientific Officer, and we have a background in haematology and immuno-oncology. CTIPharma specialises in producing human tissue models for pharmaceutical and healthcare development, with a strong focus on applications in oncology, immune-therapies and regenerative medicine. Our clients include pharmaceutical companies like Eli-Lilly and Sanofi, and biotech companies developing immunotherapies like Transgene, a part of the BioMerieux Group. We also have clients among academic and clinical centres in France and around the world. For example, we work with the centre Léon Bérard in Lyon, which specialises in oncology. Other partners are Marseille Hospital and A-Star Institute in Singapore.
Another business unit is called CTISkin. This specialises in making human skin models. We investigate the efficacy of ingredients, medical devices, or even finished cosmetics or consumer care products on artificial skin produced by 3D bioprinting. Examples of customers are BASF, Beiersdorf, Kose, the no 3 cosmetics company in Japan and Asia, Yves Rocher, and many others around the world. We have many different types of clients like large corporations, small startups, SMEs that develop medical or beauty devices that help with healing and skincare.
CTIPharma and CTISkin work in similar ways as far as technology is concerned. We work with ex-vivo models, in which a piece of tissue is directly worked on to create a bioassay. For example, you could cut a solid tumor into pieces and investigate biological pathways by flow cytometry or microscopy. Another technology we use is in-vitro models. Here we dissociate pieces of tumor or skin and isolate all the different cell types. In tumors, these could be cancer cells, cancer stem cells, cancer-associated fibroblasts, immune cells, and endothelial cells. We sort and characterise them immunophenotypically using flow cytometry. Once we have sorted and separated them, we can ‘amplify’ them in the laboratory. We can investigate one cell type or a do a co-culture, for instance, of cancer cells and cancer-associated fibroblasts, and study the efficacy of drug candidates on such models.
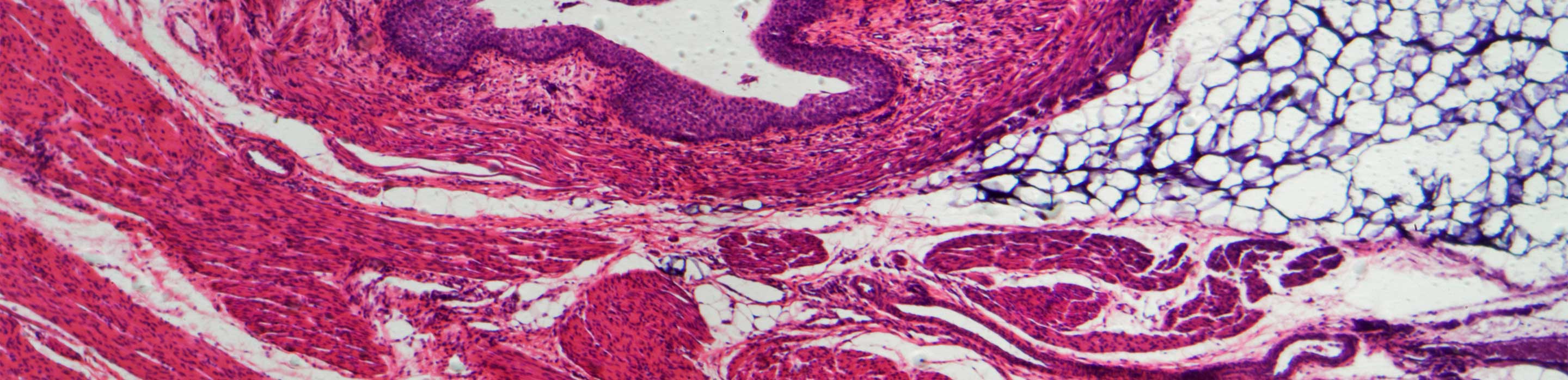
Another set of models that we have are called 3D reconstructed or 3D tissue-engineered models. Earlier the researchers in our lab would do all this manually. In the last five years, we have shifted to the use of 3D bioprinting. In this technique, we use the in-vitro collection of cells to reassemble a 3D structure which would be equivalent to a tumor or a piece of skin. One big advantage of this technology is that you can create a model of any composition by modulating the proportion of the cell types. You can also make your model highly reproducible by starting with one piece of a tumor, expanding it in the laboratory, and then 3D printing hundreds of identical tumors. We can also do the same thing with skin, where we can 3D print skin models to create equivalents of the epidermis using keratinocytes and fibroblasts, and include other cell types like macrophages, that are characterised by flow cytometry.
So we work with three types of models – ex-vivo, in vitro, and 3D printed models.
A third business unit is a production unit called CTIBiosourcing. This unit delivers the models to our customer’s laboratories.
What place does flow cytometry have in your current research and plans for the future?
Flow cytometry has a very strong place in our company. As a preamble, I have been working with Prof. McGuckin since 1996, who in turn, has been working with BD for almost 35 years. BD has been a very strong partner of our academic and clinical lab when we were in academia in the UK and the US. This relationship continued when we came to France eleven years ago to set up CTIBiotech. Flow cytometry is very important in our laboratory to characterise the different cell types, whether solid tumors, skin cells, or hematopoietic stem cells. We have developed many panels to study the cell types and we even publish on this, though we are a company. For instance, we have developed a flow cytometric characteristic analysis panel for mesenchymal stem-cells. The idea here was to help clinical projects to use as little of the sample as possible, to make a phenotypic map of the mesenchymal stem-cell types depending on their origin– bone marrow, fat tissue, or umbilical cord, for example. We can also characterise them according to the cell therapy procedure they would go under.
Another strong application that is growing, and something our customers are very interested in, is the cytometric bead array. The advantage of this is that one can use a very small amount of the cell-culture supernatant to characterise 30+ cytokines in one experiment. In our opinion, it is a very time and cost-effective technology.
We also use flow cytometry to study the cell cycle and have been the first in the world to study sebocytes using flow cytometric technology. These are the cell populations that make up the sebaceous glands that produce sebum, the oily liquid that is seen on healthy skin. Sebocytes are important in cosmetics (for oily or dry skin), dermatological conditions like eczema and psoriasis, where one has a decreased production of sebum, and acne. They are also important in the clinic for burn victims, who lose the capacity to produce sebum, rendering their skin dry and brittle. We are the first group in the world to make a 3D model of the sebaceous gland along with a 2D model of human primary sebocytes. The sebum production by the sebocytes can be characterised with two fluorescent reagents. One is called Nile-red which is specific to lipids, and the other is by Hoechst, which stains the DNA in the cell. With our flow cytometer, we have designed a protocol using which we can quantify the shift in the maturation of sebocytes, which is a unique application of the technique.
What are the pain points in flow cytometry that you would like BD to address?
A good idea would be to continue diversifying the cytometric bead array mix so there is more choice – may be to increase the number of oncology settings, neurotoxicity, or neurogenesis. You could also perhaps support custom designs of the cytometric bead arrays better. Maybe panel design? I know you have developed several tools for that, but it would also be a good idea to do more in-house training. We have a couple of specialist flow cytometrists in-house but we would like to train more people. We can do that internally but my experience with BD is that your team is very good at making intermediate flow cytometrists from the beginners. It would be better for us if you could help us do that in-house instead of sending people to your training centres.
We would also like to see better cell sorting. We have found that cell sorting is not always suitable for very small populations of cells. Presently, we are using immune-magnetic selection. Maybe if there is some innovation in practical, user-friendly cell sorting, we could switch back to it from the immune-magnetic selection.
What exactly is the pain in cell sorting?
To give an example of something I have experienced - I would spend about 24 hours preparing a cell population that is very rare or of interest, only to find that everything is wasted due to a bug in the fluidics of the cell sorter. Maybe flow cytometric cell sorting is good for large populations of cells but if you are looking for a small, very specific, and rare stem-cell subset diluted in a large cell population, then it is not so efficient.
Are you doing your panels yourselves now?
Yes, mostly. But we are always open to suggestions.
And what do you do if you need any help for panels design?
We tend to use the tools available online. We are also close to our local salesperson, for specific requests.
How many colours are you using?
We usually use three to four and can go up to eight easily when we use the mesenchymal stem cell panels. These are limited by the lasers and filters we have.
And would you say you need more?
We don’t need more from a routine perspective. We can already do a lot with eight colors.
Why did you choose BD? Was it because our solutions were answering to your needs?
As you understood from my initial introduction, I have been using BD all of my life. I have also worked on tools made by other companies, but BD is very robust. I like the way the instruments are designed. You have a very good worldwide customer service, and I have experienced that in three countries – France, the US, and the UK. The standardization in terms of technical customer services is excellent, in my opinion. Trust in the technology is key. Also, the laser maintenance, fluidics, and the footprint of the devices have continuously improved in your company. Therefore, we have chosen to stay with BD. Moreover, here in France, the team in Pont-de-Claix is very supportive and helpful, whether in panel design or technology solutions, even in acquiring new machines.
Where does CTIBiotech see itself in the next five years?
Right now we do 60% of our business outside of France. We have a strong presence in Asia, especially Japan. We have two main ambitions. The first is to strengthen our business in North America through partnerships and collaborations. We also want to establish production labs in Japan, the US and Canada to be closer to our existing and potential customers.
There has been quite a lot of research on COVID-19 where people have used flow cytometry to study the response of the immune system to the virus. What is your take on the ongoing research?
It is not something we do, as we are not allowed to work with P3 viruses in our facility. However, we do provide immunized skin models produced by 3D printing to the Pasteur Institute and Ecole Polytechnique in Paris. They work not only on COVID-19 but also on multidrug-resistant Staphylococcus aureus. We collaborate with them by providing models of healthy tissues that can be infected. Another project we are working on is to provide models of skin epidermis with the dermis underneath and nasal or airway mucosa. These models can also be used for such research.
In my opinion, what I find interesting is the cytokine storm. Your technologies can support such research, particularly with the cytometric bead array and cytokine kits which can cover a large range of inflammatory cytokines in a few minutes. It is a very powerful technology to support COVID-19 research.
View our selection of immunoassay reagents.