Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
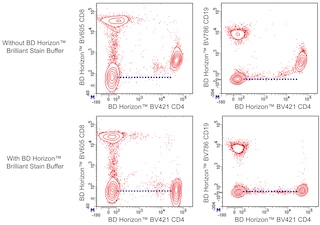
For optimal and reproducible results, BD Horizon Brilliant Stain Buffer should be used anytime two or more BD Horizon Brilliant dyes (including BD OptiBuild Brilliant reagents) are used in the same experiment. Fluorescent dye interactions may cause staining artifacts which may affect data interpretation. The BD Horizon Brilliant Stain Buffer was designed to minimize these interactions. More information can be found in the Technical Data Sheet of the BD Horizon Brilliant Stain Buffer (Cat. No. 563794).
Product Notices
- This antibody was developed for use in flow cytometry.
- The production process underwent stringent testing and validation to assure that it generates a high-quality conjugate with consistent performance and specific binding activity. However, verification testing has not been performed on all conjugate lots.
- Researchers should determine the optimal concentration of this reagent for their individual applications.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- BD Horizon Brilliant Stain Buffer is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,575,303; 8,354,239.
- BD Horizon Brilliant Ultraviolet 395 is covered by one or more of the following US patents: 8,158,444; 8,575,303; 8,354,239.
Companion Products
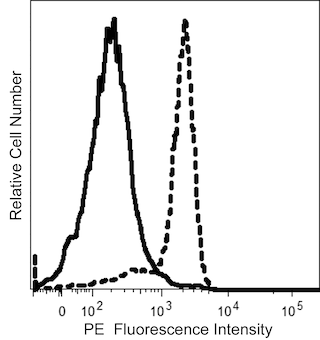


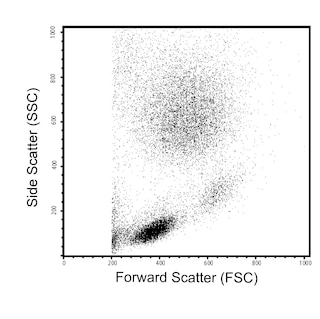

The 8G12 monoclonal antibody specifically recognizes CD34, a 105-120 kDa single-chain type I transmembrane glycoprotein. The 8G12 antibody recognizes an epitope on CD34 distinct from the one recognized by clone My10. CD34 is expressed on immature hematopoietic precursor cells and all hematopoietic colony-forming cells in bone marrow and blood, including unipotent (CFU-GM, BFU-E) and pluripotent progenitors (CFU-GEMM, CFU-Mix, and CFUBlast). The CD34 antigen is a differentiation stage-specific leucocyte antigen. Terminal deoxynucleotidyl transferase-positive B- and T-lymphoid precursors in normal bone marrow are CD34+. The CD34 antigen is present on early myeloid cells that express the CD33 antigen but lack the CD14 and CD15 antigens and on early erythroid cells that express the CD71 antigen and dimly express the CD45 antigen. The CD34 antigen is also found on capillary endothelial cells and approximately 1% of human thymocytes. Normal peripheral blood lymphocytes, monocytes, granulocytes, and platelets do not express CD34. CD34 density is highest on early hematopoietic progenitor cells and decreases as cells mature. The antigen is absent on fully differentiated hematopoietic cells. Uncommitted CD34+ progenitor cells are CD38- and lack lineage-specific antigens such as CD71, CD33, CD10, and CD5, while CD34+ cells that are lineage-committed express the CD38 antigen in high density. Most CD34+ cells reciprocally express either the CD45RO or CD45RA antigens, with the CD45RO+ population being the more primitive. Approximately 60% of acute B-lymphoid leukemias and acute myeloid leukemias (AML) and 1% to 5% of acute T-lymphoid leukemias express CD34. CD34 is not expressed on chronic lymphoid leukemias or lymphomas.
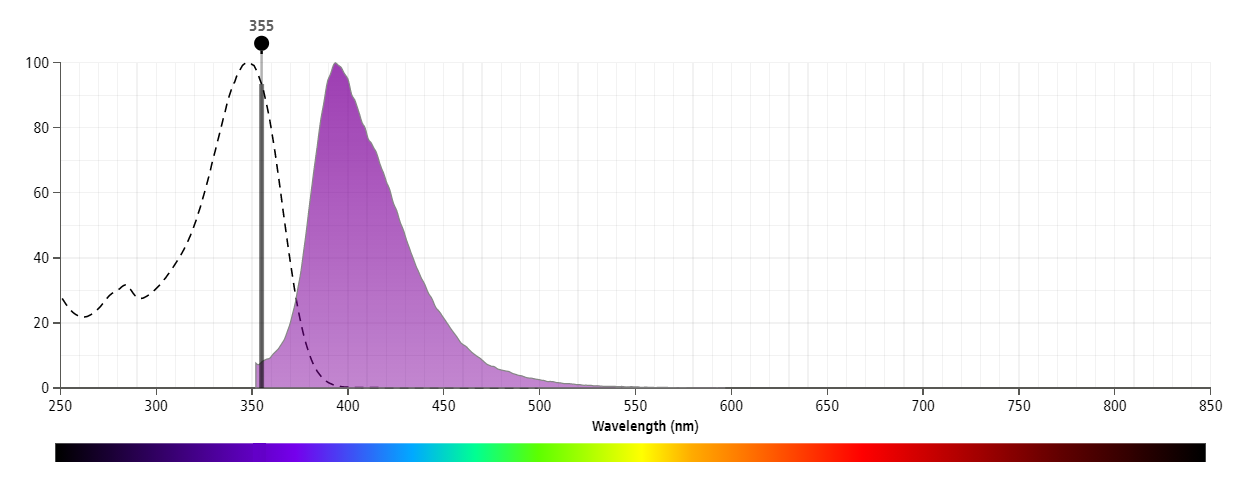
The antibody was conjugated to BD Horizon™ BUV395 which is part of the BD Horizon Brilliant™ Ultraviolet family of dyes. This dye has been exclusively developed by BD Biosciences to have minimal spillover into other detectors, making it an optimal choice for multicolor flow cytometry. With an Ex Max at 348 nm and an Em Max at 395 nm, BD Horizon BUV395 can be excited with a 355 nm laser and detected with a 379/28 filter.
Development References (17)
-
Lansdorp PM, Dougherty GJ, Humphries RK. CD34 epitopes. In: Knapp W. W. Knapp .. et al., ed. Leucocyte typing IV : white cell differentiation antigens. Oxford New York: Oxford University Press; 1989:826-827.
-
Andrews RG, Singer JW, Bernstein ID. Precursors of colony-forming cells in humans can be distinguished from colony-forming cells by expression of the CD33 and CD34 antigens and light scatter properties.. J Exp Med. 1989; 169(5):1721-31. (Biology). View Reference
-
Brocklebank AM, Sparrow RL. Enumeration of CD34+ cells in cord blood: a variation on a single-platform flow cytometric method based on the ISHAGE gating strategy.. Cytometry. 2001; 46(4):254-61. (Biology). View Reference
-
Gore SD, Kastan MB, Civin CI. Normal human bone marrow precursors that express terminal deoxynucleotidyl transferase include T-cell precursors and possible lymphoid stem cells.. Blood. 1991; 77(8):1681-90. (Biology). View Reference
-
Greaves MF, Titley I, Colman SM, et al. CD34 cluster workshop report. In: Schlossman SF. Stuart F. Schlossman .. et al., ed. Leucocyte typing V : white cell differentiation antigens : proceedings of the fifth international workshop and conference held in Boston, USA, 3-7 November, 1993. Oxford: Oxford University Press; 1995:840-846.
-
Hurwitz CA, Loken MR, Graham ML, et al. Asynchronous antigen expression in B lineage acute lymphoblastic leukemia.. Blood. 1988; 72(1):299-307. (Biology). View Reference
-
Kurtzberg J, Denning SM, Nycum LM, Singer KH, Haynes BF. Immature human thymocytes can be driven to differentiate into nonlymphoid lineages by cytokines from thymic epithelial cells.. Proc Natl Acad Sci USA. 1989; 86(19):7575-9. (Biology). View Reference
-
Lansdorp PM, Sutherland HJ, Eaves CJ. Selective expression of CD45 isoforms on functional subpopulations of CD34+ hemopoietic cells from human bone marrow.. J Exp Med. 1990; 172(1):363-6. (Clone-specific: Flow cytometry). View Reference
-
Lanza F, Moretti S, Papa S, Malavasi F, Castoldi G. Report on the Fifth International Workshop on Human Leukocyte Differentiation Antigens, Boston, November 3-7, 1993.. Haematologica. 79(4):374-86. (Clone-specific: Flow cytometry). View Reference
-
Leary AG, Strauss LC, Civin CI, Ogawa M. Disparate differentiation in hemopoietic colonies derived from human paired progenitors.. Blood. 1985; 66(2):327-32. (Biology). View Reference
-
Loken MR, Shah VO, Dattilio KL, Civin CI. Flow cytometric analysis of human bone marrow. II. Normal B lymphocyte development. Blood. 1987; 70(5):1316-1324. (Biology). View Reference
-
Loken MR, Shah VO, Dattilio KL, Civin CI. Flow cytometric analysis of human bone marrow: I. Normal erythroid development.. Blood. 1987; 69(1):255-63. (Biology). View Reference
-
Peschel C, Köller U. Cluster report: CD34. In: Knapp W. W. Knapp .. et al., ed. Leucocyte typing IV : white cell differentiation antigens. Oxford New York: Oxford University Press; 1989:817-818. View Reference
-
Ryan D, Kossover S, Mitchell S, Frantz C, Hennessy L, Cohen H. Subpopulations of common acute lymphoblastic leukemia antigen-positive lymphoid cells in normal bone marrow identified by hematopoietic differentiation antigens.. Blood. 1986; 68(2):417-25. (Biology). View Reference
-
Siena S, Bregni M, Brando B, et al. Flow cytometry for clinical estimation of circulating hematopoietic progenitors for autologous transplantation in cancer patients.. Blood. 1991; 77(2):400-9. (Clone-specific: Flow cytometry). View Reference
-
Terstappen LW, Huang S, Safford M, Lansdorp PM, Loken MR. Sequential generations of hematopoietic colonies derived from single nonlineage-committed CD34+CD38- progenitor cells. Blood. 1991; 77(6):1218-1227. (Clone-specific: Flow cytometry, Fluorescence activated cell sorting). View Reference
-
Terstappen LW, Safford M, Könemann S, et al. Flow cytometric characterization of acute myeloid leukemia. Part II. Phenotypic heterogeneity at diagnosis.. Leukemia. 1992; 6(1):70-80. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.