-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- Germany (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from United States.
Would you like to stay on the current country site or be switched to your country?
BD Pharmingen™ Purified Rat Anti-Mouse CD86
Clone GL1 (RUO)

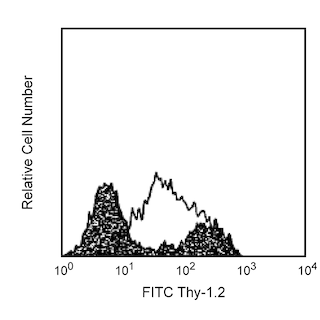
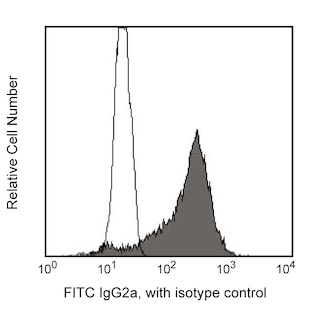
Upregulation of membrane CD86 (B7-2) on activated B lymphocytes. Freshly isolated (left panel) or 72-hour LPS-stimulated BALB/c splenocytes (right panel) were pretreated with Mouse BD Fc Block™ purified anti-mouse CD16/CD32 mAb 2.4G2 (Cat. No. 553141/553142) and either unstained (shaded histograms) or stained with purified GL1 mAb (open histograms) followed by FITC-conjugated anti-rat IgG2a mAb RG7/1.30 (Cat. No. 553896, shaded and open histograms). Flow cytometry was performed on a BD FACScan™ Flow Cytometry System. Resting lymphocytes (left panel) or activated blasts (right panel) were selected according to light-scatter profile.


Upregulation of membrane CD86 (B7-2) on activated B lymphocytes. Freshly isolated (left panel) or 72-hour LPS-stimulated BALB/c splenocytes (right panel) were pretreated with Mouse BD Fc Block™ purified anti-mouse CD16/CD32 mAb 2.4G2 (Cat. No. 553141/553142) and either unstained (shaded histograms) or stained with purified GL1 mAb (open histograms) followed by FITC-conjugated anti-rat IgG2a mAb RG7/1.30 (Cat. No. 553896, shaded and open histograms). Flow cytometry was performed on a BD FACScan™ Flow Cytometry System. Resting lymphocytes (left panel) or activated blasts (right panel) were selected according to light-scatter profile.

Upregulation of membrane CD86 (B7-2) on activated B lymphocytes. Freshly isolated (left panel) or 72-hour LPS-stimulated BALB/c splenocytes (right panel) were pretreated with Mouse BD Fc Block™ purified anti-mouse CD16/CD32 mAb 2.4G2 (Cat. No. 553141/553142) and either unstained (shaded histograms) or stained with purified GL1 mAb (open histograms) followed by FITC-conjugated anti-rat IgG2a mAb RG7/1.30 (Cat. No. 553896, shaded and open histograms). Flow cytometry was performed on a BD FACScan™ Flow Cytometry System. Resting lymphocytes (left panel) or activated blasts (right panel) were selected according to light-scatter profile.


BD Pharmingen™ Purified Rat Anti-Mouse CD86

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Mouse BD Fc Block™ purified anti-CD16/CD32 mAb 2.4G2 (Cat. No. 553141/553142) may help to reduce non-specific binding of GL1 antibody to cells bearing Fcγ-receptors. When performing immunofluorescent staining in the presence of Mouse BD Fc Block™, a second step antibody which does not cross-react with the 2.4G2 antibody (Rat IgG2b, κ) must be used. We have found that FITC-conjugated mouse anti-rat IgG2a (clone RG7/1.30, Cat. No. 553896) works well for this purpose.
Immunohistochemistry: For IHC, we recommend the use of purified GL1 mAb in our special formulation for immunohistochemistry, Cat. No. 550542).
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Sodium azide is a reversible inhibitor of oxidative metabolism; therefore, antibody preparations containing this preservative agent must not be used in cell cultures nor injected into animals. Sodium azide may be removed by washing stained cells or plate-bound antibody or dialyzing soluble antibody in sodium azide-free buffer. Since endotoxin may also affect the results of functional studies, we recommend the NA/LE (No Azide/Low Endotoxin) antibody format, if available, for in vitro and in vivo use.
Data Sheets
Companion Products
The GL1 antibody specifically recognizes the B7-2 (CD86) costimulatory molecule expressed on a broad spectrum of leukocytes, including B lymphocytes, T lymphocytes, thioglycollate-induced peritoneal macrophages, dendritic cells and astrocytes. CD86 is expressed at low levels by freshly explanted peripheral B and T cells, and its expression is substantially increased by a variety of T cell- and B cell-specific stimuli with a peak expression after 18-42 hours of culture. In contrast to most naive CD4+ T cells, memory CD4+ T cells express B7-2, both at the mRNA and protein level. CD86, a ligand for CD28 and CD152 (CTLA-4), is one of the accessory molecules that plays an important role in T cell-B cell costimulatory interactions. It has been shown to be involved in immunoglobulin class-switching and triggering of mouse NK cell-mediated cytotoxicity. CD80 (B7-1) is an alternate ligand for CD28 and CD152 (CTLA-4). GL1 antibody reportedly blocks MLR and stimulation of T cells by natural antigen-presenting cells. In addition, a mixture of anti-B7-1 and anti B7-2 (GL1) mAbs reportedly inhibits the in vitro interaction of CTLA-4 with its ligand and the in vivo priming of cytotoxic T lymphocytes.
This antibody is routinely tested by flow cytometric analysis. Other applications were tested at BD Biosciences Pharmingen during antibody development only or reported in the literature.
Development References (16)
-
Bluestone JA. New perspectives of CD28-B7-mediated T cell costimulation. Immunity. 1995; 2(6):555-559. (Biology). View Reference
-
Borriello F, Sethna MP, Boyd SD, et al. B7-1 and B7-2 have overlapping, critical roles in immunoglobulin class switching and germinal center formation. Immunity. 1997; 6(3):303-313. (Biology). View Reference
-
Freeman GJ, Borriello F, Hodes RJ, et al. Uncovering of functional alternative CTLA-4 counter-receptor in B7-deficient mice. Science. 1993; 262(5135):907-909. (Biology). View Reference
-
Hakamada-Taguchi R, Kato T, Ushijima H, Murakami M, Uede T, Nariuchi H. Expression and co-stimulatory function of B7-2 on murine CD4+ T cells. Eur J Immunol. 1998; 28(3):865-873. (Biology). View Reference
-
Hathcock KS, Laszlo G, Dickler HB, Bradshaw J, Linsley P, Hodes RJ. Identification of an alternative CTLA-4 ligand costimulatory for T cell activation. Science. 1993; 262(5135):905-907. (Immunogen: Blocking, Immunoprecipitation). View Reference
-
Hathcock KS, Laszlo G, Pucillo C, Linsley P, Hodes RJ. Comparative analysis of B7-1 and B7-2 costimulatory ligands: expression and function. J Exp Med. 1994; 180(2):631-640. (Clone-specific: Blocking). View Reference
-
Herold KC, Vezys V, Koons A, Lenschow D, Thompson C, Bluestone JA. CD28/B7 costimulation regulates autoimmune diabetes induced with multiple low doses of streptozotocin. J Immunol. 1997; 158(2):984-991. (Clone-specific: Blocking). View Reference
-
Inaba K, Witmer-Pack M, Inaba M, et al. The tissue distribution of the B7-2 costimulator in mice: abundant expression on dendritic cells in situ and during maturation in vitro. J Exp Med. 1994; 180(5):1849-1860. (Clone-specific: Blocking, Immunohistochemistry). View Reference
-
Krummel MF, Allison JP. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med. 1995; 182(2):459-465. (Clone-specific: Blocking). View Reference
-
Larsen CP, Ritchie SC, Hendrix R, et al. Regulation of immunostimulatory function and costimulatory molecule (B7-1 and B7-2) expression on murine dendritic cells. J Immunol. 1994; 152(11):5208-5219. (Biology). View Reference
-
Lenschow DJ, Su GH, Zuckerman LA, et al. Expression and functional significance of an additional ligand for CTLA-4. Proc Natl Acad Sci U S A. 1993; 90(23):11054-11058. (Biology). View Reference
-
Liu Y, Wenger RH, Zhao M, Nielsen PJ. Distinct costimulatory molecules are required for the induction of effector and memory cytotoxic T lymphocytes. J Exp Med. 1997; 185(2):251-262. (Clone-specific: Blocking). View Reference
-
Martin-Fontecha A, Assarsson E, Carbone E, Karre K, Ljunggren HG. Triggering of murine NK cells by CD40 and CD86 (B7-2). J Immunol. 1999; 162(10):5910-5916. (Biology). View Reference
-
McAdam AJ, Schweitzer AN, Sharpe AH. The role of B7 co-stimulation in activation and differentiation of CD4+ and CD8+ T cells. Immunol Rev. 1998; 165:231-247. (Biology). View Reference
-
Nikcevich KM, Gordon KB, Tan L, et al. IFN-gamma-activated primary murine astrocytes express B7 costimulatory molecules and prime naive antigen-specific T cells. J Immunol. 1997; 158(2):614-621. (Biology). View Reference
-
Turley SJ, Inaba K, Garrett WS, et al. Transport of peptide-MHC class II complexes in developing dendritic cells. Science. 2000; 288(5465):522-527. (Clone-specific: Electron microscopy, Immunofluorescence). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.