Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/551323Image1.png)
![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/24651C_551323_image1.png)
![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/551323Image1.png)

![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/24651C_551323_image1.png)
Schematic representation of the MHC class I:Ig dimeric protein.
![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/551323Image1.png)
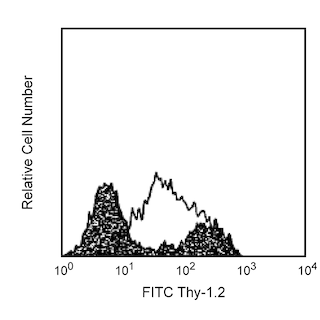
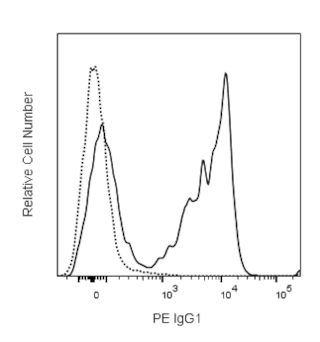
Flow cytometric analysis of a T-cell hybridoma using DimerX I H-2Db:Ig. Mouse DimerX I H-2Db:Ig was incubated with a 160-molar excess of a specific peptide MOG 44-54 (FSRVVHLYRNG, Panel A) or irrelevant peptide Influenza A34 NP 366-374 (ASNENMETM, Panel B) at 37°C overnight. Peptide-loaded H-2Db:Ig was then used for the immunofluorescent staining of MOG TH10 hybridoma cells, along with FITC conjugated anti-mouse CD3e mAb 145-2C11 (Cat. no. 553061/553062) followed by PE-conjugated anti-mouse IgG1 mAb A85-1 (Cat. no. 550083). Flow cytometry was performed on a BD FACSCalibur™ flow cytometry system.

![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/24651C_551323_image1.png)
BD™ DimerX DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig
![DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig](/content/dam/bdb/products/global/reagents/flow-cytometry-reagents/research-reagents/single-color-antibodies-ruo/551xxx/5513xx/551323_base/551323Image1.png)
BD™ DimerX DimerX I: Recombinant Soluble Dimeric Mouse H-2D[b]:Ig

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
This H-2D[b]:Ig fusion protein has been tested by immunofluorescent staining (≤ 4 µg H-2D[b]:Ig/million cells) (see Figure 2) and flow cytometric analysis of antigen-specific T cells to assure specificity and reactivity. It is necessary to load the H-2D[b] portions of the dimeric protein with a relevant peptide of interest prior to immunofluorescent staining of T cells. H-2D[b]:Ig complexes are effectively loaded by incubation with excess relevant (specific) or irrelevant (control) peptides (see Protocol 1). Peptide-loaded H-2D[b]:Ig may be used for immunofluorescent staining (see Protocol 2). Since applications vary, each investigator must determine dilutions appropriate for individual use.
Protocol 1: Peptide Loading of H-2D[b]:Ig Dimeric Protein
Several peptide-loading protocols have been described. The method used at BD Biosciences Pharmingen involves passive loading of excess peptide in solution with H-2D[b]:Ig protein. We have found that passive loading works particularly well in the case of high-affinity peptides. For lower-affinity peptides, an increase in the molar ratio of peptide to H-2D[b]:Ig may improve loading, as determined by flow cytometric analysis. It is suggested that for each peptide, parameters such as the dose of H-2D[b]:Ig per million cells, molar ratio of peptide to H-2D[b]:Ig, and peptide loading time be determined empirically by the investigator. Parameters for peptide binding to H-2D[b] have been reported, including those peptides routinely used at BD Biosciences Pharmingen for evaluation of H-2D[b]:Ig. While this DimerX product contains β2 Microglobulin, for investigators requiring excess recombinant Human β2 Microglobulin, we recommend BD Biosciences Cat. no. 551089.
Peptide preparation and loading:
1. The molecular weight (MW) of a peptide of interest will need to be determined. A peptide's MW can be estimated by multiplying its number
(n) of amino acids (AA) by 130 daltons (d) per amino acid:
MW of peptide (d) = n (AA) x 130 (d/AA)
2. A stock of peptide may be prepared at 20 mg/ml in DMSO. Dilute the peptide solution to 2 mg/ml in sterile DPBS, pH 7.2 for use in the
H-2D[b]:Ig loading protocol.
3. Mix H-2D[b]:Ig protein with specific or control peptide at 40, 160, or 640 molar (M) excess.
The following calculation, using an 8 amino acid peptide (8mer) as an example, may be used:
Dp = Molecular Weight of peptide: eg, 8 amino acids x 130 = 1,040 daltons.
DDb = Molecular Weight of H-2Db:Ig = 250,000 daltons.
R = desired excess molar ratio, eg, 160.
Mp = micrograms (µg) peptide of interest.
MDb = micrograms (µg) H-2D b:Ig in the reaction. A typical amount of peptide-loaded H-2Db:Ig to use for flow cytometry staining is 0.25
to 4 µg/million cells (test).
Mp = MDb x R x Dp÷DDb= 4 µg x 160 x 1,040 d÷250,000 d = 2.66 µg
Therefore, one would add 2.66 µg of peptide and 4 µg of H-2D[b] :Ig in solution for the optimal peptide loading of H-2D[b]:Ig.
Mix peptide and H-2D[b]:Ig together in PBS, pH 7.2, incubate at 37°C overnight. The peptide-loaded H-2D[b]:Ig can be stored at
4°C for up to 1 week.
Protocol 2: Immunofluorescent Staining Protocol
1. Prepare peptide-loaded H-2D[b] protein staining cocktail by mixing 0.25 -4 µg of peptide-loaded H-2D[b] protein/test with 0.25 -4 µg of
PE-conjugated A85-1 mAb (anti-mouse IgG1, Cat. no. 550083)/test at a ratio of 1:1 or 1:2 of dimer:A85-1 mAb. Incubate the mixture
for 60 minutes at RT, protect from exposure to light.
2. Add 0.25 - 4 µg of purified mouse IgG1 isotype control mAb A111-3 (Cat. no. 553485)/test to the staining cocktail (see Step 1 above).
Incubate the staining cocktail for 30 minutes at RT, protect from exposure to light.
3. Resuspend mouse cells in FACS staining buffer [eg, DPBS, 1% FCS, 0.09% NaN3 or BD Pharmingen™ Stain Buffer (FBS), Cat.
no. 554656], containing the appropriate amount of Mouse BD Fc Block™ purified anti-mouse CD16/CD32 mAb 2.4G2 (Cat.
no. 553141/553142), at a concentration of approximately 10^6 cells per 50 µl. Incubate 10 minutes at 4°C. Add ~1 × 10^6 cells
per staining tube (eg, 12 x 75 mm tube, BD Falcon™ Cat. no. 352008).
4. Add 50 µl FACS buffer containing the optimal per test amount of the staining cocktail to each sample, plus any other cell-surface
marker-specific antibodies to be used. Incubate 60 minutes at 4°C.
5. Wash cells 2× with 2 ml FACS buffer, centrifuge for 5 minutes at 250 × g, and discard supernatant. Resuspend cell pellet in approximately
0.5 ml staining buffer in a tube appropriate for the flow cytometer.
Protocol 3: Alternative: Immunofluorescent Staining Protocol
1. Resuspend mouse cells in FACS staining buffer [eg, DPBS, 1% FCS, 0.09% NaN3 or BD Pharmingen™ Stain Buffer (FBS), Cat.
no. 554656], containing the appropriate amount of Mouse BD Fc Block™ purified anti-mouse CD16/CD32 mAb 2.4G2 (Cat.
no. 553141/553142), at a concentration of approximately 10^6 cells per 50 µl. Incubate 10 minutes at 4°C. Add ~1 × 10^6 cells per
staining tube (eg, 12 x 75 mm tube, BD Falcon™ Cat. no. 352008).
2. Add 0.25 to 4 µg of peptide-loaded H-2D[b]:Ig protein to cell suspension. Incubate 60 minutes at 4°C.
3. Wash cells 1× with 2 ml FACS buffer, centrifuge for 5 minutes at 250 × g, and aspirate supernatant.
4. Resuspend cells in 100 µl FACS buffer containing appropriately diluted fluorescent secondary reagent. We typically use PE-conjugated
A85-1 mAb (anti-mouse IgG1, Cat. no. 550083). Incubate 30 - 60 minutes at 4°C.
5. Wash cells 2× with 2 ml FACS buffer, centrifuge for 5 minutes at 250 × g, and discard supernatant. Resuspend cell pellet in approximately
0.5 ml staining buffer in a tube appropriate for the flow cytometer.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Companion Products



The H-2D[b]:Ig fusion protein consists of three extracellular major histocompatibility complex (MHC) class I H-2D[b] domains that are fused to the VH regions of mouse IgG1 (see Figure 1). In order for the MHC class I to be functional, ie, capable of binding peptides, ß2 Microglobulin (ß2M) must be present. For this reason, BD™ DimerX consists of recombinant H-2D[b]:Ig fusion protein, supplemented with recombinant ß2M. Recombinant MHC molecules, like the DimerX fusion protein, are useful for studying T-cell function by immunofluorescent staining and flow cytometric analysis of antigen-specific T cells.
The MHC gene locus encodes a group of highly polymorphic, cell-surface proteins that play a broad role in the immune response to protein antigens. MHC molecules function by binding and presenting small antigenic protein fragments to antigen-specific receptors expressed by T cells (TCR). Human (human leukocyte antigen/HLA) and mouse (histocompatibility 2/H-2) MHC molecules are structurally and functionally related proteins that comprise two major classes. Class I MHC molecules consist of two separate polypeptide chains. The class I α chain is an MHC encoded, transmembrane polypeptide containing three extracellular domains: α1, α2, and α3. The second chain consists of a non-MHC encoded polypeptide called ß2M. Since ß2M does not contain a transmembrane domain, it associates with the achain through noncovalent interaction. Functionally, class I MHC molecules can bind peptides derived from intracellular antigens (eg, viral and some bacterial antigens) that are specifically recognized by CD8+ T cells. Class II MHC molecules consist of two different transmembrane proteins that can bind peptide fragments derived from extracellular proteins (eg, bacteria and fungi) and are specifically recognized by CD4+ T cells. TCR recognize both processed peptides bound to MHC, as well as regions of the MHC molecule itself. CD4 and CD8 accessory molecules strengthen formation of the TCR-MHC complex through their interaction with non-polymorphic regions of the MHC molecule.
Development References (7)
-
Dal Porto J, Johansen TE, Catipovic B, et al. A soluble divalent class I major histocompatibility complex molecule inhibits alloreactive T cells at nanomolar concentrations. Proc Natl Acad Sci U S A. 1993; 90(14):6671-6675. (Biology). View Reference
-
Mendel I, Kerlero de Rosbo N, Ben-Nun A. encephalomyelitis in H-2b mice: fine specificity and T cell receptor V beta expression of encephalitogenic T cells. Eur J Immunol. 1995; 25(7):1951-1959. (Biology). View Reference
-
Murali-Krishna K, Altman JD, Suresh M, et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity. 1998; 8(2):177-187. (Biology). View Reference
-
Rammensee HG, Friede T, Stevanoviíc S. MHC ligands and peptide motifs: first listing. Immunogenetics. 1995; 41(4):178-228. (Biology). View Reference
-
Schneck JP, Slansky JE, O'Herrin SM, Greten TF . Monitoring antigen-specific T cells using MHC-Ig dimers. In: Coligan J, Kruisbeek D, Margulies EM, Shevach EM, Strober W, ed. Current Protocols in Immunology. New York: John Wiley & Sons, Inc; 2000:17.2.1-17.2.17.
-
Selin LK, Lin MY, Kraemer KA, et al. Attrition of T cell memory: selective loss of LCMV epitope-specific memory CD8 T cells following infections with heterologous viruses. Immunity. 1999; 11(6):733-742. (Biology). View Reference
-
Sun D, Zhang Y, Wei B, Peiper SC, Shao H, Kaplan HJ. Encephalitogenic activity of truncated myelin oligodendrocyte glycoprotein (MOG) peptides and their recognition by CD8+ MOG-speci®c T cells on oligomeric MHC class I molecules. Int Immunol. 2003; 26(10):2470-2479. (Biology: Flow cytometry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.