Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

Flow cytometric analysis of IL-4 expressed in activated mouse splenocytes. Splenocytes from C57BL/6 mice were enriched for CD4+ T cells by positive selection using Purified NA/LE Rat Anti-Mouse CD4 antibody-coated plates (GK1.5, Cat. No.553726;10 μg/ml) for 1 hr at 4°C. The CD4+ T cells were harvested and stimulated with plate-bound Purified NA/LE Hamster Anti-Mouse CD3e (145-2C11, Cat. No. 553057;10 μg/ml) and soluble Purified NA/LE Hamster Anti-Mouse CD28 (37.51, Cat. No. 553294; 2 μg/ml) antibody and Recombinant Mouse IL-2 (Cat. No. 550069; 10 ng/ml) and IL-4 (Cat. No. 550067; 50 ng/ml) for 2 days. The cells were expanded in IL-2 and IL-4 for 3 days and then washed and stimulated for 4 hrs with PMA (Sigma, Cat. No. P-8139; 5 ng/ml) and ionomycin (Sigma, Cat. No. P-8139; 500 ng) in the presence of BD GolgiPlug™ Protein Transport Inhibitor (Containing Brefeldin A) (Cat. No. 555029). The activated cells were fixed with BD Cytofix™ Fixation Buffer (Cat. No. 554655), permeabilized using the BD Perm/Wash™ Permeabilization Buffer (Cat. No. 554723) and then stained either with a BD Horizon™ PE-CF594 Rat IgG1, κ Isotype Control (Cat. No. 562309, Left Panel) or with the BD Horizon™ PE-CF594 Rat Anti-Mouse IL-4 antibody (Cat. No. 562450, Right Panel). Two-color dot plots showing IL-4 (or Ig isotype control staining) versus autofluorescence were derived from gated events with the forward and light scattering characteristics of intact lymphocytes. Flow cytometry was performed using a BD LSR™ II Flow Cytometry System.
.png)

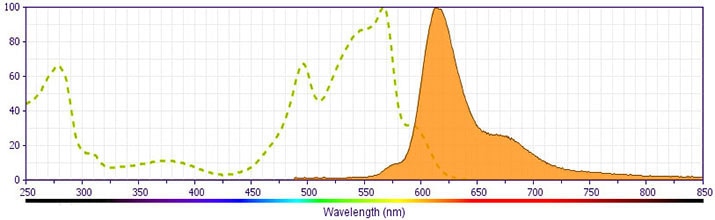
BD Horizon™ PE-CF594 Rat Anti-Mouse IL-4
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Immunofluorescent Staining and Flow Cytometric Analysis: The APC- conjugated 11B11 antibody (Cat. No. 562045/554436) can be used for immunofluorescent staining and flow cytometric analysis to identify and enumerate IL-4 producing cells within mixed cell populations. The use of an isotype control, such as rat IgG1 isotype control, APC-R3-34 (Cat. 554686) is recommended. For specific methodology, please visit our web site, www.bdbiosciences.com,.
BD® CompBeads can be used as surrogates to assess fluorescence spillover (compensation). When fluorochrome conjugated antibodies are bound to BD® CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cells and BD® CompBeads to ensure that BD® CompBeads are appropriate for your specific cellular application.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- An isotype control should be used at the same concentration as the antibody of interest.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Please observe the following precautions: Absorption of visible light can significantly alter the energy transfer occurring in any tandem fluorochrome conjugate; therefore, we recommend that special precautions be taken (such as wrapping vials, tubes, or racks in aluminum foil) to prevent exposure of conjugated reagents, including cells stained with those reagents, to room illumination.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Texas Red is a registered trademark of Molecular Probes, Inc., Eugene, OR.
- CF™ is a trademark of Biotium, Inc.
- When excited by the yellow-green (561-nm) laser, the fluorescence may be brighter than when excited by the blue (488-nm) laser.
- This product is provided under an Agreement between BIOTIUM and BD Biosciences. The manufacture, use, sale, offer for sale, or import of this product is subject to one or more patents or pending applications owned or licensed by Biotium, Inc. This product, and only in the amount purchased by buyer, may be used solely for buyer’s own internal research, in a manner consistent with the accompanying product literature. No other right to use, sell or otherwise transfer (a) this product, or (b) its components is hereby granted expressly, by implication or by estoppel. This product is for research use only. Diagnostic uses require a separate license from Biotium, Inc. For information on purchasing a license to this product including for purposes other than research, contact Biotium, Inc., 3159 Corporate Place, Hayward, CA 94545, Tel: (510) 265-1027. Fax: (510) 265-1352. Email: btinfo@biotium.com.
- Because of the broad absorption spectrum of the tandem fluorochrome, extra care must be taken when using multi-laser cytometers, which may directly excite both PE and CF™594.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
Companion Products



.png?imwidth=320)

Interleukin-4 (IL-4) is a pleiotropic cytokine that has many roles, such as inducing the differentiation of naïve helper T cells (Th0 cells) to Th2 cells, stimulating activated B-cell and T-cell proliferation, and promoting immunoglobulin class switching to IgG1 and IgE in mouse B-cells. IL-4 is expressed by CD4 T-cells, mast cells, basophils and eosinophils. IL-4 was previously known as B-Cell Differentiation Factor (BCDF) or B-cell Stimulatory Factor (BSF1). The 11B11 monoclonal antibody specifically binds to mouse IL-4. The immunogen used to generate the 11B11 hybridoma was partially purified mouse IL-4 prepared from the supernatant of Phorbol 12-Myristate 13-Acetate (PMA)-stimulated EL-4 cells. The 11B11 antibody is reportedly a neutralizing antibody.
Development References (10)
-
Abrams J. Immunoenzymetric assay of mouse and human cytokines using NIP-labeled anti-cytokine antibodies. Curr Protoc Immunol. 2001; 1:6.20-6.21. (Clone-specific: ELISA). View Reference
-
Abrams JS, Roncarolo MG, Yssel H, Andersson U, Gleich GJ, Silver JE. Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples. Immunol Rev. 1992; 127:5-24. (Methodology: Flow cytometry). View Reference
-
Assenmacher M, Schmitz J, Radbruch A. Flow cytometric determination of cytokines in activated murine T helper lymphocytes: expression of interleukin-10 in interferon-gamma and in interleukin-4-expressing cells. Eur J Immunol. 1994; 24(5):1097-1101. (Clone-specific: Flow cytometry). View Reference
-
Haak-Frendscho M, Brown JF, Iizawa Y, Wagner RD, Czuprynski CJ. Administration of anti-IL-4 monoclonal antibody 11B11 increases the resistance of mice to Listeria monocytogenes infection. J Immunol. 1992; 148(12):3978-3985. (Clone-specific: Neutralization). View Reference
-
Lindqvist C, Lundstrom H, Oker-Blom C, Akerman KE. Enhanced IL-4-mediated D10.G4.1 proliferation with suboptimal concentrations of anti-IL-4 receptor monoclonal antibodies. J Immunol. 1993; 150(2):394-398. (Clone-specific: Neutralization). View Reference
-
Ohara J, Paul WE. Production of a monoclonal antibody to and molecular characterization of B-cell stimulatory factor-1. Nature. 1985; 315(6017):333-336. (Immunogen). View Reference
-
Openshaw P, Murphy EE, Hosken NA, et al. Heterogeneity of intracellular cytokine synthesis at the single-cell level in polarized T helper 1 and T helper 2 populations. J Exp Med. 1995; 182(5):1357-1367. (Clone-specific: Flow cytometry). View Reference
-
Sadick MD, Heinzel FP, Holaday BJ, Pu RT, Dawkins RS, Locksley RM. Cure of murine leishmaniasis with anti-interleukin 4 monoclonal antibody. Evidence for a T cell-dependent, interferon gamma-independent mechanism. J Exp Med. 1990; 171(1):115-127. (Clone-specific: Neutralization). View Reference
-
Sander B, Hoiden I, Andersson U, Moller E, Abrams JS. Similar frequencies and kinetics of cytokine producing cells in murine peripheral blood and spleen. Cytokine detection by immunoassay and intracellular immunostaining. J Immunol Methods. 1993; 166(2):201-214. (Clone-specific: ELISA, Flow cytometry). View Reference
-
Swain SL, Weinberg AD, English M, Huston G. IL-4 directs the development of Th2-like helper effectors. J Immunol. 1990; 145(11):3796-3806. (Clone-specific: Neutralization). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.