Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




Flow cytometric analysis of CD14 expression on J774A.1 cells. J774A.1 cells (Mouse monocyte/macrophage cells; ATCC TIB-67) were stained either with a APC-Cy7 Rat IgG1, κ isotype control (shaded) or with the APC-Cy7 Rat Anti-Mouse CD14 antibody (unshaded). Histograms were derived from gated events with the forward and side light scatter characteristics of viable J774A.1 cells. Flow cytometry was performed on a BD™ LSR II flow cytometry system.


BD Pharmingen™ APC-Cy™7 Rat Anti-Mouse CD14

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Flow Cytometry: Investigators should note that Purified Rat Anti-Mouse CD16/CD32 (Mouse BD Fc Block™) (Cat. Nos. 553141/553142) and antibodies of the rat IgG2b isotype may potentially interfere with the reactivity of the APC-Cy™7 Rat Anti-Mouse CD14 antibody (clone rmC5-3) in a concentration-dependent manner. For alternative methods for inhibition of non-specific reactivity, investigators may find the use of purified mouse IgG at a 10-100-fold excess to be more appropriate.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Warning: Some APC-Cy7 and PE-Cy7 conjugates show changes in their emission spectrum with prolonged exposure to formaldehyde. If you are unable to analyze fixed samples within four hours, we recommend that you use BD™ Stabilizing Fixative (Cat. No. 338036).
- Please observe the following precautions: Absorption of visible light can significantly alter the energy transfer occurring in any tandem fluorochrome conjugate; therefore, we recommend that special precautions be taken (such as wrapping vials, tubes, or racks in aluminum foil) to prevent exposure of conjugated reagents, including cells stained with those reagents, to room illumination.
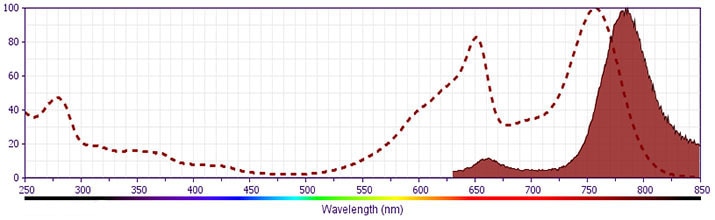
- APC-Cy7 tandem fluorochrome emission is collected in a detector for fluorescence wavelengths of 750 nm and higher.
- APC-Cy7 is a tandem fluorochrome composed of Allophycocyanin (APC), which is excited by laser lines between 595 and 647 nm and serves as an energy donor, coupled to the cyanine dye Cy7™, which acts as an energy acceptor and fluoresces at 780 nm. BD Biosciences Pharmingen has maximized the fluorochrome energy transfer in APC-Cy7, thus maximizing its fluorescence emission intensity, minimizing residual emission from APC, and minimizing required electronic compensation in multilaser-laser flow cytometry systems. Note: Although every effort is made to minimize the lot-to-lot variation in residual emission from APC, it is strongly recommended that every lot be tested for differences in the amount of compensation required and that individual compensation controls are run for each APC-Cy7 conjugate.
- Cy is a trademark of GE Healthcare.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- An isotype control should be used at the same concentration as the antibody of interest.
Companion Products
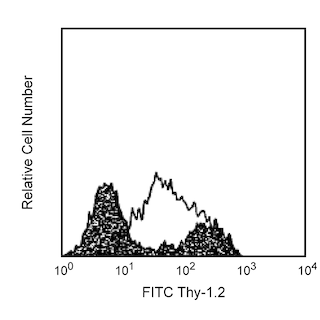

The rmC5-3 monoclonal antibody specifically binds to residues 308-322 of the hydrophilic region of mouse CD14. CD14 is a 53-55 kDa glycophosphatidylinositol (GPI)-linked glycoprotein belonging to the leucine-rich glycoprotein repeat superfamily of cell-surface proteins. It is a receptor for the complex of lipopolysaccharide (LPS or endotoxin, from gram-negative bacteria) with LPS-binding protein (LBP, a plasma protein). CD14 serves as a receptor for LPS that can play a role in the cellular production of proinflammatory cytokines such as IL-1 and TNF. CD14 can be involved in the development of endotoxic shock and LPS-stimulated bone resorption, and promotes, possibly indirectly, bacterial dissemination. Flow cytometric analysis demonstrates that rmC5-3 antibody stains J774A.1 (mouse macrophage line), WEHI-265.1 (mouse monocytic line), peritoneal resident macrophages, Kupffer cells, and cultured bone marrow-derived macrophages and dendritic cells, but not unstimulated splenic macrophages, dendritic cells, neutrophils, or blood monocytes. This staining pattern is similar to that of the alternate anti-mouse CD14 mAb 4C1/CD14, which recognizes a different CD14 epitope, and differs from that of the human, where CD14 expression is characteristic of circulating monocytes and neutrophils. Therefore, data suggests that CD14 expression by leukocyte populations may differ in mice and humans. Peritoneal cells from naive mice, 3-day thioglycollate-elicited peritoneal exudate, as well as 4-hour LPS-activated peritoneal cells, contain a population of Mac-1 (CD11b)-high cells which double-stain with rmC5-3 antibody. Levels of CD14 expression on Kupffer cells and bone marrow-derived macrophages and dendritic cells of LPS-sensitive mice are increased by in vivo and in vitro LPS treatments, an effect which may be mediated by TNF. Preliminary evidence suggests that CD14 may be up-regulated on mouse blood neutrophils. In agreement with the observations that CD14 is shed from activated human and mouse monocytes, rmC5-3 mAb detects soluble CD14 in the serum of LPS-treated mice in a time-dependent manner.
Development References (18)
-
Adachi Y, Satokawa C, Saeki M, et al. Inhibition by a CD14 monoclonal antibody of lipopolysaccharide binding to murine macrophages. J Endotoxin Res. 1999; 5(3):139-146. (Biology).
-
Akashi S, Saitoh S, Wakabayashi Y, et al. Lipopolysaccharide interaction with cell surface Toll-like receptor 4-MD-2: higher affinity than that with MD-2 or CD14. J Exp Med. 2003; 198(7):1035-1042. (Biology). View Reference
-
Fearns C, Kravchenko VV, Ulevitch RJ, Loskutoff DJ. Murine CD14 gene expression in vivo: extramyeloid synthesis and regulation by lipopolysaccharide. J Exp Med. 1995; 181(3):857-866. (Biology). View Reference
-
Fearns C, Loskutoff DJ. Role of tumor necrosis factor alpha in induction of murine CD14 gene expression by lipopolysaccharide. Infect Immun. 1997; 65(11):4822-4831. (Biology). View Reference
-
Ferrero E, Hsieh CL, Francke U, Goyert SM. CD14 is a member of the family of leucine-rich proteins and is encoded by a gene syntenic with multiple receptor genes. J Immunol. 1990; 145(1):331-336. (Biology). View Reference
-
Haziot A, Ferrero E, Kontgen F, et al. Resistance to endotoxin shock and reduced dissemination of gram-negative bacteria in CD14-deficient mice. Immunity. 1996; 4(4):407-414. (Biology). View Reference
-
Le Roy D, Di Padova F, Adachi Y, Glauser MP, Calandra T, Heumann D. Critical role of lipopolysaccharide-binding protein and CD14 in immune responses against gram-negative bacteria. J Immunol. 2001; 167(5):2759-2765. (Biology). View Reference
-
Mahnke K, Becher P, Ricciardi-Castagnoli P, Luger TA, Schawrz T Grabbe S. CD14 is expressed by subsets of murine dendritic cells and upregulated by lipopolysaccharide. In: Ricciardi-Castagnoli P, ed. Dendritic Cells in Fundamental and Clinical Immunology. New York: Plenum Press; 1997:145-159.
-
Matsuura K, Ishida T, Setoguchi M, Higuchi Y, Akizuki S, Yamamoto S. Upregulation of mouse CD14 expression in Kupffer cells by lipopolysaccharide. J Exp Med. 1994; 179(5):1671-1676. (Immunogen: Western blot). View Reference
-
Miyata Y, Takeda H, Kitano S, Hanazawa S. Porphyromonas gingivalis lipopolysaccharide-stimulated bone resorption via CD14 is inhibited by broad-spectrum antibiotics. Infect Immun. 1997; 65(9):3513-3519. (Biology). View Reference
-
Nasu N, Yoshida S, Akizuki S, Higuchi Y, Setoguchi M, Yamamoto S. Molecular and physiological properties of murine CD14. Int Immunol. 1991; 3(2):205-213. (Biology). View Reference
-
Pulendran B, Lingappa J, Kennedy MK, et al. Developmental pathways of dendritic cells in vivo: distinct function, phenotype, and localization of dendritic cell subsets in FLT3 ligand-treated mice. J Immunol. 1997; 159(5):2222-2231. (Biology). View Reference
-
Stewart CC. Methods for studying the ontogeny of monnuclear phagocytes. In: Weir DM, Herzenberg LA, Blackwell C, ed. Weir's Handbook of Experiemental Immunology. Blackwell Science Publications; 1986:44.1-44.17.
-
Takakuwa T, Knopf HP, Sing A, Carsetti R, Galanos C, Freudenberg MA. Induction of CD14 expression in Lpsn, Lpsd and tumor necrosis factor receptor-deficient mice. Eur J Immunol. 1996; 26(11):2686-2692. (Biology). View Reference
-
Takamatsu S, Nakashima I, Nakano K. Modulation of endotoxin-induced histamine synthesis by cytokines in mouse bone marrow-derived macrophages. J Immunol. 1996; 156(2):778-785. (Biology). View Reference
-
Takezawa R, Watanabe Y, Akaike T. Direct evidence of macrophage differentiation from bone marrow cells in the liver: a possible origin of Kupffer cells. J Biochem (Tokyo). 1995; 118(6):1175-1183. (Biology). View Reference
-
Tasaka S, Ishizaka A, Yamada W, et al. Effect of CD14 blockade on endotoxin-induced acute lung injury in mice. Am J Respir Cell Mol Biol. 2003; 29(2):252-258. (Biology). View Reference
-
Ziegler-Heitbrock HW. Heterogeneity of human blood monocytes: the CD14+ CD16+ subpopulation. Immunol Today. 1996; 17(9):424-428. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.