Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
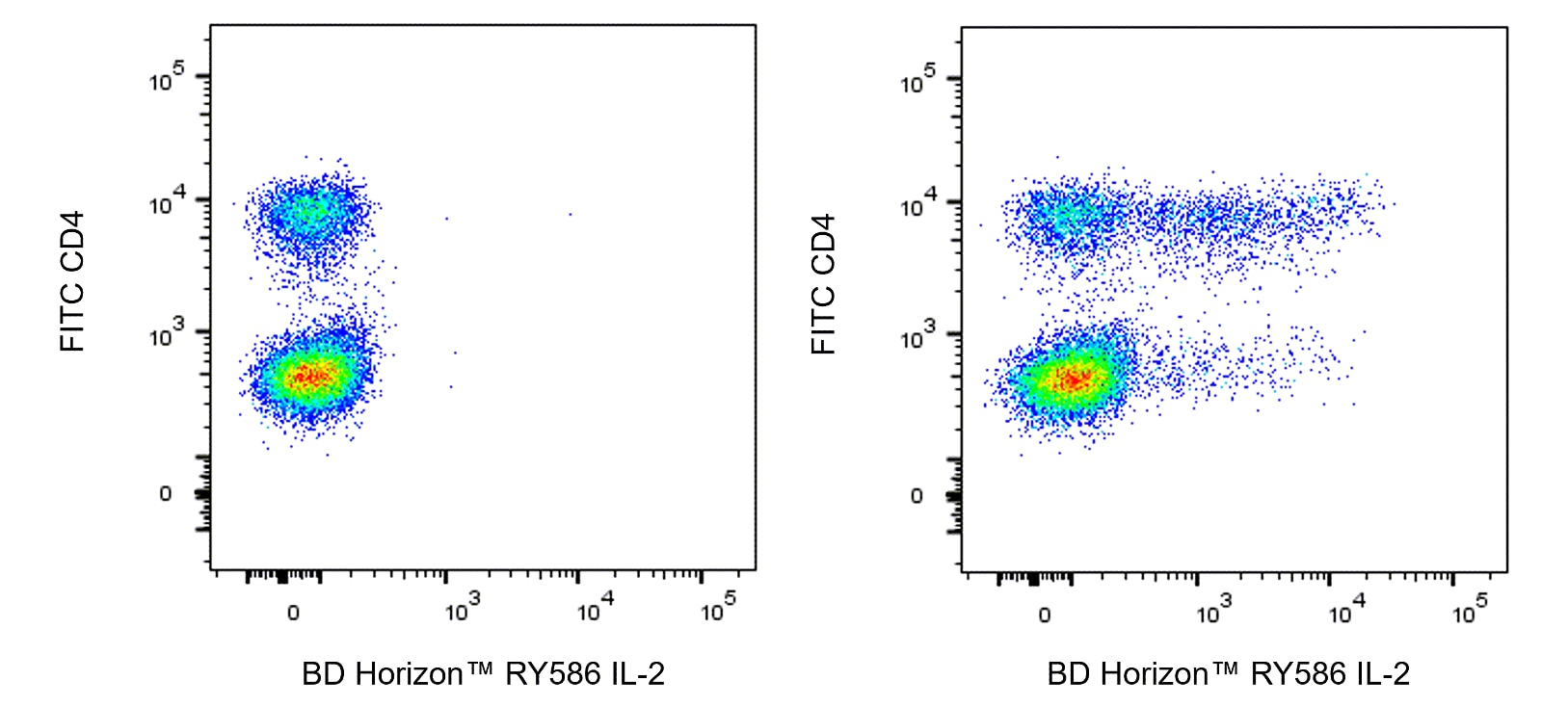



Two color flow cytometric analysis of IL-2 expression in Stimulated Mouse splenic leucocytes. BALB/c Mouse splenocytes were stimulated for 5 hours with Phorbol 12-Myristate 13-Acetate (PMA; Sigma P-8139; 50 ng/ml) and Ionomycin (Sigma I-0634; 1 μg/ml) in the presence of BD GolgiStop™ Protein Transport Inhibitor (containing Monensin) [Cat. No. 554724]. The cells were fixed with BD Cytofix™ Fixation Buffer (Cat. No. 554655), washed, permeabilized and stained in BD Perm/Wash™ Buffer (Cat. No. 554723) with FITC Rat Anti-Mouse CD4 antibody (Cat. No. 553046/553047) and with either BD Horizon™ RY586 Rat IgG2b, κ Isotype Control (Cat. No. 568160; Left Plot) or BD Horizon™ RY586 Rat Anti-Mouse IL-2 antibody (Cat. No. 568543/568544; Right Plot) at 0.125 µg/test using the BD Biosciences Intracellular Cytokine Staining protocol. The bivariate pseudocolor density plot showing the correlated expression of IL-2 (or Ig Isotype control staining) versus CD4 was derived from gated events with the forward and side-light scatter characteristics of intact stimulated leucocytes. Flow cytometry and data analysis were performed using a BD LSRFortessa™ X-20 Cell Analyzer System and FlowJo™ software. Data shown on this Technical Data Sheet are not lot specific.


BD Horizon™ RY586 Rat Anti-Mouse IL-2

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
BD® CompBeads can be used as surrogates to assess fluorescence spillover (compensation). When fluorochrome conjugated antibodies are bound to BD® CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cells and BD® CompBeads to ensure that BD® CompBeads are appropriate for your specific cellular application.
Product Notices
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- An isotype control should be used at the same concentration as the antibody of interest.
- CF™ is a trademark of Biotium, Inc.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- For U.S. patents that may apply, see bd.com/patents.
Companion Products






The JES6-5H4 monoclonal antibody specifically binds to mouse interleukin-2 (IL-2), a multifunctional cytokine that plays pivotal roles in immunity and tolerance. It is produced by activated T cells and affects the activation, growth, proliferation and/or differentiation of various cell types including T and B lymphocytes and their precursors, LAK cells, NK cells, and monocytes/macrophages. IL-2 mediates its biological activities by binding to IL-2 receptor complexes. The intermediate affinity IL-2R is comprised of IL-2Rβ (CD122) and common gamma chain (γc; CD132) subunits, whereas the high-affinity IL-2R is comprised of IL-2Rα (CD25), IL-2Rβ, and γc subunits. The JES6-5H4 monoclonal antibody binds to IL-2 and neutralizes its biological activity.
Development References (5)
-
Abrams J. Immunoenzymetric assay of mouse and human cytokines using NIP-labeled anti-cytokine antibodies. Curr Protoc Immunol. 2001; 1:6.20-6.21. (Clone-specific: ELISA). View Reference
-
Abrams JS, Roncarolo MG, Yssel H, Andersson U, Gleich GJ, Silver JE. Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples. Immunol Rev. 1992; 127:5-24. (Clone-specific: ELISA, Immunoprecipitation). View Reference
-
Karulin AY, Hesse MD, Tary-Lehmann M, Lehmann PV. Single-cytokine-producing CD4 memory cells predominate in type 1 and type 2 immunity.. J Immunol. 2000; 164(4):1862-72. (Clone-specific: Flow cytometry). View Reference
-
Prussin C, Metcalfe DD. Detection of intracytoplasmic cytokine using flow cytometry and directly conjugated anti-cytokine antibodies. J Immunol Methods. 1995; 188(1):117-128. (Methodology: Flow cytometry). View Reference
-
Sander B, Hoiden I, Andersson U, Moller E, Abrams JS. Similar frequencies and kinetics of cytokine producing cells in murine peripheral blood and spleen. Cytokine detection by immunoassay and intracellular immunostaining. J Immunol Methods. 1993; 166(2):201-214. (Clone-specific: ELISA). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.