Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




Analysis of cleaved PARP in activated human T leukemia cells. Jurkat cells were either treated with camptothecin (right panel) or untreated (left panel). The cells were fixed and permeabilized, then stained with PE Mouse anti-Cleaved PARP according to the protocol included in this data sheet. Flow cytometry was performed on a BD™ FACSCalibur flow cytometry system.


BD Pharmingen™ PE Mouse Anti-Cleaved PARP (Asp214)

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Camptothecin (an extract of the Chinese tree Camptotheca acuminata) is a potent inhibitor of topoisomerase I, a molecule required for DNA synthesis. Camptothecin has been shown to induce apoptosis in a dose dependent manner in vitro. Camptothecin is used at BD Biosciences Pharmingen as a general method for inducing apoptosis.
Materials
-1.0 µM stock solution of camptothecin (Sigma; Cat. No. C-9911) in DMSO.
-Jurkat cell line (ATCC TIB-152), proliferating, at 1 x 10^6 cells/ml.
-Either Cytofix/Cytoperm™ Fixation/Permeablization Kit (Cat. No. 554714) or Cytofix/Cytoperm™ solution (Cat. No. 554722) plus Perm/Wash™ buffer (Cat. No. 554723).
Procedure
1. Add camptothecin (4-6 mM final concentration) per 1 x 10^6 proliferating Jurkat cells. If desired, a control aliquot of untreated cells should also be prepared.
2. Incubate the cells for 4 hours at 37°C.
3. Wash the cells (camptothecin-treated and control aliquots) twice with cold PBS; then resuspend them in Cytofix/Cytoperm™ solution at 2 x 10^6 cells/ml.
4. Incubate the cells for 20 minutes on ice.
5. Pellet the cells, and aspirate and discard the Cytofix/Cytoperm™ solution.
6. Wash the cells twice at room temperature with 0.5 ml Perm/Wash™ buffer per 1 x 10^6 cells, and discard the supernatants.
7. Resuspend the cells in Perm/Wash™ buffer at 10 x 10^6 /ml.
8. Aliquot test samples of 1 x 10^6 cells per 100-µl test.
9. Add 20 µl antibody per test, and incubate for 30 minutes at room temperature.
10. Wash each test in 1.0 ml Perm/Wash™ Buffer and discard the supernatant.
11. Resuspend each test in 0.5 ml Perm/Wash™ Buffer and analyze by flow cytometry.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
Companion Products




PARP (Poly [ADP-Ribose] Polymerase) is a 113-kDa nuclear chromatin-associated enzyme that catalyzes the transfer of ADP-ribose units from NAD+ to a variety of nuclear proteins including topoisomerases, histones, and PARP itself. The catalytic activity of PARP is increased in cells following DNA damage, and PARP is thought to play an important role in mediating the normal cellular response to DNA damage. Additionally, PARP is a target of the caspase protease activity associated with apoptosis. The PARP protein consists of an N-terminal DNA-binding domain (DBD) and a C-terminal catalytic domain separated by a central automodification domain. During apoptosis, Caspase-3 cleaves PARP at a recognition site (Asp Glu Val Asp Gly) in the DBD to form 24- and 89-kDa fragments. This process separates the DBD (which is mostly in the 24-kDa fragment) from the catalytic domain (in the 89-kDa fragment) of the enzyme, resulting in the loss of normal PARP function. It has been proposed that inactivation of PARP directs DNA-damaged cells to undergo apoptosis rather than necrotic degradation, and the presence of the 89-kDa PARP cleavage fraction is considered to be a marker of apoptosis.
A peptide corresponding to the N-terminus of the cleavage site (Asp 214) of human PARP was used as the immunogen. The F21-852 monoclonal antibody reacts only with the 89-kDa fragment of human PARP-1 that is downstream of the Caspase-3 cleavage site (Asp214) and contains the automodification and catalytic domains. It does not react with intact human PARP-1. Cross-reactivity with other members of the PARP superfamily is unknown. Recognition of cleaved PARP in mouse cells has been demonstrated, and it may also cross-react with a number of other species due to the conserved nature of the molecule.
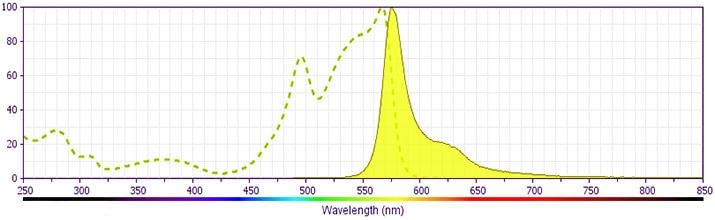
Development References (5)
-
Amé J-C, Spenlehauer C, de Murcia G. The PARP superfamily. Bioessays. 2004; 26:882-893. (Biology).
-
Boulares AH, Yakovlev AG, Ivanova V, et al. Role of Poly(ADP-ribose) polymerase (PARP) cleavage in apoptosis. Caspase 3-resistant PARP mutant increases rates of apoptosis in transfected cells. J Biol Chem. 1999; 274(33):22932-22940. (Biology).
-
Cherney BW, McBride OW, Chen D, et al. cDNA sequence, protein structure, and chromosomal location of the human gene for poly(ADP-ribose) polymerase. Proc Natl Acad Sci U S A. 1987; 84(23):8370-8374. (Biology). View Reference
-
D'Amours D, Desnoyers S, D'Silva I, Poirier GG. Poly(ADP-ribosyl)ation reactions in the regulation of nucelar functions. Biochem J. 1999; 342:249-268. (Biology).
-
Soldani G, Scovassi AI. Poly(ADP-ribose) polymerase-1 cleavage during apoptosis: an update. Apoptosis. 2002; 7:321-328. (Biology).
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.