-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
-
- BD® OMICS-Guard Sample Preservation Buffer
- BD® AbSeq Assay
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ ATAC-Seq Assays
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Accessory Kits
- BD® OMICS-One Protein Panels
- Poland (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from United States.
Would you like to stay on the current country site or be switched to your country?
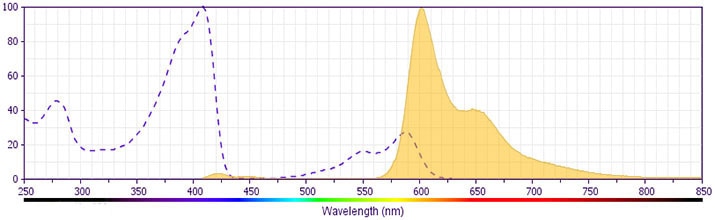
BD® CD11b (D12) BV605
Clone D12 (CE_IVD)


Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
The antibody reagent is stable until the expiration date shown on the label when stored at 2° to 8°C. Do not use after the expiration date. Do not freeze the reagent or expose it to direct light during storage or incubation with cells. Keep the outside of the reagent vial dry.Do not use the reagent if you observe any change in appearance. Precipitation or discoloration indicates instability or deterioration.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- Human donor specific background has been observed in relation to the presence of anti-polyethylene glycol (PEG) antibodies, developed as a result of certain vaccines containing PEG, including some COVID-19 vaccines. We recommend use of BD Horizon Brilliant™ Stain Buffer in your experiments to help mitigate potential background. For more information visit https://www.bdbiosciences.com/en-us/support/product-notices.
Data Sheets
Recently Viewed
Development References (20)
-
Lanier LL, Phillips JH. A map of the cell surface antigens expressed on resting and activated human natural killer cells. In: Reinherz EL. Ellis L. Reinherz .. et al., ed. Leukocyte typing II. New York: Springer-Verlag; 1986:157-170.
-
Aires A, Teixeira MDA, Lau C, et al. A pilot study on the usefulness of peripheral blood flow cytometry for the diagnosis of lower risk myelodysplastic syndromes: the "MDS thermometer".. BMC Hematol. 2018; 18:6. View Reference
-
Amirghofran Z, Zakerinia M, Shamseddin A. Significant association between expression of the CD11b surface molecule and favorable outcome for patients with acute myeloblastic leukemia.. Int J Hematol. 2001; 73(4):502-506. View Reference
-
Bernstein ID, Self S. Joint report of the Myeloid Section of the Second International Workshop on Human Leukocyte Differentiation Antigens. In: Reinherz EL, Haynes BF, Nadler LM, Bernstein ID, ed. Leukocyte Typing II: Human Myeloid and Hematopoietic Cells. New York, NY: Springer-Verlag; 1986:1-25.
-
Centers for Disease Control and Prevention. 2007 Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings. Available: https://www.cdc.gov/infectioncontrol/guidelines/isolation/index.html March 12, 2019. View Reference
-
Churchill HRO, Fuda FS, Xu J, et al. Leukocyte immunoglobulin-like receptor B1 and B4 (LILRB1 and LILRB4): Highly sensitive and specific markers of acute myeloid leukemia with monocytic differentiation.. Cytometry B Clin Cytom. 2021; 100(4):476-487. View Reference
-
Clinical and Laboratory Standards Institute. Clinical Flow Cytometric Analysis of Neoplastic Hematolymphoid Cells. In: CLSI. CLSI, ed. CLSI document H43-A2. Wayne, PA: Clinical and Laboratory Standards Institute; 2007:1-81. View Reference
-
Clinical and Laboratory Standards Institute. Collection of Diagnostic Venous Blood Specimens, 7th ed. In: CLSI. CLSI, ed. CLSI document GP41-A7. Wayne, PA: Clinical and Laboratory Standards Institute; 2017:1-85. View Reference
-
Clinical and Laboratory Standards Institute. Interference Testing in Clinical Chemistry: Approved Guidelines. Second Edition. In: CLSI. CLSI, ed. CLSI document EP07-A2. Wayne, PA: Clinical and Laboratory Standards Institute; 2005:1-12.
-
Haycocks NG, Lawrence L, Cain JW, Zhao XF. Optimizing antibody panels for efficient and cost-effective flow cytometric diagnosis of acute leukemia.. Cytometry B Clin Cytom. 2011; 80(4):221-9. View Reference
-
Jackson AL, Warner NL. Preparation, staining, and analysis by flow cytometry of peripheral blood leukocytes. In: Rose NR, Friedman H, Fahey JL, ed. Manual of Clincial Laboratory Immunology, Third Edition. Washington DC: American Society for Microbiology; 1986:226-235.
-
Kroll MH. Evaluating interference caused by lipemia.. Clin Chem. 2004; 50(11):1968-9. View Reference
-
Nikolac N. Lipemia: causes, interference mechanisms, detection and management.. Biochem Med (Zagreb). 2014; 24(1):57-67. View Reference
-
Repo H, Jansson SE, Leirisalo-Repo M. Anticoagulant selection influences flow cytometric determination of CD11b upregulation in vivo and ex vivo.. J Immunol Methods. 1995; 185(1):65-79. View Reference
-
Rizzatti EG, Garcia AB, Portieres FL, Silva DE, Martins SL, Falcão RP. Expression of CD117 and CD11b in bone marrow can differentiate acute promyelocytic leukemia from recovering benign myeloid proliferation.. Am J Clin Pathol. 2002; 118(1):31-7. View Reference
-
Rothe G, Schmitz G. Consensus protocol for the flow cytometric immunophenotyping of hematopoietic malignancies. Leukemia. 1996; 10:877-895.
-
Seegmiller AC, Kroft SH, Karandikar NJ, McKenna RW. Characterization of immunophenotypic aberrancies in 200 cases of B acute lymphoblastic leukemia.. Am J Clin Pathol. 2009; 132(6):940-9. View Reference
-
Shalekoff S, Page-Shipp L, Tiemessen CT. Effects of anticoagulants and temperature on expression of activation markers CD11b and HLA-DR on human leukocytes.. Clin Diagn Lab Immunol. 1998; 5(5):695-702. View Reference
-
Stelzer GT, Marti G, Hurley A, McCoy PJ, Lovett EJ, Schwartz A. US-Canadian consensus recommendations on the immunophenotypic analysis of hematologic neoplasia by flow cytometry: standardization and validation of laboratory procedures. Cytometry. 1997; 30:214-230.
-
Zola H. CD10 Workshop Panel report. In: Schlossman SF. Stuart F. Schlossman .. et al., ed. Leucocyte typing V : white cell differentiation antigens : proceedings of the fifth international workshop and conference held in Boston, USA, 3-7 November, 1993. Oxford: Oxford University Press; 1995:505-507.
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
![]() Documents are subject to revision without notice. Please verify you have the correct revision of the document, and always refer back to BD's eIFU website for the latest and most up to date information.
Documents are subject to revision without notice. Please verify you have the correct revision of the document, and always refer back to BD's eIFU website for the latest and most up to date information.

