Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?





Analysis of Stat5 (pY694) in in human peripheral blood lymphocytes. Human peripheral blood mononuclear cells (PBMC) were either left untreated (unshaded) or treated (shaded) with 100 ng/ml (final concentration) of BD Pharmingen™ Recombinant Human IL-2 (Cat. No. 554603) for 15 minutes at 37°C. The PBMC were fixed with BD Cytofix™ Fixation Buffer (Cat. No. 554655) for 10 minutes at 37°C, permeabilized (BD Phosflow™ Perm Buffer III, Cat. No. 558050) on ice for 30 minutes and were then stained with Pacific Blue™ Mouse anti-Stat5 (pY694). For data analysis, lymphocytes were selected by scatter profile. Flow cytometry was performed on a BD FACSCanto™ II flow cytometry system.



BD™ Phosflow Pacific Blue™ Mouse anti-Stat5 (pY694)

BD™ Phosflow Pacific Blue™ Mouse anti-Stat5 (pY694)

监管状态图例
未经BD明确书面授权,严禁使用未经许可的任何商品。
准备和存储
推荐的实验流程
This antibody conjugate is suitable for intracellular staining of human whole blood (using BD Phosflow™ Lyse/Fix Buffer) and peripheral blood mononuclear cells and cell lines (using BD Cytofix™ Fixation Buffer).
商品通知
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
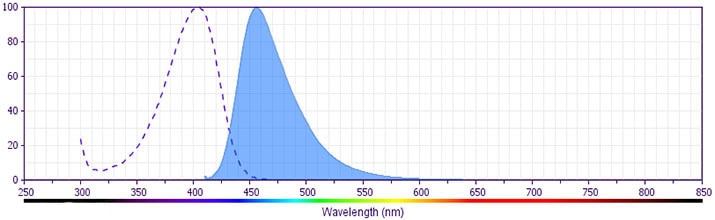
- Pacific Blue™ has a maximum absorption of 416 nm and maximum emission of 451 nm. Before staining with this reagent, please confirm that your flow cytometer is capable of exciting the fluorochrome and discriminating the resulting fluorescence.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Pacific Blue™ is a trademark of Molecular Probes, Inc., Eugene, OR.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
配套商品

Stat (Signal transducer and activators of transcription) proteins mediate the biological activity of cytokines, including interleukins, interferons, erythropoietin, and growth factors. Ligand-receptor interaction activates constitutively-associated JAK family kinases and subsequent recruitment/activation of Stat proteins by tyrosine phosphorylation. Active Stat proteins then move to the nucleus to promote transcription of cytokine-inducible genes. Seven Stat proteins have been cloned, each of which is differentially expressed and/or activated in a cytokine-specific and cell type-specific manner. Stat5 has been characterized and shown to be encoded by two separate genes, Stat5a and Stat5b, which share over 90% identity at the amino acid level. Stat5a has been shown to be involved in lactogenesis and mammary development, while Stat5b has been shown to be involved in growth hormone signaling and liver gene expression. Both Stat5a and Stat5b are involved in IL-2 induced peripheral T cell proliferation. The peptide hormone, prolactin, binds to the prolactin receptor (PRLR) to initiate the lactogenic response. There are at least three forms of PRLR; however, only the long form activates the 92-kDa Stat5 protein by inducing phosphorylation at Y694. Once phosphorylated, Stat5 becomes an essential transcription factor which binds to the β-casein gene promoter. The presence of an SH2 domain within Stat5 suggests that it may directly interact with protein tyrosine kinases (PTKs) such as JAK2.
The 47 monoclonal antibody recognizes the phosphorylated Y694 of Stat5a. The homologous phosphorylation site in Stat5b is Y699.
研发参考 (4)
-
Bromberg J, Darnell JE. The role of STATs in transcriptional control and their impact on cellular function. Oncogene. 2000; 19(21):2468-2473. (Biology). 查看参考
-
Gouilleux F, Wakao H, Mundt M, Groner B. Prolactin induces phosphorylation of Tyr694 of Stat5 (MGF), a prerequisite for DNA binding and induction of transcription. EMBO J. 1994; 13(18):4361-4369. (Biology). 查看参考
-
Imada K, Leonard WJ. The Jak-STAT pathway. Mol Immunol. 2000; 37:1-11. (Biology). 查看参考
-
Riou C, Yassine-Diab B, Van grevenynghe J, et al. Convergence of TCR and cytokine signaling leads to FOXO3a phosphorylation and drives the survival of CD4+ central memory T cells.. J Exp Med. 2007; 204(1):79-91. (Clone-specific). 查看参考
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.