-
Training
- Flow Cytometry Basic Training
-
Product-Based Training
- BD FACSDiscover™ S8 Cell Sorter Product Training
- Accuri C6 Plus Product-Based Training
- FACSAria Product Based Training
- FACSCanto Product-Based Training
- FACSLyric Product-Based Training
- FACSMelody Product-Based Training
- FACSymphony Product-Based Training
- HTS Product-Based Training
- LSRFortessa Product-Based Training
- Advanced Training
-
- BD FACSDiscover™ S8 Cell Sorter Product Training
- Accuri C6 Plus Product-Based Training
- FACSAria Product Based Training
- FACSCanto Product-Based Training
- FACSLyric Product-Based Training
- FACSMelody Product-Based Training
- FACSymphony Product-Based Training
- HTS Product-Based Training
- LSRFortessa Product-Based Training
- United States (English)
-
Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Put all BD® AbSeq Reagents to be pooled into a Latch Rack for 500 µL Tubes (Thermo Fisher Scientific Cat. No. 4900). Arrange the tubes so that they can be easily uncapped and re-capped with an 8-Channel Screw Cap Tube Capper (Thermo Fisher Scientific Cat. No. 4105MAT) and the reagents aliquoted with a multi-channel pipette.
BD® AbSeq tubes should be centrifuged for ≥ 30 seconds at 400 × g to ensure removal of any content in the cap/tube threads prior to the first opening.
Product Notices
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- The production process underwent stringent testing and validation to assure that it generates a high-quality conjugate with consistent performance and specific binding activity. However, verification testing has not been performed on all conjugate lots.
- Illumina is a trademark of Illumina, Inc.
- Please refer to bd.com/genomics-resources for technical protocols.
- This reagent has been pre-diluted for use at the recommended volume per test. Typical use is 2 µl for 1 × 10^6 cells in a 200-µl staining reaction.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- For U.S. patents that may apply, see bd.com/patents.
Companion Products
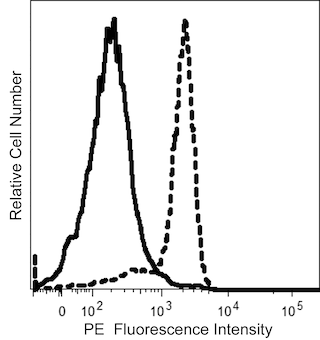



The EMab-134 monoclonal antibody specifically recognizes the epidermal growth factor receptor (EGF-R). The EGF-R is a transmembrane glycoprotein of approximately 170 kDa that is expressed on most cells. It consists of a glycosylated extracellular domain which binds to epidermal growth factor (EGF), a transmembrane domain, and an intracellular domain with tyrosine-kinase activity essential for signal transduction. The epitope recognized by EMab-134 is an extracellular peptide that is not affected by glycosylation. The EGF-R plays an important role in the growth and differentiation of many cellular types. Transforming growth factor α (TGFα), vaccinia virus growth factor, and related growth factors can also bind to and signal through the EGF-R.
Development References (5)
-
Itai S, Yamada S, Kaneko MK, Chang YW, Harada H, Kato Y. Establishment of EMab-134, a Sensitive and Specific Anti-Epidermal Growth Factor Receptor Monoclonal Antibody for Detecting Squamous Cell Carcinoma Cells of the Oral Cavity. Monoclon Antib Immunodiagn Immunother. 2017; 36(6):272-281. (Immunogen: ELISA, Flow cytometry, Immunohistochemistry, Western blot). View Reference
-
Kaneko MK, Yamada S, Itai S, et al. Elucidation of the critical epitope of an anti-EGFR monoclonal antibody EMab-134.. Biochem Biophys Rep. 2018; 14:54-57. (Clone-specific: Flow cytometry, Immunohistochemistry, Western blot). View Reference
-
Kovacs E, Zorn JA, Huang Y, Barros T, Kuriyan J. A structural perspective on the regulation of the epidermal growth factor receptor.. Annu Rev Biochem. 2015; 84:739-64. (Biology). View Reference
-
Leahy DJ. Structure and function of the epidermal growth factor (EGF/ErbB) family of receptors.. Adv Protein Chem. 2004; 68:1-27. (Biology). View Reference
-
Messa C, Russo F, Notarnicola M, Di Leo A. Demonstration of epidermal growth factor receptor in colorectal adenocarcinoma by enzyme immunoassay. Digestion. 1994; 55(2):103-107. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.